1. The rate law for the reaction 2NO2(g) + F2(g) are two proposed mechanisms for this...
Fantastic news! We've Found the answer you've been seeking!
Question:
![1. The rate law for the reaction 2NO2(g) F2(g) 2NO2F(g) is rate kNO2][F2]. Below are two proposed mechanisms for this reactio](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/08/6308b52b60f78_0516308b52b1821f.jpg)
![Step l: NO (g) + Br2 (g) NOBr2 (g) Step 2: NOBr2 (g) + NO (g) 2 NOBr (g) The rate law for the reaction is rate kINO [Br2] A.](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/08/6308b52c4eef9_0516308b52bec50f.jpg)
Transcribed Image Text:
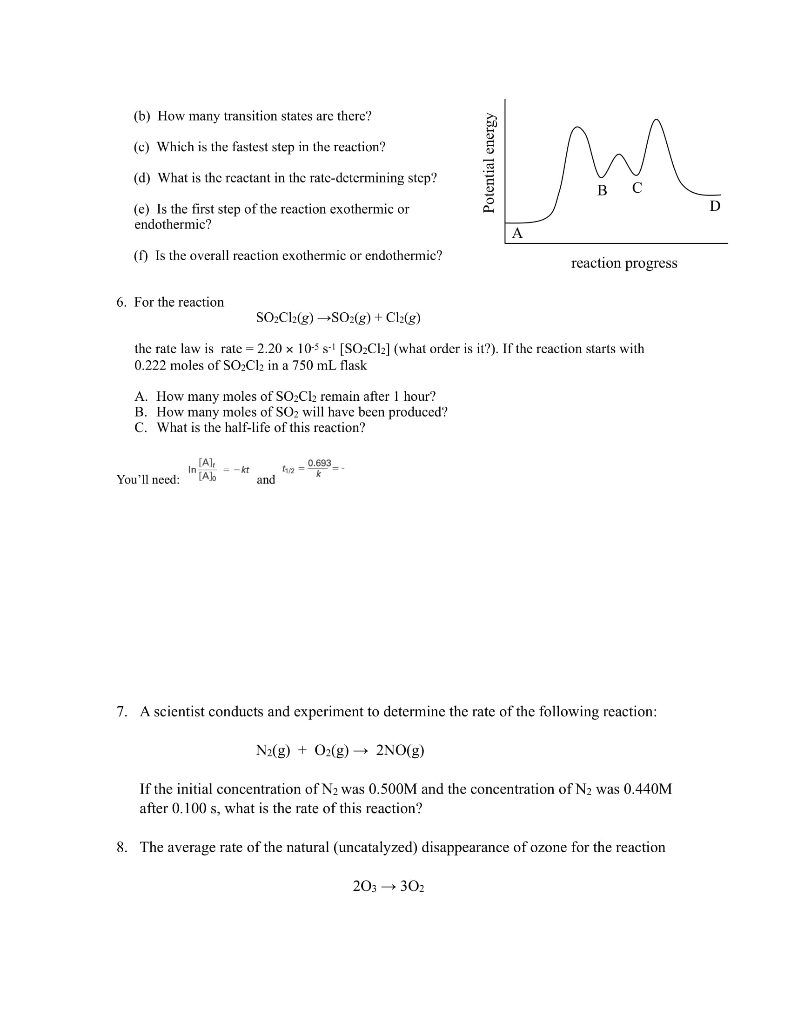
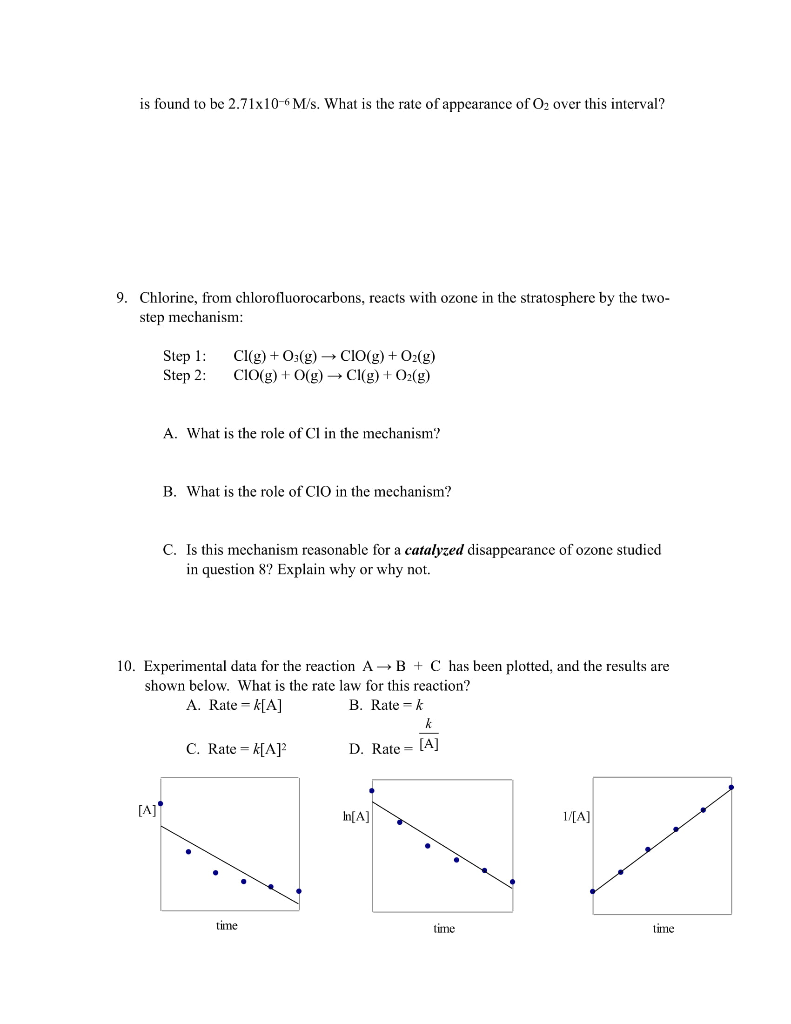
1. The rate law for the reaction 2NO2(g) + F2(g) are two proposed mechanisms for this reaction. plausible. (a) Step 1: NO2 + F2 Step 2: NO₂+ F (b) Step 1: NO2 + F2 Step 2: NO₂F2 Step 3: F+NO₂ (a) I2 I + H₂ H₂I + I k2 k3 k₁ NO₂F+F (Slow) k₂ NO₂F k₁ k3 2NO₂F(g) is rate = k[NO2][F2]. Below Show that both of these mechanisms are NO₂F2 (Fast) NO₂F (Fast) NO₂F + F (Slow) 2. Derive the overall reaction and the rate law for each mechanism shown below. k₁ K₁ 21 K₂ (fast) H₂I (fast) 2HI (slow) (Fast) (b) NO + Brz NOBr2 + NO k₂ NOBrz (slow) 2NOBr (fast) 3. The reaction 2 NO (g) + Brz (g) →2 NOBr (g) is believed to follow the mechanism: Step 1: Step 2: The rate law for the reaction is rate = k[NO]²[Br₂]. NO (g) + Br2 (g) →NOBr2 (g) NOBr2 (g) + NO (g) - 2 NOBr (g) A. Which is the rate-determining step in the mechanism? B. Why is it believed that the overall reaction is NOT an elementary process? (Hint: collision state theory) 4. For the reaction 4A + B + C₂ A2B + 2AC, the following initial rates of reaction were found. Determine the rate law and the value of k (with units!) for this reaction. Experiment 1 2 3 4 [A]o (mol/L) 0.20 0.20 0.30 0.20 [B]o (mol/L) 0.30 0.60 0.30 0.60 [C₂]0 (mol/L) 0.20 0.20 0.20 0.60 Initial Rate (mol/ L-s) 6.8 x 10-6 2.7 x 10-5 1.0 x 10-5 2.7 x 10-5 5. Below is the reaction profile for the reaction of A to D. Use it to answer the following questions. (a) How many intermediates are there in this reaction? (b) How many transition states are there? (c) Which is the fastest step in the reaction? (d) What is the reactant in the rate-determining step? (e) Is the first step of the reaction exothermic or endothermic? (f) Is the overall reaction exothermic or endothermic? 