1. Add curved arrows to both structures to show the delocalization of electron pairs needed to form...
Fantastic news! We've Found the answer you've been seeking!
Question:
1. Add curved arrows to both structures to show the delocalization of electron pairs needed to form the other resonance contributor.
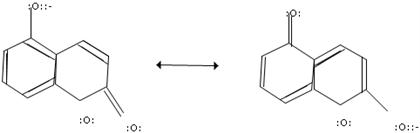
2a.) Draw the predominant resonance contributor for the following compound; include lone pairs of electrons, formal charges, and hydrogen atoms.
b) Add curved arrows to both structures to show the delocalization of electron pairs.
Related Book For 

Organic Chemistry A Short Course
ISBN: 978-1111425562
13th edition
Authors: Harold Hart, Christopher M. Hadad, Leslie E. Craine, David J. Hart
Posted Date:




