In order to study the photochemical decay of aqueous bromine in bright sunlight, a small quantity of
Question:
In order to study the photochemical decay of aqueous bromine in bright sunlight, a small quantity of liquid bromine was dissolved in water contained in a glass battery jar and placed in direct sunlight. The following data were obtained at 25?C:
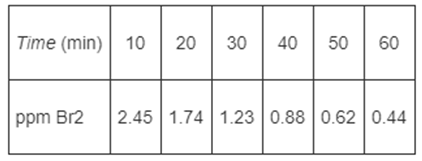
(a) Determine whether the reaction rate is zero, first, or second order in bromine, and calculate the reaction rate constant in units of your choice.
(b) Assuming identical exposure conditions, calculate the required hourly rate of injection of bromine (in pounds per hour) into a sunlit body of water, 25,000 gal in volume, in order to maintain a sterilizing level of bromine of 1.0 ppm.
(c) Apply one or more of the six ideas in Table to this problem. (Note: ppm = parts of bromine per million parts of brominated water by weight. In dilute aqueous solutions, 1 ppm = 1 milligram per liter.) (From California Professional Engineers' Exam.)
Table P-3 Practicing Creative Thinking


Mathematical Statistics with Applications in R
ISBN: 978-0124171138
2nd edition
Authors: Chris P. Tsokos, K.M. Ramachandran




