1. The magnitude of the electrostatic force between two identical ions that are separated by a...
Fantastic news! We've Found the answer you've been seeking!
Question:

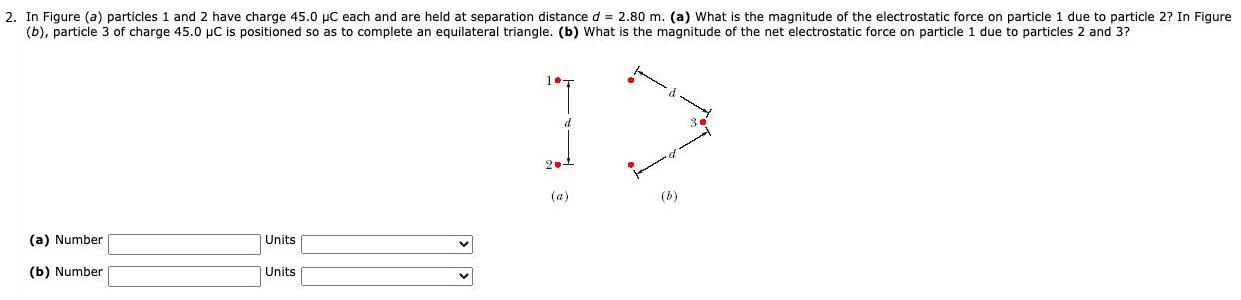


Transcribed Image Text:
1. The magnitude of the electrostatic force between two identical ions that are separated by a distance of 6.5 x 10-10 m is 78.65 x 109 N. (a) What is the charge of each ion? (b) How many electrons are "missing" from each ion (thus giving the ion its charge imbalance)? (a) Number Units (b) Number Units 2. In Figure (a) particles 1 and 2 have charge 45.0 µC each and are held at separation distance d = 2.80 m. (a) What is the magnitude of the electrostatic force on particle 1 due to particle 2? In Figure (b), particle 3 of charge 45.0 µC is positioned so as to complete an equilateral triangle. (b) What is the magnitude of the net electrostatic force on particle 1 due to particles 2 and 3? d 2 (a) (b) (a) Number Units (b) Number Units 3. Two particles are fixed on an x axis. Particle 1 of charge 59.0 µC is located at x = -7.55 cm; particle 2 of charge Q is located at x = 31.4 cm. Particle 3 of charge magnitude 53.7 µC is released from rest on the y axis at y = 7.55 cm. What is the value of Q if the initial acceleration of particle 3 is in the positive direction of (a) the x axis and (b) the y axis? (a) Number Units (b) Number Units 4. Two equally charged particles, held 4.3 x 103 m apart, are released from rest. The initial acceleration of the first particle is observed to be 8.0 m/s2 and that of the second to be 11 m/s?. If the mass of the first particle is 8.5 x 10 kg, what are (a) the mass of the second particle and (b) the magnitude of the charge of each particle? (a) Number Units (b) Number Units 1. The magnitude of the electrostatic force between two identical ions that are separated by a distance of 6.5 x 10-10 m is 78.65 x 109 N. (a) What is the charge of each ion? (b) How many electrons are "missing" from each ion (thus giving the ion its charge imbalance)? (a) Number Units (b) Number Units 2. In Figure (a) particles 1 and 2 have charge 45.0 µC each and are held at separation distance d = 2.80 m. (a) What is the magnitude of the electrostatic force on particle 1 due to particle 2? In Figure (b), particle 3 of charge 45.0 µC is positioned so as to complete an equilateral triangle. (b) What is the magnitude of the net electrostatic force on particle 1 due to particles 2 and 3? d 2 (a) (b) (a) Number Units (b) Number Units 3. Two particles are fixed on an x axis. Particle 1 of charge 59.0 µC is located at x = -7.55 cm; particle 2 of charge Q is located at x = 31.4 cm. Particle 3 of charge magnitude 53.7 µC is released from rest on the y axis at y = 7.55 cm. What is the value of Q if the initial acceleration of particle 3 is in the positive direction of (a) the x axis and (b) the y axis? (a) Number Units (b) Number Units 4. Two equally charged particles, held 4.3 x 103 m apart, are released from rest. The initial acceleration of the first particle is observed to be 8.0 m/s2 and that of the second to be 11 m/s?. If the mass of the first particle is 8.5 x 10 kg, what are (a) the mass of the second particle and (b) the magnitude of the charge of each particle? (a) Number Units (b) Number Units
Expert Answer:

