1. 25 cm of iron (II) sulphate reacts with 24.3 cm3 of 0.02M potassium manganate (VII)...
Fantastic news! We've Found the answer you've been seeking!
Question:
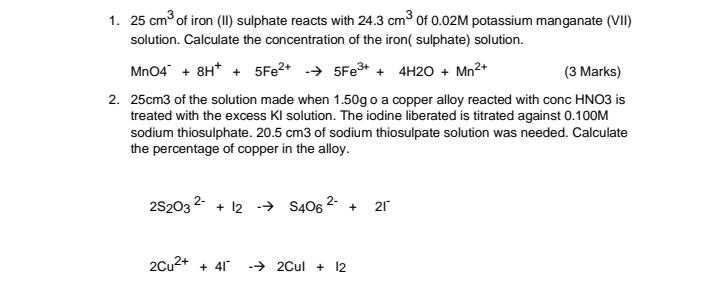
Transcribed Image Text:
1. 25 cm of iron (II) sulphate reacts with 24.3 cm3 of 0.02M potassium manganate (VII) solution. Calculate the concentration of the iron( sulphate) solution. Mn04 + 8H* + 5FE2+ -→ 5FE3+ + 4H20 + Mn2+ (3 Marks) 2. 25cm3 of the solution made when 1.50g o a copper alloy reacted with conc HNO3 is treated with the excess KI solution. The iodine liberated is titrated against 0.10OM sodium thiosulphate. 20.5 cm3 of sodium thiosulpate solution was needed. Calculate the percentage of copper in the alloy. 2S203 2 + 12 -→ S406 2- 21 + 2Cu2+ + 41 -→ 2Cul + 12 1. 25 cm of iron (II) sulphate reacts with 24.3 cm3 of 0.02M potassium manganate (VII) solution. Calculate the concentration of the iron( sulphate) solution. Mn04 + 8H* + 5FE2+ -→ 5FE3+ + 4H20 + Mn2+ (3 Marks) 2. 25cm3 of the solution made when 1.50g o a copper alloy reacted with conc HNO3 is treated with the excess KI solution. The iodine liberated is titrated against 0.10OM sodium thiosulphate. 20.5 cm3 of sodium thiosulpate solution was needed. Calculate the percentage of copper in the alloy. 2S203 2 + 12 -→ S406 2- 21 + 2Cu2+ + 41 -→ 2Cul + 12
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
1. 25 cm of iron (II) sulphate reacts with 24.3 cm Of 0.02M potassium manganate (VII) solution. Calculate the concentration of the iron( sulphate) solution. Mn04 + 8H* + 5FE2+ -> 5FE3+ + 4H20 + Mn2+...
-
1. 25 cm of iron (II) sulphate reacts with 24.3 cm Of 0.02M potassium manganate (VII) solution. Calculate the concentration of the iron( sulphate) solution. Mn04 + 8H* + 5FE2* - 5Fe* + 4H20 + Mn2* (3...
-
HNO3 reacts with iron metal according to Fe(s) + 2HNO3(aq) ( Fe(NO3)2(aq) + H2(g) In a reaction vessel, 23.8 g of Fe are reacted but only 446 mL of H2 are collected over water at 25C and a pressure...
-
Dozier Industries Inc. manufactures only one product. For the year ended December 31, 2014, the contribution margin increased by $38,500 from the planned level of $1,386,000. The president of Dozier...
-
Fill in the blanks. a. Based on the least-squares criterion, the line that best fits a set of data points is the one having the possible sum of squared errors. b. The line that best fits a set of...
-
Selected operating information on three different companies for a recent year is given below: Required: For each company, state whether the company would have a favorable or unfavorable volume...
-
Dirk Coles checkbook lists the following: Dirk Coles September bank statement shows the following: Requirements 1. Prepare Dirk Coles bank reconciliation on September 30, 2010. How much cash does...
-
In Problem 4.16, find: a. EVPI for the cost of making the processor. b. EVPI for the cost of subcontracting the processor. c. EVPI for both uncertain events.
-
Youd deposit $ 3 5 0 a month into an account earning an APR of 2 . 3 % compounded monthly. How much will you have at the end of 5 years?
-
Red's Furniture Manufacturing produces a line of tables and chairs from specialty hardwoods. It makes three different styles of chairs, and each chair takes about the same amount of direct labour...
-
Overhead Activities and Drivers for 2022 Overhead Activities Driver Budgeted Drivers Setups Setups 62,000 Quality Control Batches checked 77,500 Maintenance Machine hours 2,850,000 Transportation /...
-
At 7:40 a.m. on June 27, Officer Steve Sjerven was in the crossover preparing to turn south on Highway 65 to help the driver of an apparently disabled car. As he waited to turn, he saw a red pickup...
-
Kam Ltd has the following equity balances at the end of June 2022. Share Capital A 100,000 shares issued at $5 paid to $2.6 Share Capital B 100,000 shares fully paid @ $4 Total $260,000 400,000...
-
Sarah Jackson Antiques issued its 11%, 10-year bonds payable at a price of $327,550 (face value is $400,000). The company uses the straight-line amortization method for the bond discount or premium....
-
A construction crew needs a crane to lift something that weights 2,024.91 newtons up to the top floor of a building that is 22.23 meters above the ground. The supervisor needs a crane that can do...
-
What are the five determinants of a credit score? How are credit ratings different than credit scoring?
-
Disruptive Innovation explains how firms may fail even if management are acting rationally. Critically discuss whether this is the case using the Netflix and Blockbuster case studies.
-
The production budget of Artest Company calls for 80,000 units to be produced. If it takes 30 minutes to make one unit and the direct labor rate is $16 per hour, what is the total budgeted direct...
-
A sample of natural gas is 80.0% CH4 and 20.0% C2H6 by mass. What is the heat from the combustion of 1.00 g of this mixture? Assume the products are CO2(g) and H2O(l).
-
A cylinder with circular cross section has a radius of 2.56 cm and a height of 56.32 cm. What is the volume of the cylinder? Express the answer to the correct number of significant figures.
-
a. If the molar solubility of cobalt(II) hydroxide is 5.4 106 mol/L in pure water, what is its Ksp value?
-
What are the responsibilities of the plaintiff and the defendant in a 1934 Act suit?
-
What are the conditions associated with liability under Rule \(10 \mathrm{~b}-5\) of the 1934 Act?
-
Identify actions which the auditor may take to avoid litigation.
Lessons Learned At The Shore A Journey Through Retirement 1st Edition - ISBN: 1092595295 - Free Book

Study smarter with the SolutionInn App


