1. Lil reacts with O to give I and LiO in a closed vessel at constant...
Fantastic news! We've Found the answer you've been seeking!
Question:

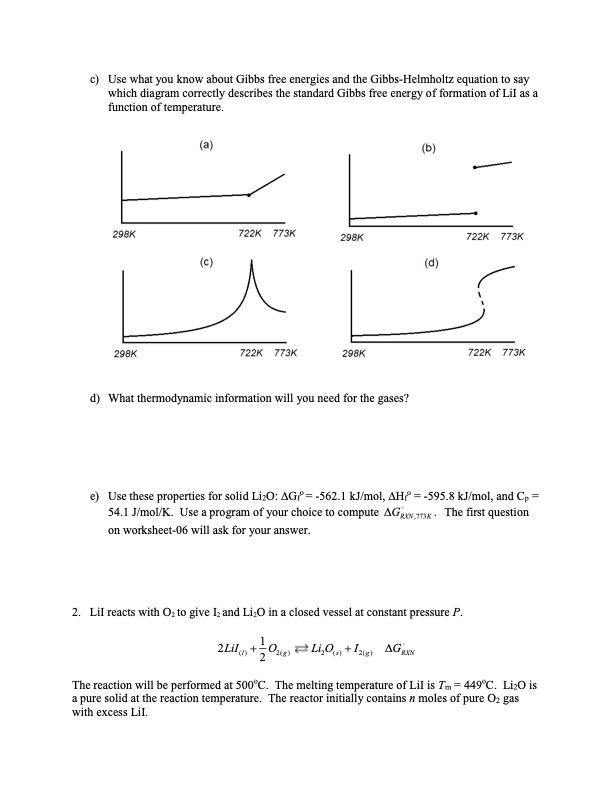

Transcribed Image Text:
1. Lil reacts with O to give I and LiO in a closed vessel at constant pressure P. 2Lil+0) Li0+12) AGRON The reaction will be performed at 500C. The melting temperature of Lil is Tm=449C. LiO is a pure solid at the reaction temperature. The reactor initially contains n moles of pure O2 gas with excess Lil. a) You will need some unusual thermodynamic information to compute AGRXN". Which curve accurately describes the standard enthalpy of formation vs. temperature for Lil? (a) (b) 298K 298K (c) 722K 773K 298K 722K 773K 298K (d) 722K 773K 722K 773K b) Lil is a solid at the reference temperature (25C) with properties AG=-266.9 kJ/mol and AH=-270.48 kJ/mol. The heat capacity of the solid is Cp,solid 54.4 J/mol/K. Lil melts at 449C and the heat of fusion is AHm=14.63 kJ/mol. The liquid heat capacity is Cp.liquid=63.2 J/mol/K. You must integrate the Gibbs-Helmholtz equation to compute AGRXN, but first match each property to the feature it controls in the enthalpy vs. temperature diagram. Please select the correct form of plot from (a) and correspond a list of features and options (AH298K Cold Clawd and AH) to properties of the curve (i.e. intercept, slope, gap, ...). c) Use what you know about Gibbs free energies and the Gibbs-Helmholtz equation to say which diagram correctly describes the standard Gibbs free energy of formation of Lil as a function of temperature. (a) (b) 298K 722K 773K 298K (c) 298K 722K 773K 298K d) What thermodynamic information will you need for the gases? 722K 773K (d) 722K 773K e) Use these properties for solid LiO: AG=-562.1 kJ/mol, AH? =-595.8 kJ/mol, and Cp = 54.1 J/mol/K. Use a program of your choice to compute AGNK. The first question on worksheet-06 will ask for your answer. 2. Lil reacts with O to give I, and LiO in a closed vessel at constant pressure P. 2Lil + LiO +12 AGN The reaction will be performed at 500C. The melting temperature of Lil is Tm=449C. LiO is a pure solid at the reaction temperature. The reactor initially contains n moles of pure O2 gas with excess Lil. a) In the previous worksheet, you computed AGAN,773K Give your answer here in kJ/mol. b) Using K = exp[-AGS/kT] you can compute a dimensionless equilibrium constant. Construct the mass action ratio of activities and equate it to your undetermined constant K773k. Based on your expression, what units would the equilibrium constant have for those engineers who prefer to absorb the reference pressures into the equilibrium constant? c) Construct a stoichiometric table. This time use conversion. d) Find the equilibrium conversion Xo, as a function of Ko, n, P, P. e) Use the correct value of Keq to estimate the mole fraction of Oz in the gas phase at equilibrium. Let the total pressure be latm. 1. Lil reacts with O to give I and LiO in a closed vessel at constant pressure P. 2Lil+0) Li0+12) AGRON The reaction will be performed at 500C. The melting temperature of Lil is Tm=449C. LiO is a pure solid at the reaction temperature. The reactor initially contains n moles of pure O2 gas with excess Lil. a) You will need some unusual thermodynamic information to compute AGRXN". Which curve accurately describes the standard enthalpy of formation vs. temperature for Lil? (a) (b) 298K 298K (c) 722K 773K 298K 722K 773K 298K (d) 722K 773K 722K 773K b) Lil is a solid at the reference temperature (25C) with properties AG=-266.9 kJ/mol and AH=-270.48 kJ/mol. The heat capacity of the solid is Cp,solid 54.4 J/mol/K. Lil melts at 449C and the heat of fusion is AHm=14.63 kJ/mol. The liquid heat capacity is Cp.liquid=63.2 J/mol/K. You must integrate the Gibbs-Helmholtz equation to compute AGRXN, but first match each property to the feature it controls in the enthalpy vs. temperature diagram. Please select the correct form of plot from (a) and correspond a list of features and options (AH298K Cold Clawd and AH) to properties of the curve (i.e. intercept, slope, gap, ...). c) Use what you know about Gibbs free energies and the Gibbs-Helmholtz equation to say which diagram correctly describes the standard Gibbs free energy of formation of Lil as a function of temperature. (a) (b) 298K 722K 773K 298K (c) 298K 722K 773K 298K d) What thermodynamic information will you need for the gases? 