Question: 1. Provide a stepwise synthesis for the following. (3 points) 2. Provide the mechanism for following reaction. (3 points) O 3. Provide the reagents
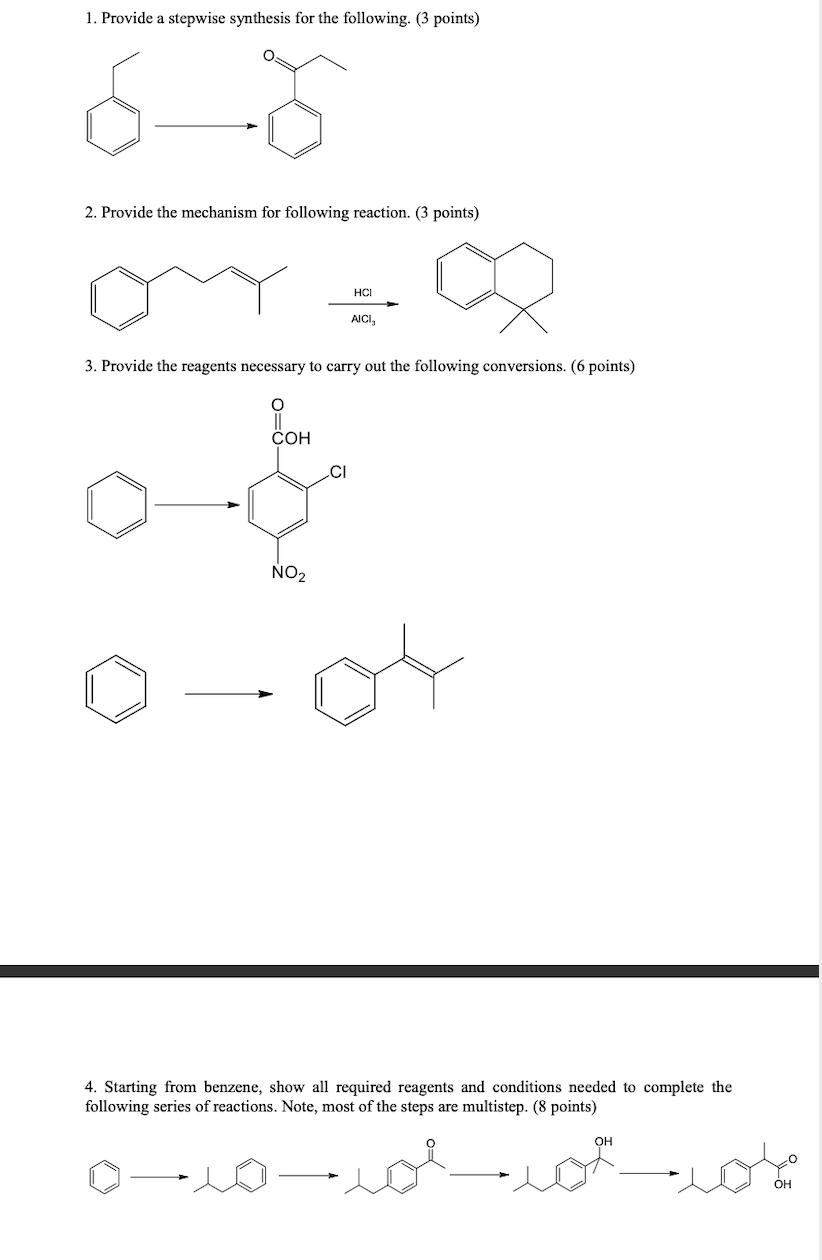
1. Provide a stepwise synthesis for the following. (3 points) 2. Provide the mechanism for following reaction. (3 points) O 3. Provide the reagents necessary to carry out the following conversions. (6 points) COH NO HCI CI AICI, 4. Starting from benzene, show all required reagents and conditions needed to complete the following series of reactions. Note, most of the steps are multistep. (8 points) OH jot jot OH
Step by Step Solution
3.31 Rating (160 Votes )
There are 3 Steps involved in it
Answer CH3 convention of A 1 Ethyl benzene urdises benzylic carbon to benzoic acid mec... View full answer

Get step-by-step solutions from verified subject matter experts


