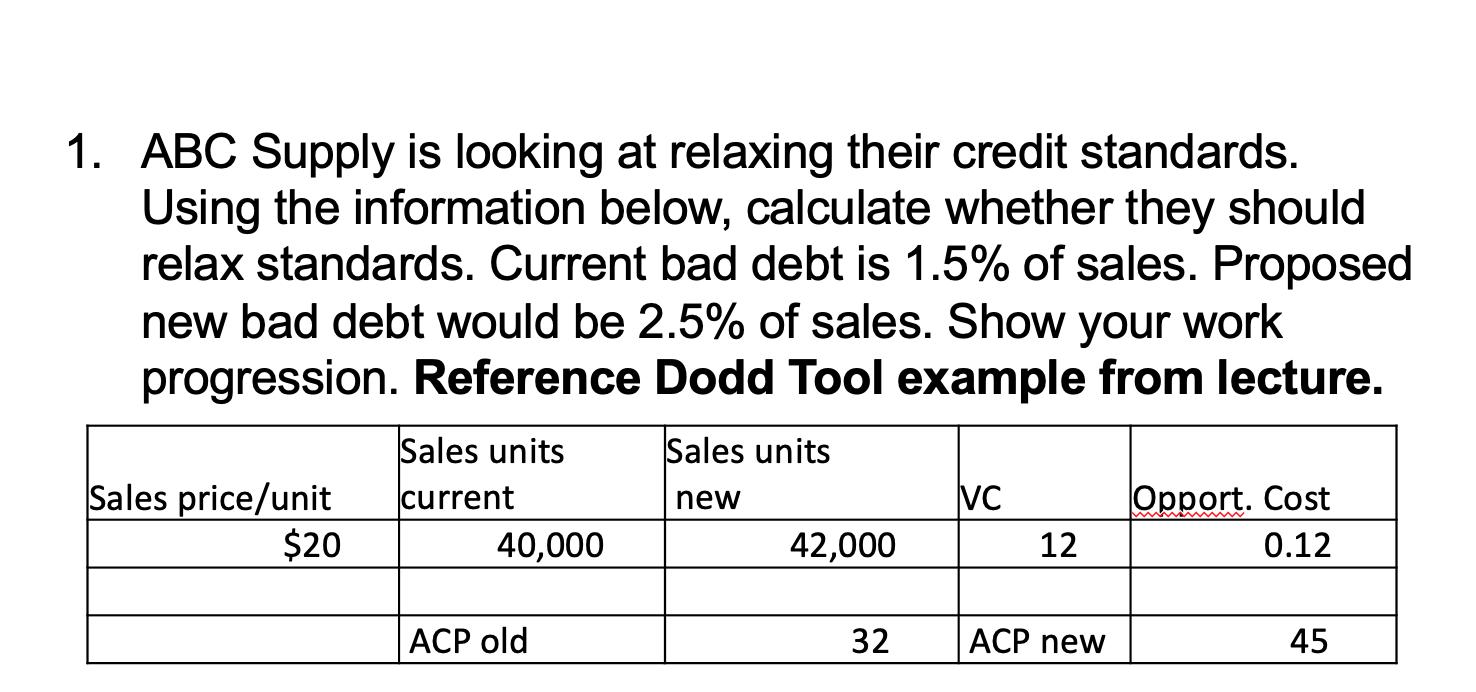
1. ABC Supply is looking at relaxing their credit standards. Using the information below, calculate whether...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1. ABC Supply is looking at relaxing their credit standards. Using the information below, calculate whether they should relax standards. Current bad debt is 1.5% of sales. Proposed new bad debt would be 2.5% of sales. Show your work progression. Reference Dodd Tool example from lecture. Sales price/unit $20 Sales units current 40,000 ACP old Sales units new 42,000 32 VC 12 ACP new Opport. Cost 0.12 45 1. ABC Supply is looking at relaxing their credit standards. Using the information below, calculate whether they should relax standards. Current bad debt is 1.5% of sales. Proposed new bad debt would be 2.5% of sales. Show your work progression. Reference Dodd Tool example from lecture. Sales price/unit $20 Sales units current 40,000 ACP old Sales units new 42,000 32 VC 12 ACP new Opport. Cost 0.12 45
Expert Answer:
Answer rating: 100% (QA)
To determine whether ABC Supply should relax their credit standards we need to compare the cost of b... View the full answer

