Question: The bond angle between two hybrid orbitals is 105. The percentage of s-character of hybrid orbital is between (A) 50-55% (B) 9-12% (C) 22-23%
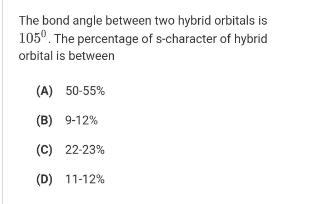
The bond angle between two hybrid orbitals is 105. The percentage of s-character of hybrid orbital is between (A) 50-55% (B) 9-12% (C) 22-23% (D) 11-12%
Step by Step Solution
3.34 Rating (157 Votes )
There are 3 Steps involved in it
Hie f Scharactes of f SCharacter of ... View full answer

Get step-by-step solutions from verified subject matter experts


