Question: 1. Shown below is the IR spectrum of eugenol. Highlight the peaks assigned to O-H, C-H a stretching by annotating the spectrum. 100 90
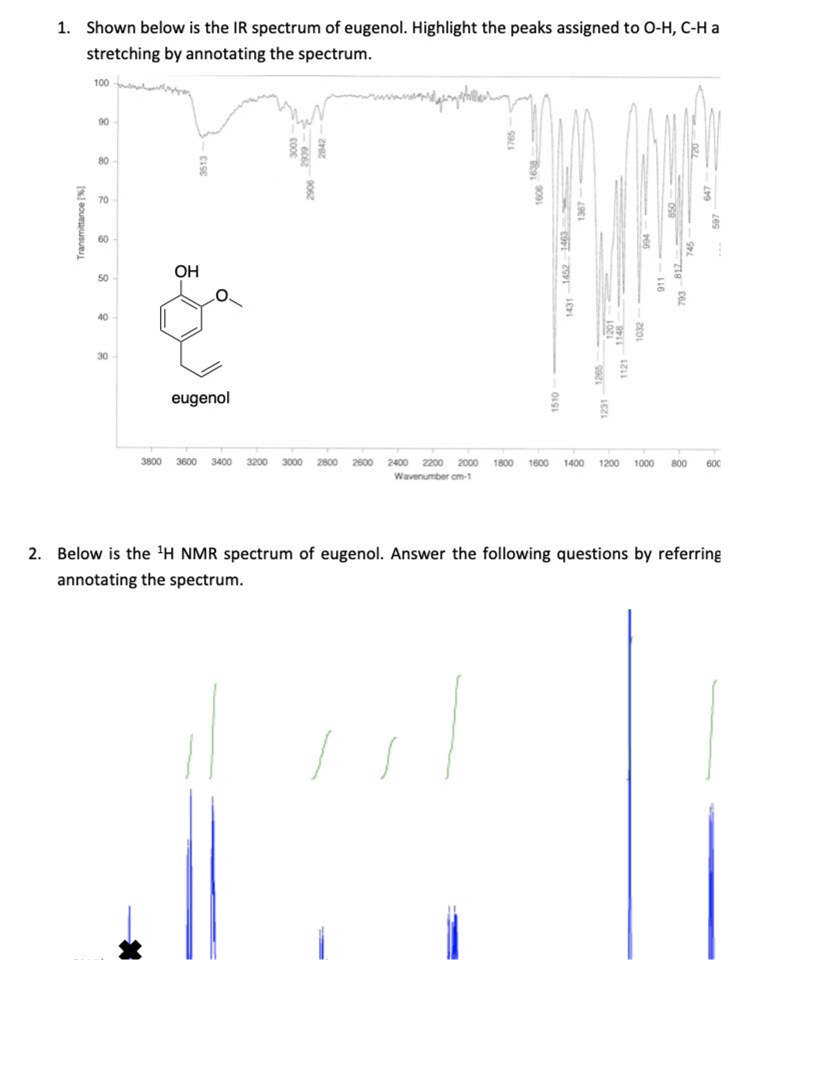

![80 70 60 50 40 30 Transmittance [%] OH eugenol 3800 3600](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/03/62396f67d6dcc_23962396f6784049.jpg)
1. Shown below is the IR spectrum of eugenol. Highlight the peaks assigned to O-H, C-H a stretching by annotating the spectrum. 100 90 80 70 60 50 40 30 Transmittance [%] OH eugenol 3800 3600 3400 3200 2800 1400 1200 1000 800 600 2400 2200 2000 1800 1600 Wavenumber cm-1 2. Below is the H NMR spectrum of eugenol. Answer the following questions by referring annotating the spectrum. 1431-1452 1510- 1231-1265 a. Identify which proton environment causes the peak at 5.51 ppm. b. Identify which proton environment causes the peak at 3.88 ppm. C. Which peaks correspond to the aromatic protons? Indicate them on the spectrum. d. Which peaks correspond to the methylene (CH2) protons between the aromatic ri and the alkene? Indicate them on the spectrum. 3. a. Predict the m/z (mass-to-charge ratio) values of the [M]* and [M+11+ peaks under EIMS for eugenol. b. Assuming 100% abundance of the [M]* ion peak, calculate the relative abundance of the [M+11+ ion peak (C abundance is 1.1%). 4. A fragment is observed at m/z = 149. Predict the formula of this fragment. A typical eukaryotic cell cycle consists of mitosis and fertilization. mitosis and meiosis. O prophase, metaphase, anaphase, and telophase. interphase, mitosis, and cytokinesis. G1, the Sphase, and G2.
Step by Step Solution
3.51 Rating (154 Votes )
There are 3 Steps involved in it
e 6 b H dH alkene bund Hh Hi g 1 ppm a H proton of OH gru show peak at 551 OH grup show broader ... View full answer

Get step-by-step solutions from verified subject matter experts


