1. Identify the electrophilic site in each of the following molecules by selecting e electrophilic atom. 2....
Question:
1. Identify the electrophilic site in each of the following molecules by selecting e electrophilic atom.
 2.
2.
2. For the Bronsted acid-base reaction shown below, determine the conjugate acid-base pairs.
Then give the curved-arrow notation for the reaction in the left-to-right direction. (To draw the arrows, click on the reaction to get into the edit mode, then click on the curved arrow icon.)
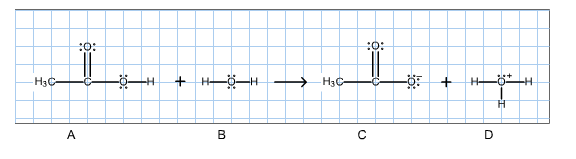
Fill in the blanks with the letters A,B,C and D, representing the species in the reaction above.

For the following reaction, indicate which reactant is the Lewis acid an which is the Lewis base.
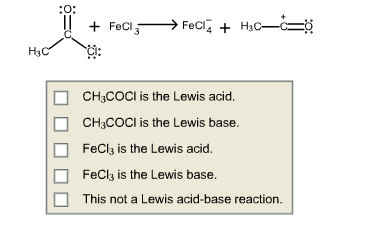
4.
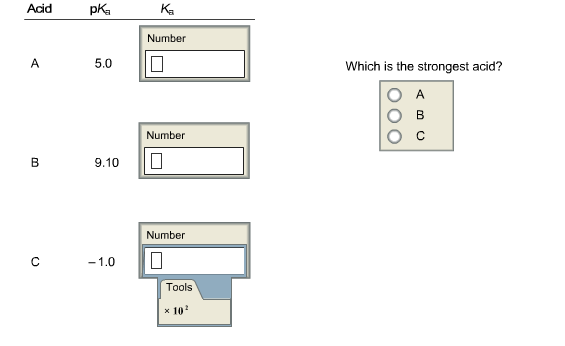
Determine the dissociation constants for the following acids. Express the answers in proper scientific notation where appropriate.
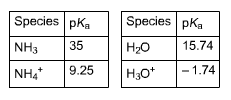
5. Complete the equations for the following equilibria and calculate Kea where the Keq expression includes IF1201. Be sure to enter Keg in proper scientific notation.


Contemporary Business
ISBN: 9781118161739
1st Canadian Edition
Authors: Louis E. Boone, Micheal H. Khan, DAvid L. Kurtz, Brahm M. Canzer