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these mathematics questions
-
The magnitude of the electrostatic force between two identical ions that are separated by u distance of 5.0 x 1010 m is 3.7 x 109 N. (a) What is the charge of each ion? (b) How many electrons are...
-
The magnitude of the electrostatic force between two point charges Q and q of the same sign is given by F(x) = kQq/x 2 , where x is the distance (measured in meters) between the charges and k = 9 10...
-
What would be the magnitude of the electrostatic force between two 1.00 C point charges separated by a distance of? (a) 1.00 m and (b) 1.00 km if such point charges existed (they do not) and this...
-
CASE STUDY. Case Study Chapters 1 and 2. Please post both case studies in Assignment Drop Box as one MS Word apa formate document. Note: See template provided for case study papers. Chapter 1 - Listo...
-
Rasheed wishes to postpone for 90 days the payment of $450 that he owes to Roxanne. If money now earns 6.75%, what amount can he reasonably expect to pay at the later date?
-
A number of matrices are defined as Answer the following questions regarding these matrices;(a) What are the dimensions of the matrices?(b) Identify the square, column, and row matrices.(c) What are...
-
This case deals with several issues regarding contract formation under the UCC. Logan and Kanawha Coal agreed to purchase coal from Detherage via a fax dated March 9, 2010. The fax stated that it had...
-
1. Assume you are a financial analyst who works for a major brokerage company that is heavily invested in Bre-X Minerals. a. In what ways would investigating management and directors help determine...
-
Suzanne Michaels from Howie's Pizza was asked to calculate the break-even point for a new line of gourmet pizzas (prebaked, ready to take home). The selling price will be $25 per pizza. The labor...
-
A man from Vietnam is in your office because his 11-year-old daughter has been having trouble in school. The school suggested the daughter be tested by your agency. You are doing the intake, but only...
-
Frankie and Johnnie are social work colleagues counseling women in a shelter for battered women. Frankie has seen Johnnie out on dates with some of the women from the shelter. While Frankie and...
-
Tooth enamel has a chemical formula of Ca5 (PO4)3 (OH). What is the percent composition of each element. Give 4 sig figs for each percentage. Ca = P = O = H = do % % % %
-
Given that atomic radius for copper is 0 . 1 2 8 nm , calculate the linear density for copper in [ 1 0 0 ] direction. ( Copper has FCC crystal structure )
-
Does a cation gain protons to form a positive charge or does it lose electrons? a) The protons in the nucleus of a cation formed due to the loss of protons and electrons. b) The cation gains protons...
-
A balanced net ionic equation for the reaction of AgNO3(aq) with KCl(aq). a. AgNO3(aq) + KCl(aq) AgCl(aq) + KNO3(s) b. Ag+(aq) + Cl-(aq) AgCl(s) c. Ag+(aq) + NO3-(aq) + K+(aq) + Cl-(aq) AgCl(s) +...
-
Identify the number of structural isomers that have the formula, C3H8. A) 1 B) 2 C) 3 D) 4 E) 5
-
According to the International Organization for Standardization (ISO9000:2000), how is quality defined? Group of answer choices Conformance to requirements: a project's processes and deliverables...
-
Assessing simultaneous changes in CVP relationships Braun Corporation sells hammocks; variable costs are $75 each, and the hammocks are sold for $125 each. Braun incurs $240,000 of fixed operating...
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
High-mass radio-nuclides, which may be either alpha or beta emitters, belong to one of four decay chains, depending on whether their mass number A is of the form 4n,4n + 1, 4n + 2, or 4n + 3, where n...
-
One long wire lies along an x axis and carries a current of 30 A in the positive x direction. A second long wire is perpendicular to the xy plane, passes through the point (0, 4.0 m, 0) and carries a...
-
Why is gifting an important estate planning tool? Why are assets that grow in value recommended as gifts?
-
Following his death in 2012, Zane Wulster's gross taxable estate was valued at $3,300,000. He has made a total of $200,000 of gifts that exceeded the annual gift tax exclusion. a. What is the amount...
-
Explain how the portable estate exemption, resulting from the 2010 Tax Relief Act, altered estate planning using family trusts.

Study smarter with the SolutionInn App


