1. a. Calculate the rate constant at the higher temperature used in Experiment 4. b. Calculate the...
Fantastic news! We've Found the answer you've been seeking!
Question:
1. a. Calculate the rate constant at the higher temperature used in Experiment 4.
b. Calculate the activtion energy for the reaction.
c. What do your results demotrate about the effect of a catalyst on the rate of reaction.

Transcribed Image Text:
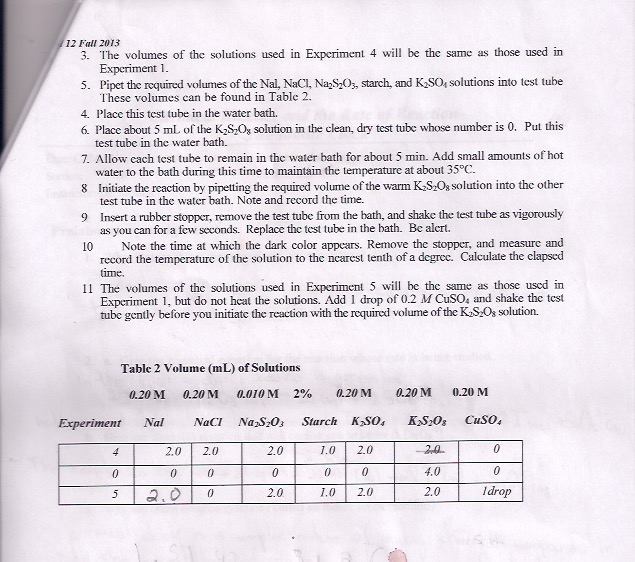
12 Fall 2013 3. The volumes of the solutions used in Experiment 4 will be the same as those used in Experiment 1. 5. Pipet the required volumes of the Nal, NaCl, Na₂S₂O3, starch, and K₂SO, solutions into test tube These volumes can be found in Table 2. 4. Place this test tube in the water bath. 6. Place about 5 mL of the K₂S₂Ox solution in the clean, dry test tube whose number is 0. Put this test tube in the water bath. 7. Allow each test tube to remain in the water bath for about 5 min. Add small amounts of hot water to the bath during this time to maintain the temperature at about 35°C. 8 Initiate the reaction by pipetting the required volume of the warm K₂S₂O, solution into the other test tube in the water bath. Note and record the time. 9 Insert a rubber stopper, remove the test tube from the bath, and shake the test tube as vigorously as you can for a few seconds. Replace the test tube in the bath. Be alert. 10 Note the time at which the dark color appears. Remove the stopper, and measure and record the temperature of the solution to the nearest tenth of a degree. Calculate the clapsed time. 11 The volumes of the solutions used in Experiment 5 will be the same as those used in Experiment 1, but do not heat the solutions. Add 1 drop of 0.2 M CuSO4 and shake the test tube gently before you initiate the reaction with the required volume of the K₂S₂Os solution. Table 2 Volume (mL) of Solutions 0.20 M 0.20 M Experiment Nal 4 0 5 2.0 0 NaCl 2.0 0 0 0.010 M 2% 0.20 M Na₂S₂O3 2.0 0 2.0 Starch K₂SO4 1.0 2.0 0 0 1.0 2.0 0.20 M K₂S208 -2,0 4.0 2.0 0.20 M CuSO4 0 0 Idrop 12 Fall 2013 3. The volumes of the solutions used in Experiment 4 will be the same as those used in Experiment 1. 5. Pipet the required volumes of the Nal, NaCl, Na₂S₂O3, starch, and K₂SO, solutions into test tube These volumes can be found in Table 2. 4. Place this test tube in the water bath. 6. Place about 5 mL of the K₂S₂Ox solution in the clean, dry test tube whose number is 0. Put this test tube in the water bath. 7. Allow each test tube to remain in the water bath for about 5 min. Add small amounts of hot water to the bath during this time to maintain the temperature at about 35°C. 8 Initiate the reaction by pipetting the required volume of the warm K₂S₂O, solution into the other test tube in the water bath. Note and record the time. 9 Insert a rubber stopper, remove the test tube from the bath, and shake the test tube as vigorously as you can for a few seconds. Replace the test tube in the bath. Be alert. 10 Note the time at which the dark color appears. Remove the stopper, and measure and record the temperature of the solution to the nearest tenth of a degree. Calculate the clapsed time. 11 The volumes of the solutions used in Experiment 5 will be the same as those used in Experiment 1, but do not heat the solutions. Add 1 drop of 0.2 M CuSO4 and shake the test tube gently before you initiate the reaction with the required volume of the K₂S₂Os solution. Table 2 Volume (mL) of Solutions 0.20 M 0.20 M Experiment Nal 4 0 5 2.0 0 NaCl 2.0 0 0 0.010 M 2% 0.20 M Na₂S₂O3 2.0 0 2.0 Starch K₂SO4 1.0 2.0 0 0 1.0 2.0 0.20 M K₂S208 -2,0 4.0 2.0 0.20 M CuSO4 0 0 Idrop
Expert Answer:
Answer rating: 100% (QA)
NaI moles in 2 mL 0221000 00004 moles persulphate moles in 2mL 0221000 00004 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the rate constant at 500 K for the second-order gas-phase reaction between Cl and H 2 , given the frequency factor, A-8.1x10 -10 dm 3 mol -1 s -1 and activation energy E a = 23 kJ mol.
