1- identigy FTIR spectrum of your product and 2- % Transmittance complete the H-NMR and find...
Fantastic news! We've Found the answer you've been seeking!
Question:
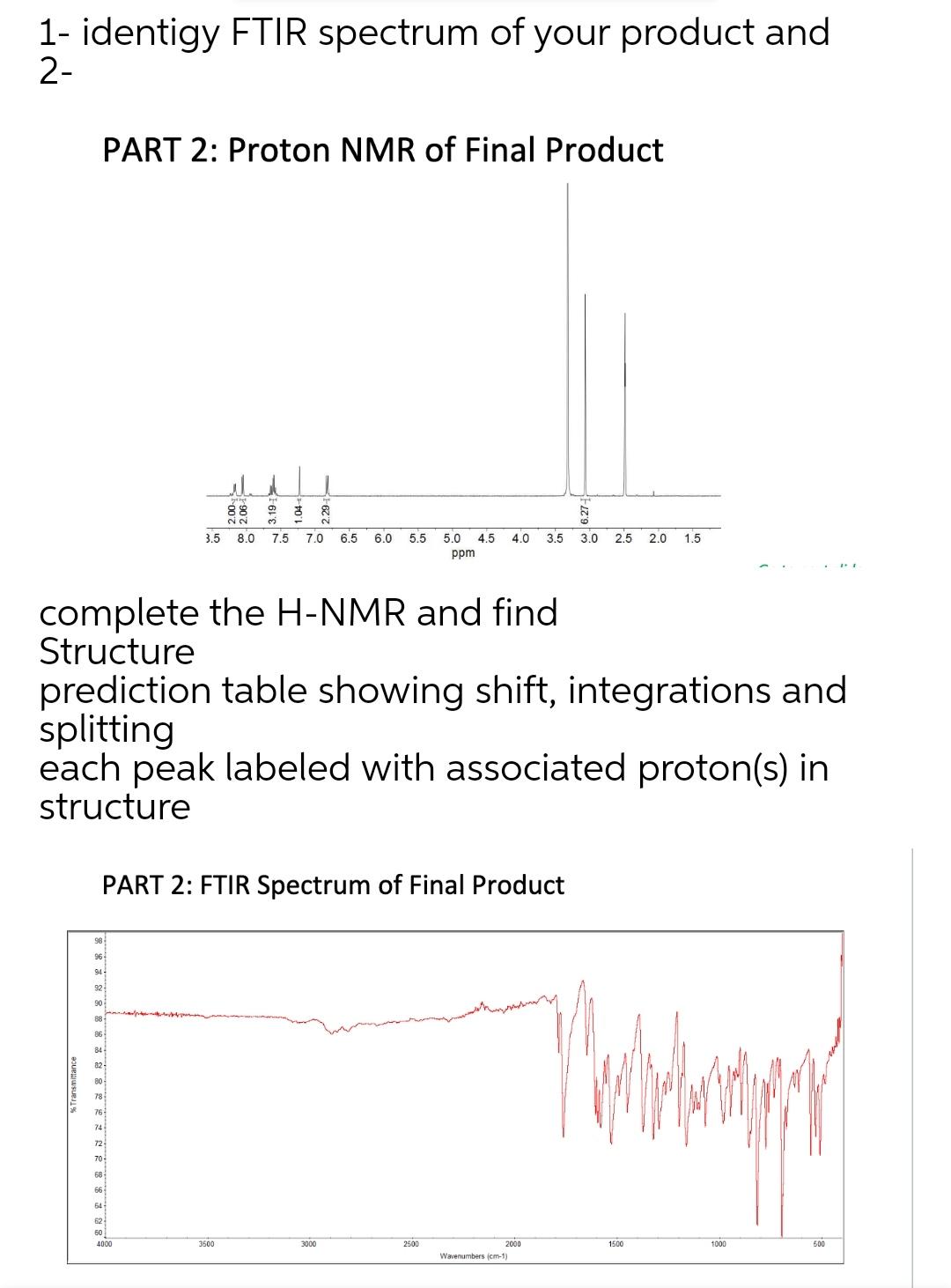
Transcribed Image Text:
1- identigy FTIR spectrum of your product and 2- % Transmittance complete the H-NMR and find Structure prediction table showing shift, integrations and splitting each peak labeled with associated proton(s) in structure 98 PART 2: Proton NMR of Final Product 86 90 PART 2: FTIR Spectrum of Final Product 82 80 78 76 68 66 64 H 10 ष्ठ 3 2 3.5 8.0 7.5 7.0 6.5 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 ppm 3500 3000 2500 2000 Wavenumbers (cm-1) Talahanay 1500 1000 500 1- identigy FTIR spectrum of your product and 2- % Transmittance complete the H-NMR and find Structure prediction table showing shift, integrations and splitting each peak labeled with associated proton(s) in structure 98 PART 2: Proton NMR of Final Product 86 90 PART 2: FTIR Spectrum of Final Product 82 80 78 76 68 66 64 H 10 ष्ठ 3 2 3.5 8.0 7.5 7.0 6.5 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 ppm 3500 3000 2500 2000 Wavenumbers (cm-1) Talahanay 1500 1000 500
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In a proton NMR spectrum the peak from TMS was found to show 'wiggles' characteristic of truncation of the FID. However, the other peaks in the spectrum showed no such artefacts. Explain.
