Question: Camila Fashion is a clothing retailer owned by proprietor, Christine. The business just started its operation on last August 2022. The following transactions were
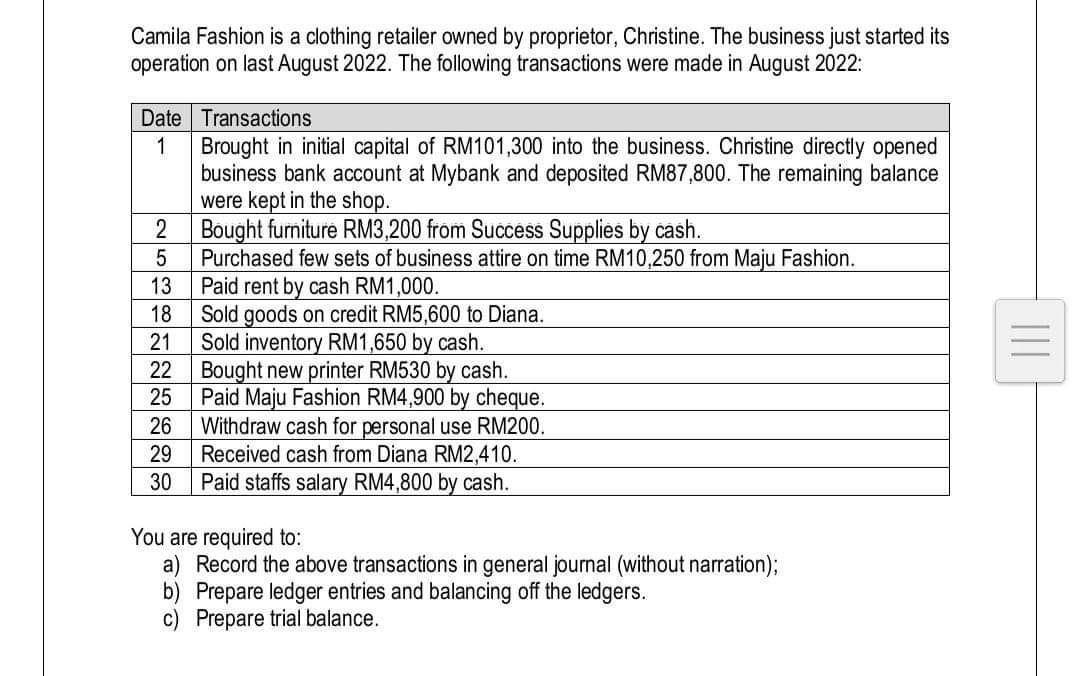
Camila Fashion is a clothing retailer owned by proprietor, Christine. The business just started its operation on last August 2022. The following transactions were made in August 2022: Date Transactions 1 2 5 13 18 21 22 25 26 29 30 Brought in initial capital of RM101,300 into the business. Christine directly opened business bank account at Mybank and deposited RM87,800. The remaining balance were kept in the shop. Bought furniture RM3,200 from Success Supplies by cash. Purchased few sets of business attire on time RM10,250 from Maju Fashion. Paid rent by cash RM1,000. Sold goods on credit RM5,600 to Diana. Sold inventory RM1,650 by cash. Bought new printer RM530 by cash. Paid Maju Fashion RM4,900 by cheque. Withdraw cash for personal use RM200. Received cash from Diana RM2,410. Paid staffs salary RM4,800 by cash. You are required to: a) Record the above transactions in general journal (without narration); b) Prepare ledger entries and balancing off the ledgers. c) Prepare trial balance.
Step by Step Solution
3.42 Rating (149 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


