Question: QUESTION 9 The Group 8A elements Oare unreactive and are rarely found in combination with other elements. are liquids at room temperature. are good
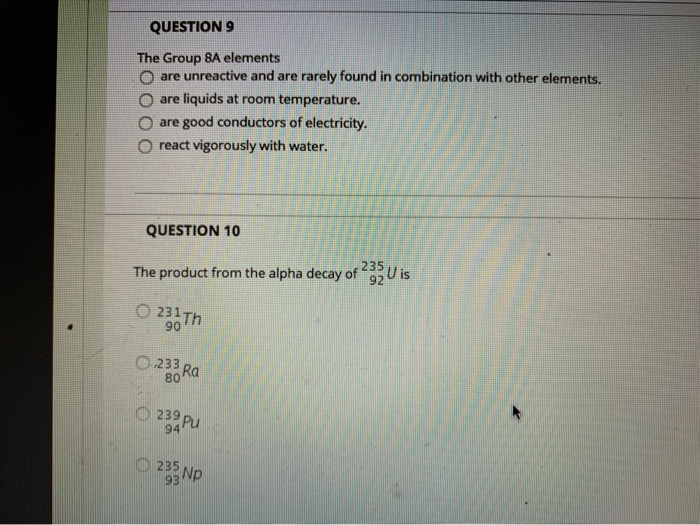

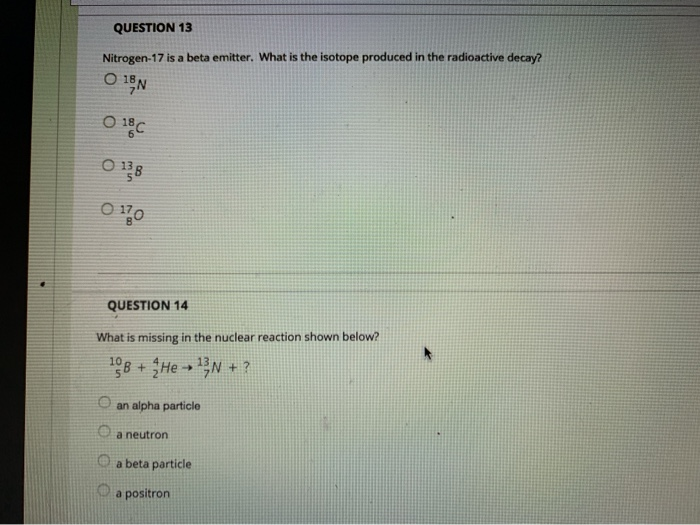

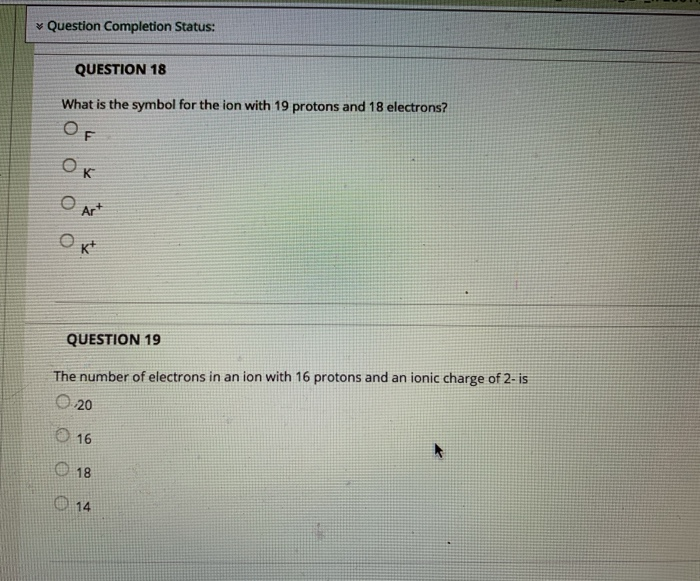
QUESTION 9 The Group 8A elements Oare unreactive and are rarely found in combination with other elements. are liquids at room temperature. are good conductors of electricity. react vigorously with water. QUESTION 10 The product from the alpha decay of O231 Th 90 O233 80 Ra 23 Pu 94 235 93 Np 235 92 U is QUESTION 11 The process in which a nucleus spontaneously breaks down by emitting radiation is known as O a chain reaction O radioactive decay O fusion O fission QUESTION 12 Which is NOT a way to minimize your exposure to radiation? Ostaying a longer time 'wearing a lead apron waring lead-lined gloves keeping a good distance QUESTION 13 Nitrogen-17 is a beta emitter. What is the isotope produced in the radioactive decay? O BN 0 180 0 138 0170 QUESTION 14 What is missing in the nuclear reaction shown below? 8+HeN+? O an alpha particle a neutron a beta particle a positron QUESTION 15 The octet rule indicates that O atoms lose, gain, or share valence electrons to have 8 valence electrons. all of the shells in an atom hold a maximum of 8 electrons. the noble gases react with other compounds to get 8 valence electrons. all of the noble gases have eight total electrons. QUESTION 16 In ionic compounds, O metals; cations metals; anions nonmetals; anions 'metals; polyatomic ions lose their valence electrons to form positively charged. Question Completion Status: QUESTION 18 What is the symbol for the ion with 19 protons and 18 electrons? OF OK O Art Okt QUESTION 19 The number of electrons in an ion with 16 protons and an ionic charge of 2- is 020 16 18 14
Step by Step Solution
3.52 Rating (162 Votes )
There are 3 Steps involved in it
The detailed answer for the above question is provided below 9The group 8 A elements unreactive and rarely found in combination with other elements Th... View full answer

Get step-by-step solutions from verified subject matter experts