722K 773K (d) 722K 773K e) Use these properties for solid LiO: AG=-562.1 kJ/mol, AH? =-595.8 kJ/mol, and Cp = 54.1 J/mol/K. Use a program of your choice to compute AGNK. The first question on worksheet-06 will ask for your answer. 2. Lil reacts with O to give I, and LiO in a closed vessel at constant pressure P. 2Lil + LiO +12 AGN The reaction will be performed at 500C. The melting temperature of Lil is Tm=449C. LiO is a pure solid at the reaction temperature. The reactor initially contains n moles of pure O2 gas with excess Lil. a) In the previous worksheet, you computed AGAN,773K Give your answer here in kJ/mol. b) Using K = exp[-AGS/kT] you can compute a dimensionless equilibrium constant. Construct the mass action ratio of activities and equate it to your undetermined constant K773k. Based on your expression, what units would the equilibrium constant have for those engineers who prefer to absorb the reference pressures into the equilibrium constant? c) Construct a stoichiometric table. This time use conversion. d) Find the equilibrium conversion Xo, as a function of Ko, n, P, P. e) Use the correct value of Keq to estimate the mole fraction of Oz in the gas phase at equilibrium. Let the total pressure be latm.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Roles define the manager. Do you agree or disagree with this statement? Discuss what you think managers do.
-
Two points on a line are measured by a CMM in the x-y plane. The points have the following coordinates: (100.24, 20.57) and (50.44, 60.46), where the units are mm. The given coordinates have been...
-
During the year, TRC Corporation has the following inventory transactions. Date January 1 Transaction Beginning inventory April 7 Purchase July 16 Purchase October 6 Purchase Number of Unit Total...
-
Should a canoe paddle be made rough to get a "better grip on the water" for paddling purposes? Explain.
-
A large piece of equipment acquired on 1 January 20X5 by Kapadia Company has four major components for depreciation. Details regarding each component are given in the schedule below: Required: 1....
-
The following items are reported on a company's balance sheet: Cash $296,600 Marketable securities 185,000 Accounts receivable (net) 121,000 Inventory 127,000 Accounts payable 228,000 Determine (a)...
-
you will write a complete C++ program that will display a menu and based on user input, perform a variety of operations to process the grades using structures. This is an individual work. Program...
-
Let F = (5t +4, sin(41), 5t) Find the indefinite vector integral F F(t) dt = ( +C2, +C1, +C3)
-
How is scope affected if an auditor decides that a smaller amount of misstatement is material for the same detection risk?
-
Calculate and report the value for the Upper Control Limit (UCL) and the Lower Control Limit (LCL) of the Proportion (p) control chart. Please do all calculations and report your final answers...
-
8 0 , 0 0 0 lb / h of a 1 0 % soda solution in water will be concentrated to 5 0 % . Water vapor available at 1 0 0 psia and 3 4 0 F will be used. The third evaporator has a pressure of 2 psia. The...
-
A particle starts from the origin at t=0 with an initial velocity of 5.5 m/s along the positive axis. If the acceleration is (-3.5 +4.2 m/s, determine (a)the velocity and (b)position of the particle...
-
Using the internet reference Shockley-Zalabak, P. S. (2015) . Fundamentals of organizational communication, answer the following questions Describe a group/team meeting in which you were the leader,...
-
A parking lot charges $3 for the first hour (or part of an hour) and $2 for each succeeding hour (or part), up to a daily maximum of $10. (a) Sketch a graph of the cost of parking at this lot as a...
-
The following data were obtained for a proprietary salad dressing tested at $22^{\circ} \mathrm{C}$ in a cup and bob viscometer (cup diameter $=4.2 \mathrm{~cm}$, bob diameter $=4.01 \mathrm{~cm}$,...
-
An inclined tube manometer with a reservoir is used to measure the pressure gradient in a large pipe carrying oil $(\mathrm{SG}=0.91)$ (see Figure $\mathrm{P} 4.5)$. The pipe is inclined at an angle...
-
A mercury manometer is used to measure the pressure drop across an orifice that is mounted in a vertical pipe. A liquid with a density of $0.87 \mathrm{~g} / \mathrm{cm}^{3}$ is flowing upward...

Study smarter with the SolutionInn App


