1. If you dissolve 10.00 g of K3N (s) in 9.00 L of water (density =...
Fantastic news! We've Found the answer you've been seeking!
Question:
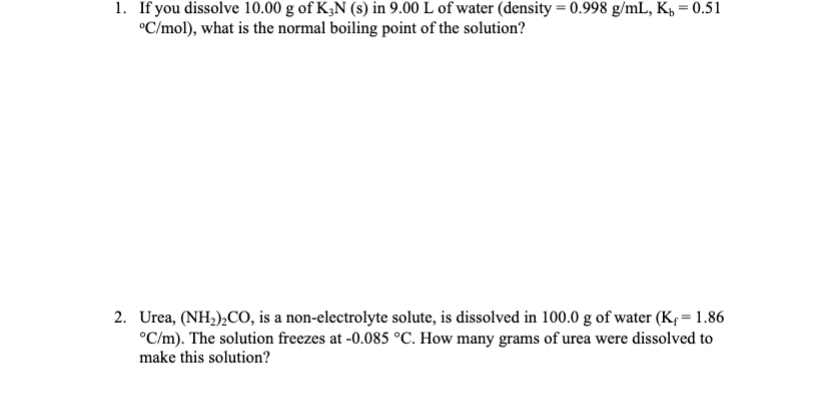
Transcribed Image Text:
1. If you dissolve 10.00 g of K3N (s) in 9.00 L of water (density = 0.998 g/mL, K = 0.51 C/mol), what is the normal boiling point of the solution? 2. Urea, (NH)CO, is a non-electrolyte solute, is dissolved in 100.0 g of water (K = 1.86 C/m). The solution freezes at -0.085 C. How many grams of urea were dissolved to make this solution? 1. If you dissolve 10.00 g of K3N (s) in 9.00 L of water (density = 0.998 g/mL, K = 0.51 C/mol), what is the normal boiling point of the solution? 2. Urea, (NH)CO, is a non-electrolyte solute, is dissolved in 100.0 g of water (K = 1.86 C/m). The solution freezes at -0.085 C. How many grams of urea were dissolved to make this solution?
Expert Answer:
Answer rating: 100% (QA)
Ill address each question separately 1 To find the normal boiling point of the solution for the compound KN we will use the formula for boiling point ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Create a cash flow sheet with Initial investment 100,000 resources expense : 5,000 labour cost: 10,000 maintenance cost : 3000 increase 2 warehouses (rental): 15,000 website development: 5,000...
-
You propose a new temperature scale with temperatures given in oM. You define 0.0oM to be the normal melting point of mercury and 100.0o to be the normal boiling point of mercury. (a) What is the...
-
Samson Company manufactures embroidered jackets. The company uses a standard cost system to control manufacturing costs. The following data represent the standard unit cost of a jacket: Fixed...
-
Reflect on the logical steps of effective change implementation. If you have not already done so, read Spector's Four Steps. Here they are below, as well. Step 1-Redesign considers alternative...
-
Find the lengths of the curves. x = (y/4) 2 - 2 ln (y/4), 4 y 12
-
While the electromagnetic interaction between the photons and the charged electrons and positrons kept them in equilibrium with each other during the early universe, show that the direct...
-
A natural gas trading company wants to develop an optimal trading plan for the next 10 days. The following table summarizes the estimated prices (per thousand cubic feet (cf)) at which the company...
-
Thompson's Hardware spent $46,370 this year on business insurance alone. If total sales were $765,500, what percent of total sales was spent on business insurance? Round to the nearest tenth.
-
Implement the following LP problem in a spreadsheet. Use Solver to solve the problem and create a Sensitivity Report. Use this information to answer the following questions: MAX: 4X1 + 2X2 Subject...
-
Imagine that you are driving your vehicle over a bridge. The width of your vehicle is equivalent to the spread or range of the data. The guardrails on each side of the bridge are your specification...
-
post covid KPis and the importance of SQDCM? Which metric is more beneficial to the overall goal of a business ? having the right metrics isn't enough data visualization is key , but why ? need...
-
Using Anwar Shaikh's explanation of 'real competition' (see Capitalism, Competition, Conflict, Crisis, chapter 8) assess the importance of the interaction between government and car manufacturers for...
-
How does celebrity endorsement work as a form of marketing communication? What are some disadvantages of using celebrity endorsement? What should this non-profit organization watch for when selecting...
-
4. Compute each of the double sums. a. ( + 3j) =0 b. =1 21
-
How much consideration does government give to its' employees' overall job satisfaction? Should (or can) government focus on employees' career development? If it did so, could we expect to see...
-
1. Review various visuals describing the Action Research models and select one that best exhibits your own understanding of the AR process. Then, explain the defining elements found within the visual...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Give the number of protons and electrons in each of the following common ions: K+, Mg2+, Fe3+, Br-, Mn2+, C4-, Cu2+.
-
`The standard enthalpies of formation of ions in aqueous solutions are obtained by arbitrarily assigning a value of zero to H+ ions; that is, Hf [H+(aq)] = 0. (a) For the following reaction ...
-
Methyl bromide (CH3Br, b.pt. 3.6C) is used as a soil fumigant to control insects and weeds. It is also a marine by-product. Photo dissociation of the C-Br bond produces Br atoms that can react with...
-
Based on this chapter and the case incident, compile a list of 10 international HR mistakes Mr. Fisher has made so far.
-
Based on what you read in this chapter of Dessler, Human Resource Management, whats the first step you would have advised the Hotel Paris to take as part of its new safety and health program, and why?
-
Write a one-page summary addressing the topic, How improving safety and health at the Hotel Paris will contribute to us achieving our strategic goals.

Study smarter with the SolutionInn App