Related Book For 

Fundamental Accounting Principles Volume II
ISBN: 978-1259066511
14th Canadian Edition
Authors: Larson Kermit, Jensen Tilly
Posted Date:
Students also viewed these finance questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Refer to the information in BE4-7 for Hébert Company. Prepare the correcting journal entries. Information in BE4-7 1. A collection of cash on account from a customer for $750 was recorded as a...
-
The pre-sinking stress field assumed to be caused by gravity alone (Problem 3.37) turns out to be wrong. The actual stress field has a tectonic component that adds a constant 8.62 MPa to the eastwest...
-
In Table 3.6, what exact score falls at the 25th percentile rank? Use the appropriate formula. Table 3.6 Cumulative Relative Frequency for Statistics Test Scores Score 98 96 94 93 92 91 90 89 88 87...
-
How does an auditor evaluate the effectiveness and reliability of a client's control procedures over revenue /receipt cycle activities?
-
Fast Turnstiles Co. is evaluating the extension of credit to a new group of customers. Although these customers will provide $180,000 in additional credit sales, 12 percent are likely to be...
-
Project Alpha would generate annual cash inflows of 200,000 after the purchase of machinery costing 556,000, with a scrap value after five years of 56,000. Project Beta would generate annual cash...
-
Pure Plant Beauty is a sole proprietorship that has a developed a new line of skin care and makeup that uses natural and organic ingredients. The company showed the following adjusted account...
-
Make this Lo Shu Magic into a 4 x 4 matrix instead of a 3 x 3 matrix? def is_lo_shu_magic_square(square)make this Lo Shu Magic into a 4 x 4 matrix instead of a 3 x 3 matrix? Every change I made...
-
Moody Corporation's bonds have a 15-year maturity, a 7.25% coupon paid semiannually, and a par value of $1,000. If the going market interest rate for bonds of similar risk and maturity is 6.00%...
-
A reaction has a rate constant of 8.2 x 10-2 M-1s-1. If you start with an initial concentration of 6.49M, what is the half-life?
-
An investor wants to purchase a 2 year treasury bond, and the broker provides the investor with the following two alternatives. a) A $10,000 10 year Treasury bond issued on March 8, 2013 that matures...
-
According to the balanced reaction below, calculate the quantity of moles of NH3 gas that form when 4.20 mol of NH4 liquid completely reacts: 3 NH4(1) 4 NH3(g) + N(g)?
-
On her birthday, February 20, 2009, Marley received a gift from her father of unimproved land located in Marshal County, IN. The land cost her father $23,000 in 1970 and had a value of $76,000 on the...
-
Given the triangle below, we will find the area of the triangle using Heron's formula. 26cm/ 16cm 40cm Q The semi-perimeter, s, is 41 cm To substitute into Heron's formula, use the 3 sides in...
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
The 2014 financial statements of Dalmac Energy Inc. follow: Assume all sales were on credit. On the December 31, 2013, balance sheet, the assets totalled $432,720, common shares were $192,000, and...
-
A company reported $576,000 of net income for 2014. It also declared $78,000 of dividends on preferred shares for the same year. At the beginning of 2014, the company had 50,000 outstanding common...
-
Liam and Katano formed a partnership by investing $95,000 and $105,000 respectively. They agreed to share income based on an allocation to Liam of an annual salary allowance of $150,000, interest...
-
What are the PMI and PRINCE2?
-
What are the differences between a framework, a methodology and a standard?
-
Individually identify what you consider to be the greatest achievements accomplished by human kind in the last decade. Now share your list with other students in the class and come up with an...

Study smarter with the SolutionInn App