6. For the reaction A. How many moles of SO₂Cl₂ remain after 1 hour? B. How many moles of SO₂ will have been produced? C. What is the half-life of this reaction? SO₂Cl2(g) →SO2(g) + Cl₂(g) the rate law is rate = 2.20 x 105 s-¹ [SO₂Cl2] (what order is it?). If the reaction starts with 0.222 moles of SO₂Cl2 in a 750 mL flask You'll need: [A], [A] = kt and 11/2 = 0.693_ m reaction progress 203 → 302 7. A scientist conducts and experiment to determine the rate of the following reaction: N₂(g) + O₂(g) →→→ 2NO(g) If the initial concentration of N₂ was 0.500M and the concentration of N₂ was 0.440M after 0.100 s, what is the rate of this reaction? 8. The average rate of the natural (uncatalyzed) disappearance of ozone for the reaction D is found to be 2.71x10-6 M/s. What is the rate of appearance of O₂ over this interval? 9. Chlorine, from chlorofluorocarbons, reacts with ozone in the stratosphere by the two- step mechanism: Step 1: Step 2: [A] Cl(g) + O3(g) → CIO(g) + O₂(g) CIO(g) + O(g) →→ Cl(g) + O₂(g) A. What is the role of Cl in the mechanism? B. What is the role of CIO in the mechanism? C. Is this mechanism reasonable for a catalyzed disappearance of ozone studied in question 8? Explain why or why not. 10. Experimental data for the reaction AB + C has been plotted, and the results are shown below. What is the rate law for this reaction? A. Rate = k[A] B. Rate k C. Rate = k[A]² time k D. Rate: [A] In[A] time 1/[A] time 1. The rate law for the reaction 2NO2(g) + F2(g) are two proposed mechanisms for this reaction. plausible. (a) Step 1: NO2 + F2 Step 2: NO₂+ F (b) Step 1: NO2 + F2 Step 2: NO₂F2 Step 3: F+NO₂ (a) I2 I + H₂ H₂I + I k2 k3 k₁ NO₂F+F (Slow) k₂ NO₂F k₁ k3 2NO₂F(g) is rate = k[NO2][F2]. Below Show that both of these mechanisms are NO₂F2 (Fast) NO₂F (Fast) NO₂F + F (Slow) 2. Derive the overall reaction and the rate law for each mechanism shown below. k₁ K₁ 21 K₂ (fast) H₂I (fast) 2HI (slow) (Fast) (b) NO + Brz NOBr2 + NO k₂ NOBrz (slow) 2NOBr (fast) 3. The reaction 2 NO (g) + Brz (g) →2 NOBr (g) is believed to follow the mechanism: Step 1: Step 2: The rate law for the reaction is rate = k[NO]²[Br₂]. NO (g) + Br2 (g) →NOBr2 (g) NOBr2 (g) + NO (g) - 2 NOBr (g) A. Which is the rate-determining step in the mechanism? B. Why is it believed that the overall reaction is NOT an elementary process? (Hint: collision state theory) 4. For the reaction 4A + B + C₂ A2B + 2AC, the following initial rates of reaction were found. Determine the rate law and the value of k (with units!) for this reaction. Experiment 1 2 3 4 [A]o (mol/L) 0.20 0.20 0.30 0.20 [B]o (mol/L) 0.30 0.60 0.30 0.60 [C₂]0 (mol/L) 0.20 0.20 0.20 0.60 Initial Rate (mol/ L-s) 6.8 x 10-6 2.7 x 10-5 1.0 x 10-5 2.7 x 10-5 5. Below is the reaction profile for the reaction of A to D. Use it to answer the following questions. (a) How many intermediates are there in this reaction? (b) How many transition states are there? (c) Which is the fastest step in the reaction? (d) What is the reactant in the rate-determining step? (e) Is the first step of the reaction exothermic or endothermic? (f) Is the overall reaction exothermic or endothermic? 6. For the reaction A. How many moles of SO₂Cl₂ remain after 1 hour? B. How many moles of SO₂ will have been produced? C. What is the half-life of this reaction? SO₂Cl2(g) →SO2(g) + Cl₂(g) the rate law is rate = 2.20 x 105 s-¹ [SO₂Cl2] (what order is it?). If the reaction starts with 0.222 moles of SO₂Cl2 in a 750 mL flask You'll need: [A], [A] = kt and 11/2 = 0.693_ m reaction progress 203 → 302 7. A scientist conducts and experiment to determine the rate of the following reaction: N₂(g) + O₂(g) →→→ 2NO(g) If the initial concentration of N₂ was 0.500M and the concentration of N₂ was 0.440M after 0.100 s, what is the rate of this reaction? 8. The average rate of the natural (uncatalyzed) disappearance of ozone for the reaction D is found to be 2.71x10-6 M/s. What is the rate of appearance of O₂ over this interval? 9. Chlorine, from chlorofluorocarbons, reacts with ozone in the stratosphere by the two- step mechanism: Step 1: Step 2: [A] Cl(g) + O3(g) → CIO(g) + O₂(g) CIO(g) + O(g) →→ Cl(g) + O₂(g) A. What is the role of Cl in the mechanism? B. What is the role of CIO in the mechanism? C. Is this mechanism reasonable for a catalyzed disappearance of ozone studied in question 8? Explain why or why not. 10. Experimental data for the reaction AB + C has been plotted, and the results are shown below. What is the rate law for this reaction? A. Rate = k[A] B. Rate k C. Rate = k[A]² time k D. Rate: [A] In[A] time 1/[A] time
Expert Answer:
Answer rating: 100% (QA)
Answer 1 For mechanism a the Rate determining step RDS is the slow step step 1 therefore rate k ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The rate law for the reaction in Exercise 22.1b was found to be v = k[A][BF, What are the units of k? Express the rate law in terms of the rates of formation and consumption of (a) A, (b) C.