-
In a 1H NMR spectrum the peak from TMS is found to occur at 500.134 271 MHz. Two other peaks in the spectrum are found at 500.135 021 and 500.137 921 MHz; compute the chemical shifts of these two...
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
After 1 0 0 0 B . C . E . trade in Greece allowed for the accumulation of wealth by Kings Aristoi Farmers Foreigners
-
Explain why some electrolyte solutions are strongly conducting, whereas others are weakly conducting.
-
Explain why and how the business combination valuation entries for liabilities will be adjusted in subsequent periods after the acquisition date.
-
What does a technician measure to set a proximity probe into a machine for the most useful range?
-
Alternative methods of joint-cost allocation, product-mix decisions. The Sunshine Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoft point; A, B, C, and...
-
A portfolio manager is considering the purchase of a bond with a 5.5% coupon rate that pays interest annually and matures in 3 years. If the required rate of return on the bond is 5%, the price of...
-
Forecasting with the Parsimonious Method and Estimating Share Value Using the ROPI Model Following are income statements and balance sheets for Cisco Systems. Cisco Systems Consolidated Statements of...
-
At a minimum, you need one quantitative (numerical) variable and one qualitative (categorical) variable (with at least two levels). Get some data from the source of your choice (at least two columns...
-
The CNO has asked to meet with you to discuss your stakeholder analysis. Prepare a stakeholder map, power/interest grid, and stakeholder analysis in advance of the meeting. Also include a one-page...
-
Assume the situation from question 13 , except now assume that banks hold a ratio of \(0.5 \%\) of excess reserves to deposits and the public keeps \(20 \%\) of its liquid assets in the form of cash....
-
You are the chief nurse executive at an organization that provides clinic services to Medicare patients; seven of the clinics are located in underserved areas. While visiting one of the clinics, you...
-
An investment company has \(\$ 1.05\) million of assets, \(\$ 50,000\) of liabilities, and 10,000 shares outstanding. a. What is its NAV? b. Suppose the fund pays off its liabilities while at the...
-
You are an NP who sees mostly posthead trauma patients and who refers many of these patients to physical and occupational services. On one routine follow-up visit, your patient claims that he has not...
-
What do analysts want to find out in assessing accrual quality? Explain briefly.
-
What is EBIT/eps analysis? What information does it provide managers?
-
Use bond-dissociation enthalpies (Table 4-2) to calculate values of for the following reactions. (a) CH3 - CH3 + I2 CH3 CH2I + HI (b) CH3 CH2 Cl + HI CH3 CH2 I + HCl (c) (CH3)3C -OH + HCl (CH3)3C...
-
(a) Two stereoisomers of a bromodecalin are shown. Although the difference between these stereoisomers may seem trivial, one isomer undergoes elimination with KOH much faster than the other. Predict...
-
Show how you would accomplish the following synthetic conversions. (a) but-1-ene 1-bromobutane (b) but-1-ene 2-bromobutane (c) 2-methylcyclohexanol 1-bromo-1-methylcyclohexane (d) 2-methylbutan-2-ol...
-
Assume that you have shorted the put option in Problem 4. a. If the stock is trading at \($8\) in three months, what will you owe? b. If the stock is trading at \($23\) in three months, what will you...
-
Assume that you have shorted the call option in Problem 2. PROBLEM 2 You own a call option on Intuit stock with a strike price of \($40.\) The option will expire in exactly three months. a. If the...
-
Redo Problem 14, but assume that Kay must pay a corporate tax rate of 21%, and that investors pay no taxes. Problem 14. Chen Brothers, Inc., sold 4 million shares in its IPO, at a price of \($18.50\)...

Study smarter with the SolutionInn App



