1. Name the following compounds Br te a) b) COH 2. Give the common and IUPAC...
Fantastic news! We've Found the answer you've been seeking!
Question:




Transcribed Image Text:
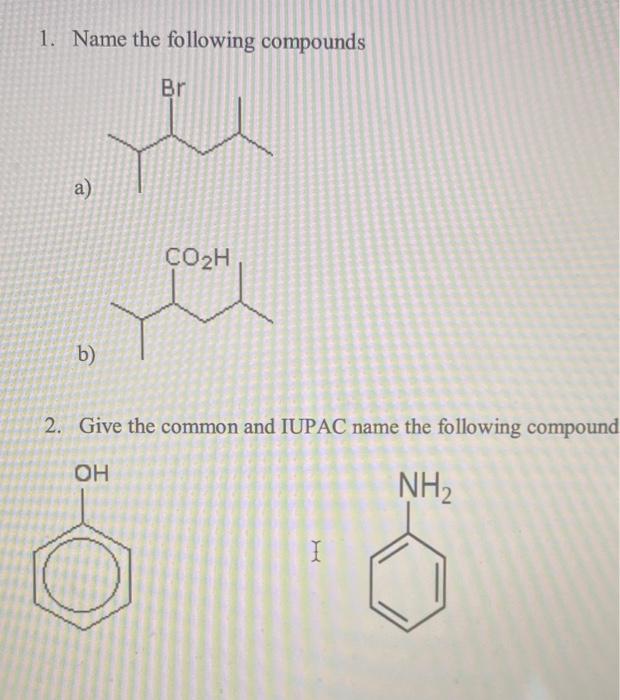
1. Name the following compounds Br te a) b) CO₂H 2. Give the common and IUPAC name the following compound OH NH₂ I 3. Give the IUPAC name of each of the following compound a) CH3CH2CH2CH CHO b) CH3COCH₂CH₂CH3 c) CH3COOCH₂CH3 4. Which of the following compounds is a carboxylic acid? a. CH3CH₂COOH b. CH3CH₂OCH3 c. CH3CH₂CH₂OH d. CH3CH2CHO 5. Which of the following compounds is a ketone? a. CH3CH₂COOH b. CH3CH2CHO C. CH3CH₂CH₂OH d. CH3COCH 3 6. What is the name of the linear hydrocarbon with the molecular formula C7H16? a. hexane b. heptane c. decane d. undecane 7. What is the IUPAC name of the following compound? a. 2-ethyl-4-methylpentane b. 2,4-dimethylhexane c. 3,5-dimethylhexane 1,1,3-trimethylpentane d. 8. What is the IUPAC name of the following compound? a. 2-isopropyl-5-methylpentane b. 5-isopropyl-2-methylpentane c. 2,3,5-trimethylhexane d. 1.2-diisopropylpropane 9. What is the IUPAC name of the following compound? a. 1,3-dimethylhexene b. 2,4-dimethyl-2-hexene c. 2,4-dimethyl-1-hexene d. 3,5-dimethyl-4-hexene 10. What is the IUPAC name of the following compound? CH3CH₂CH₂C=C-CH(CH3)2 a. 1-isopropyl-1-pentyne b. 2-methyl-3-heptyne c. 6-methyl-4-heptyne d. isopropyl propyl ethyne 1. Name the following compounds Br te a) b) CO₂H 2. Give the common and IUPAC name the following compound OH NH₂ I 3. Give the IUPAC name of each of the following compound a) CH3CH2CH2CH CHO b) CH3COCH₂CH₂CH3 c) CH3COOCH₂CH3 4. Which of the following compounds is a carboxylic acid? a. CH3CH₂COOH b. CH3CH₂OCH3 c. CH3CH₂CH₂OH d. CH3CH2CHO 5. Which of the following compounds is a ketone? a. CH3CH₂COOH b. CH3CH2CHO C. CH3CH₂CH₂OH d. CH3COCH 3 6. What is the name of the linear hydrocarbon with the molecular formula C7H16? a. hexane b. heptane c. decane d. undecane 7. What is the IUPAC name of the following compound? a. 2-ethyl-4-methylpentane b. 2,4-dimethylhexane c. 3,5-dimethylhexane 1,1,3-trimethylpentane d. 8. What is the IUPAC name of the following compound? a. 2-isopropyl-5-methylpentane b. 5-isopropyl-2-methylpentane c. 2,3,5-trimethylhexane d. 1.2-diisopropylpropane 9. What is the IUPAC name of the following compound? a. 1,3-dimethylhexene b. 2,4-dimethyl-2-hexene c. 2,4-dimethyl-1-hexene d. 3,5-dimethyl-4-hexene 10. What is the IUPAC name of the following compound? CH3CH₂CH₂C=C-CH(CH3)2 a. 1-isopropyl-1-pentyne b. 2-methyl-3-heptyne c. 6-methyl-4-heptyne d. isopropyl propyl ethyne
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Give both common and IUPAC names for the following compounds: a. CH3Br b. CH3CH2Cl c. CH2Cl2 d. (CH3)2CHBr e. CHI3 f. (CH3)3CCl g. CH3CH2CH2CH2F
-
Name the following compounds by IUPAC rules: a. b. H-C CH,CH-CH
-
Name the following compounds by the IUPAC system: a. CH3CH=C(CH2CH2CH3)2 b. (CH3)2CHCH"CHCH3 c. g. CH3-C-C-CH-CH, h. k.