-
In an experiment to determine the effect of catalyst on the yield of a certain reaction, the mean yields for reactions run with each of four catalysts were X1. = 89.88, X2. = 89.51, X3. = 86.98, and...
-
Calculate the energy change for the following nuclear reaction (in joules per mole of Give the energy change in MeV per nucleus. See Table 20.3. H) H +H Heon 97 97 98 98 01 02 03 03 04 03 04 04 OS...
-
Southern Stitches is a local casual clothing shop that makes a variety of t-shirt styles. Below is some actual vs. budget information for Southern's t-shirts for the month of May. Assume no...
-
Define what is meant by an audit trail and explain how it can be affected by the client's integration of IT.
-
There are no universally accepted definitions of financial ratios, but some of the following ratios make no sense at all. Substitute correct definitions and then explain what each ratio measures....
-
Janes Cream Soda, Inc., makes specialty soft drinks. At the end of 2010, Janes had total assets of \($300,000\) and liabilities totaling $220,000. Requirements 1. Write the companys accounting...
-
Lewiss management has been considering moving to a new downtown location, and they are concerned that these plans may come to fruition prior to the expiration of the lease. If the move occurs, Lewis...
-
Amman Company has three production lines, Production Line 1, Production Line 2 and Production Line 3. The management of the company has decided to close Production Line 3 from company's operations...
-
Two flow control loops axe shown in the drawing. Indicate whether each system is either a feedback or a feed-forward control system. Justify your answer. It can be assumed that the distance between...
-
Which of the following statements is true about the Boolean data type? A) The Boolean data type can only be assigned a value of either true or false. B) The Boolean data type can be assigned any...
-
Would it be permissible to allocate all vocational training funds for students with disabilities to separate programs? Should all vocational training programs integrate students with disabilities and...
-
Conventional product costing systems are likely to overcost high-volume products. Do you agree with this statement? Explain.
-
Is there harm done to the public by awarding a student a diploma when the student has not completed competency requirements? Is this different from a situation involving higher education? That is, is...
-
What is a job-cost sheet? Prepare a comprehensive job-cost sheet of a firm having two departments.
-
Specify the production situations that are more suitable to the process costing system.
-
Question 4 Tic-tac-toe (also known as noughts and crosses) is a game for two players, X and O, who take turns marking the spaces in a 3x3 grid. The player who succeeds in placing three of their marks...
-
What services are provided by the provincial and territorial governments?
-
In the presence of excess thiocyanate ion, SCN, the following reaction is first order in chromium(III) ion, Cr3+; the rate constant is 2.0 106/s. Cr3+(aq) + SCN(aq) Cr(SCN)2+(aq) If 85.0% reaction...
-
Ammonium sulfate is used as a nitrogen and sulfur fertilizer. It is produced by reacting ammonia with sulfuric acid. Write the balanced equation for the reaction of gaseous ammonia with sulfuric acid...
-
Explain why evaporation leads to cooling of the liquid.
-
Figure P18-39 shows two similar unit pulse waveforms. The only difference is that the first pulse is positive from \(-T / 2\) to \(T / 2\), while the second pulse goes from \(+\mathrm{A}\) to...
-
Describe the principal types of audits performed by independent auditors and indicate the established criteria and primary users of each tyr
-
Contrast the activities of independent, internal, and governmental auditors.

Study smarter with the SolutionInn App


