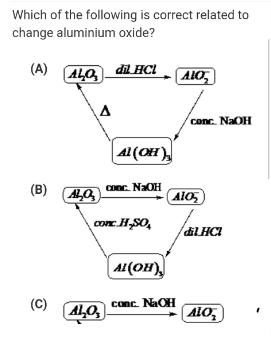
Which of the following is correct related to change aluminium oxide? (A) ALO, di HCI ALO...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Which of the following is correct related to change aluminium oxide? (A) ALO, di HCI ALO conc. NaOH Al (OH), (B) LO Conc. NaOH con H,SO, A1(OH) conc. NaOH ALO, (C) ALO, Which of the following is correct related to change aluminium oxide? (A) ALO, di HCI ALO conc. NaOH Al (OH), (B) LO Conc. NaOH con H,SO, A1(OH) conc. NaOH ALO, (C) ALO,
Expert Answer:
Related Book For 

Financial Accounting
ISBN: 978-1118978085
IFRS 3rd edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Posted Date:
Students also viewed these chemistry questions
-
Which of the following is correct for Smith Company when Smith issues 10,000 shares of $10 par value common stock and pays $29,000 cash in exchange for a building? The market price of the Smith stock...
-
Which of the following is not required by generally accepted auditing standards? a. Written representations. b. Attorney letter. c. Management letter. d. Engagement letter.
-
You should explain and justify your answer a. Why is electric discharge machining a good process for the manufacture of plastic injection mould for a model railway engine body? [10 marks] b. Once a...
-
Is there more to marketing than building product and brand awareness than what people discuss? How do marketing managers and executives manage consistent messaging across cultures?
-
The fraternities and sororities at St. Algebra College have been plagued by declining membership over the past several years and want to know if the incoming freshman class will be a fertile...
-
When preparing a Consolidated Statement of financial position the identifiable non-monetary assets of the subsidiary need to be fair valued. Which of the following assets of the subsidiary need to be...
-
Explain qualitatively why sandwich structures (Figure 1.5) have such high flexural stiffness-to-weight ratios. Describe the key parameters affecting the flexural stiffness-to-weight ratio of a...
-
A technique for cooling a multichip module involves submerging the module in a saturated fluorocarbon liquid. Vapor generated due to boiling at the module surface is condensed on the outer surface of...
-
Create an outline for a training session to help a team avoid phishing. What could be included in that training session? What are some typical signs that an email might be fraudulent?
-
The Gilbert Air-Conditioning Company is considering the purchase of a special shipment of portable air conditioners manufactured in Japan. Each unit will cost Gilbert $80, and it will be sold for...
-
Express the following ratios as ratios of natural logarithms and simplify. log 2 x log 2 x log, a log 3 x a. log 2 x log 3 X log 2 x log 16 X log x a log x a 11 b. log 16 X C. log 9,2 (Use an exact...
-
If Hyatt Corp. has accounts receivables of $654,803 and net sales of $1,932,349, what is its accounts receivable turnover? Round your final answer to two decimal places.
-
Norfolk Inc. is a $1 billion corporation with a December year-end. The Westerly Division has annual sales of $100 million, with annual before-tax operating profit of $12 million. In September, the...
-
Solve the equation. 12x+11-10=-4
-
Consider the matrices A B -2 3 4 2 3 12 05 D 3 2 0 1 1 6 For each of the following expressions, determine if it is defined. If yes, evaluate it. If no, explain why. (d) Find the matrix X that...
-
Problem 4 (6 points) Suppose g(x) is a monic polynomial of even degree d, that is g(x) = xd +bd-1xd1 + +6x+bo ...
-
Calculations Marketing Inc. issued 7.0% bonds with a par value of $370,000 and a five-year life on January 1, 2023, for $385,781. The bonds pay interest on June 30 and December 31. The market...
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
Rees Company experienced a fire on December 31, 2017, in which its financial records were partially destroyed. It has been able to salvage some of the records and has ascertained the following...
-
Below is a series of cost of goods sold sections for companies Alpha, Beta, Chi, and Decca. Instructions Fill in the lettered blanks to complete the cost of goods sold sections. Alpha Beta Chi Decca...
-
On January 1, 2017, Lennon Enterprises acquires 100% of Ono Ltd. for £220,000 in cash. The condensed statements of financial position of the two corporations immediately following the...
-
A crystal sample of bacteriorhodpsin, a light-sensitive protein found in halobacteria that responds to light energy, has crystal planes separated by 0.20 nm. If a beam of 11 keV x rays illuminates a...
-
A nickel crystals work function is measured to be 5.22 eV at 25C. As the temperature increases by 300C, the work function drops by 50 meV. By how much does this shift the threshold wavelength for...
-
Investigators have created structures consisting of linear chains of ionized atoms on a smooth surface. Electrons are restricted to travel along the chain. The energy levels of the electrons match...

Study smarter with the SolutionInn App