-
Identify each process as endothermic or exothermic and indicate the sign of H. (a) Sweat evaporating from skin (b) Water freezing in a freezer (c) Wood burning in a fire
-
Auditing Automated Controls. You are auditing payroll for Alexander Inc., which uses computerized processing for its payroll transactions; the various steps in Alexanders system follow: As employees...
-
Segmented Reporting Cineplex Corporation is a diversified company that operates in five different industries: A, B, C, D, and E. The following information relating to each segment is available for...
-
Repeat Example 7.3 using \(10 \mathrm{~kg} / \mathrm{h}\) of solvent in each stage. Data From Example 7.3:- The feed of Example 7.2 is extracted three times with pure chloroform at 298 K, using 8...
-
The stockholders equity accounts of Lawrence Company have the following balances on December 31, 2010. Common stock, $10 par, 200,000 shares issued and outstanding............$2,000,000 Paid-in...
-
A 480-g iron pan is heated on a stove to a temperature of 130 C. The pan is then dropped into a partially-filled sink of water at 20 C. The final temperature reached by the sink and the pan is 40 C....
-
An analysis of transactions for Foley & Co. was presented in E18. Assume that August is the companys first month of business. In E18, An analysis of the transactions made by Foley & Co., a certified...
-
Pheasant Corporation, a calendar year taxpayer, has $400,000 of current E & P and a deficit in accumulated E & P of $180,000. If Pheasant pays a $600,000 distribution to its shareholders on December...
-
Depreciation expense for 2021 was $1,400. Assuming zero salvage value and straight-line depreciation for Season's depreciable assets, approximately what is the useful life of Season's depreciable...
-
How do digital technologies and virtual interactions shape symbolic communication and social interactions, and what implications does this have for contemporary understandings of symbolic...
-
products : Tank Gauging, , magnetic flux meters ,Vortex meters for flow measurement ,Transmitting devices for sound waves . We have to take this product and find How can yokogawa do good than...
-
TARGET: Apply a PESTLE to the industry in which your selected company functions within. Select three categories in the PESTLE, and identify one factor for each of the three categories that is...
-
Provide an example of over-delegation, under-delegation, and refusal to accept a legitimate delegation. Identify a leadership strategy that you would utilize for each example. Analyze problem...
-
Find the present value of a perpetuity that pays n (mod 3) dollars at the end of year n. (So it pays $1 in year 1, $2 in year 2, $0 in year 3, $1 in year 4 and so on). Assume the annual effective...
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
What is the difference in energy between the two levels responsible for the violet emission line of the calcium atom at 422.7 nm?
-
Which salt would have its solubility more affected by changes in pH, silver chloride or silver cyanide?
-
Arginine vasopressin is a pituitary hormone. It helps regulate the amount of water in the blood by reducing the flow of urine from the kidneys. An aqueous solution containing 21.6 mg of vasopressin...
-
a. Show that the mean-squared forecast error \(E\left[\left(\hat{y}_{T+1}-y_{T+1} ight)^{2} \mid I_{T} ight]\) for a forecast \(\hat{y}_{T+1}\), that depends only on past information \(I_{T}\), can...
-
Consider the AR(1) model \(y_{t}=\delta+\theta y_{t-1}+e_{t}\) where \(|\theta|)=0\) and \(\operatorname{var}\left(e_{t} \mid I_{t-1} ight)=\sigma^{2}\). Let \(\bar{y}_{-1}=\sum_{t=2}^{T} y_{t}...
-
Consider a stationary model that combines the \(\operatorname{AR}(2)\) model \(y_{t}=\delta+\theta_{1} y_{t-1}+\theta_{2} y_{t-2}+e_{t}\) with an \(\mathrm{AR}(1)\) error model \(e_{t}=ho...

Study smarter with the SolutionInn App


