1. Let us assume that air comprises 78% nitrogen and 22% of oxygen, and assume that...
Fantastic news! We've Found the answer you've been seeking!
Question:
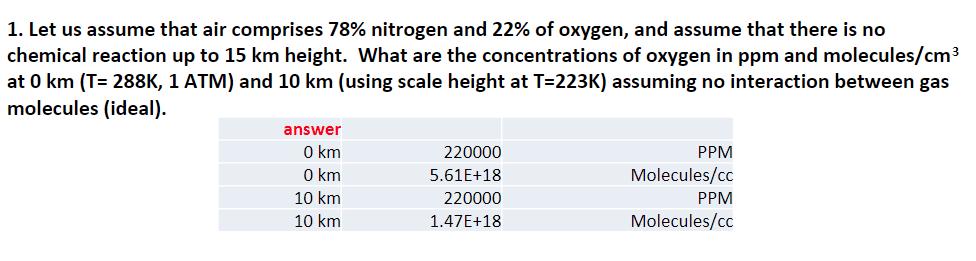

Transcribed Image Text:
1. Let us assume that air comprises 78% nitrogen and 22% of oxygen, and assume that there is no chemical reaction up to 15 km height. What are the concentrations of oxygen in ppm and molecules/cm³ at 0 km (T= 288K, 1 ATM) and 10 km (using scale height at T=223K) assuming no interaction between gas molecules (ideal). answer 0 km 0 km 10 km 10 km 220000 5.61E+18 220000 1.47E+18 PPM Molecules/cc PPM Molecules/cc ● Summary of Equations Atmospheric pressure (p) at height of z: p(z) = Poe-z/H where scale height: H(z) = RT(z)/gMair H(z): in unit of km R = 0.082 L atm K-¹ mol-¹ T(z): temperature (K) at height of z g= 9.8 m s-¹ Mair = 28.97 g mol-¹ b Black body temperature T(K) == max where b is Wien's constant: b = 2897768.5 nm K and max (nm) is the peak wavelength that the black body emits. Earth's temperature (Te, K): Te = [(1 − A)]¹/4 So: solar constant (1370 W m-²) A: planetary albedo (0.3) o: Stephan-Boltzmann constant (5.67 x 10-8 Wm-2K-4) 1. Let us assume that air comprises 78% nitrogen and 22% of oxygen, and assume that there is no chemical reaction up to 15 km height. What are the concentrations of oxygen in ppm and molecules/cm³ at 0 km (T= 288K, 1 ATM) and 10 km (using scale height at T=223K) assuming no interaction between gas molecules (ideal). answer 0 km 0 km 10 km 10 km 220000 5.61E+18 220000 1.47E+18 PPM Molecules/cc PPM Molecules/cc ● Summary of Equations Atmospheric pressure (p) at height of z: p(z) = Poe-z/H where scale height: H(z) = RT(z)/gMair H(z): in unit of km R = 0.082 L atm K-¹ mol-¹ T(z): temperature (K) at height of z g= 9.8 m s-¹ Mair = 28.97 g mol-¹ b Black body temperature T(K) == max where b is Wien's constant: b = 2897768.5 nm K and max (nm) is the peak wavelength that the black body emits. Earth's temperature (Te, K): Te = [(1 − A)]¹/4 So: solar constant (1370 W m-²) A: planetary albedo (0.3) o: Stephan-Boltzmann constant (5.67 x 10-8 Wm-2K-4)
Expert Answer:
Answer rating: 100% (QA)
number density of n at P pressure N avagadro conumber T t... View the full answer

Related Book For 

Management Foundations a pacific rim focus
ISBN: 9780070284944
3rd edition
Authors: Kathryn Bartol, Margaret Tein, Graham Matthews, Bishnu Sharma, Brenda Scott-Ladd
Posted Date:
Students also viewed these mechanical engineering questions
-
Let us assume that air is under standard conditions close to the Earth's surface. Presuming that the temperature and the molar mass of air are independent of height, find the air pressure at the...
-
Let us assume that you need a computer for your studies at university or college. In the process of buying a computer, how would you apply the decision-making algorithm/steps?
-
Let us assume that the prior distribution for the proportion y of drinks from a vending machine that overflow is. If 2 of the next 9 drinks from this machine overflow, find(a) The posterior...
-
How can a layer fold and boudinage (extend) at the same time?
-
Cerulean Corporation has two equal shareholders, Eloise and Olivia. Eloise acquired her Cerulean stock three years ago by transferring property worth $700,000, basis of $300,000, for 70 shares of the...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
In Example 17.11, assume that the promised amounts to \(A\) and \(B\) are \(\$ 110\) and \(\$ 90\), respectively. Develop the new table of results. What are the prices of \(A\) and \(\mathrm{B}\) ?...
-
You were recently approached by one of your clients, Wendy Wonders, the Chief Financial Officer of Rock Group Limited ( RGL), a Canadian public company with a 31 December year- end. RGL manufactures...
-
Suppose that there are three firms in a region that are producing a common emission. The marginal abatement cost (MAC) for each firm is given by: MAC1 240-2E1 MAC2 192-1.6E2 MAC3 = 320 -2.67E3 The...
-
The diagram below represents a process where two components are made at stations A1 and A2 (one component is made at A1 and the other at A2). These components are then assembled at station B and...
-
1. Under which set of auditing standards is the auditor required to express an opinion on compliance with laws and regulations applicable to each major program? a. AICPA standards. b. GAO standards....
-
Discuss how a firearm and ammunition use both potential and kinetic energy.
-
How might the self-fulfilling prophecy work in a positive sense? A negative sense? which is more common, and why? How can negative thoughts like this be realistically managed?
-
Direction: Make an essay about festival dance, chose one from the given questions below. Write your answer in a separate sheet of paper. Processing questions: 1. Why is there a need for festivals?...
-
Describe one type of role-play technique you could use in a specific group setting. Explain who would benefit from this technique and why. Toseland, R. W., & Rivas, R. F. (2017). An introduction to...
-
Suppose that a manager is following a base stock policy where the optimal inventory position is 10. Assume the component lead time is 2 days. At the end of day 1, there is no ordered units yet to be...
-
Consider the recorded transactions below. Transaction 1. 2. 3. 4. 5. 6. Accounts Receivable. Service Revenue Supplies Account Name Accounts Payable Cash Accounts Receivable Advertising Expense Cash...
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
Describe force-field analysis. Suggest three situations where it might be useful to help analyse a change situation.
-
Describe three levels of controls in organisations. For a familiar organisation, identify a control at each level.
-
Delineate the four principal conditions that managers need to consider when deciding what to control.
-
True or False: The most commonly used measure of inflation is the relative change in the Consumer Price Index.
-
Mike's Veneer Shop owns a vacuum press that requires annual maintenance. Mike has a contract to cover the maintenance expenses for the next 5 years. The contract calls for an annual payment of \(\$...
-
What is the relationship between inflation and deflation? Give an example of deflation experienced in your everyday life.

Study smarter with the SolutionInn App