-
The rate law for the reaction in Exercise 22.2b was reported as d[C]/dt = k[A] [B][C]-l Express the rate law in terms of the reaction rate; what are the units for k in each case?
-
The rate law for the reaction 2NOBr(g) 2NO(g) + Br2(g) at some temperature is a. If the half- life for this reaction is 2.00 s when [NOBr]0 = 0.900 M, calculate the value of k for this reaction. b....
-
Show that for a linear ODE y' + p(x)y = r(x) with continuous p and r in |x - x 0 | < a Lipschitz condition holds. This is remarkable because it means that for a linear ODE the continuity of f(x, y)...
-
Access the financial statements and related disclosure notes of Ford Motor Company from its website at corporate.ford.com. In Ford's balance sheet, deferred income taxes in 2013 are reported as both...
-
A six-foot person walks from the base of a broadcasting tower directly toward the tip of the shadow cast by the tower. When the person is 132feet from the tower and 3feet from the tip of the shadow,...
-
Refer to the statements for Google in Appendix A. For the year ended December 31, 2015, what was its debt-to-equity ratio? What does this ratio tell us? Data From Statement Google In Appendix A...
-
The budget director of Gourmet Grill Company requests estimates of sales, production and other operating data from the various administrative units every month. Selected information concerning sales...
-
1. (10) A $10 000 bond was issued on January 1st 2015 with a coupon rate of 9.7% and a redemption date of January 1st 2025. What is the purchase price of the bond on January 1st, 2022 when the yield...
-
You have been employed as a consultant to Golf R Us, Inc. The company has been having problems with account receivables and they want to use various dunning methods to better their receivables. But...
-
How Mutual Funds Reflect Market Efficiency Assumptions?Reviewthe Vanguard 500 Index Fund (VFIAX) overview, performance and fees. If an investor had invested $10,000 in VFIAX in December of 2010,...
-
Do you agree or disagree with this statement? Firms minimize costs; thus, a firm earning short-run economic profits will choose to produce at the minimum point on its average total cost curve.
-
Flexible work schedules are becoming more common in the workplace, with an increasing number of companies adopting new policies to meet a growing demand for this flexibility from todays labor force....
-
Many countries follow the local/foreigner price discrimination strategy. Why do you think it is unusual in the United States?
-
What characteristics of a company do you think influence it to use flex time with its workers versus one which has employer-directed irregular hours?
-
The demand for land is a derived demand. Think of a popular location near your school. What determines the demand for land in that area? What outputs are sold by businesses located there? Discuss the...
-
Explain that How the concept of organizational silence manifest in different types of organizations?
-
Sheldon and Leonard had a million-dollar idea. In order to make it happen, they have to do special research first. Only Kripke can help them in this matter. But Kripke is known to be the first-class...
-
For the reaction 2H2O(g) 2H2(g) + O2(g) K = 2.4 10-3 at a given temperature. At equilibrium in a 2.0- L container, it is found that [H2O(g)] = 1.1 10-1 M and [H2(g)] = 1.9 10-2 M. Calculate the...
-
Papaverine hydrochloride (abbreviated papH+Cl; molar mass = 378.85 g/ mol) is a drug that belongs to a group of medicines called vasodilators, which cause blood vessels to expand, thereby increasing...
-
Consider the hypothetical reaction A + B + 2C 2D + 3E In a study of this reaction, three experiments were run at the same temperature. The rate is defined as 2d[B]/dt. Experiment 1: [A]0 = 2.0 M [B]0...
-
Orvil Companys net profit margin percentage is 4.5%, its total asset turnover is 2.4, and its equity multiplier is 1.5. What is the companys return on equity? a. 2.8% b. 7.2% c. 16.2% d. 10.7%
-
Obtain the most recent annual report or SEC filing 10-K of a publicly traded company that interests you. It may be a local company or it may be a company in an industry that you would like to know...
-
Endless Mountain Company manufactures a single product that is popular with outdoor recreation enthusiasts. The company sells its product to retailers throughout the northeastern quadrant of the...

Study smarter with the SolutionInn App


