If 25.0 mL of a 6.0 M HNO3 solution is diluted to 200 ml, what is...
Fantastic news! We've Found the answer you've been seeking!
Question:
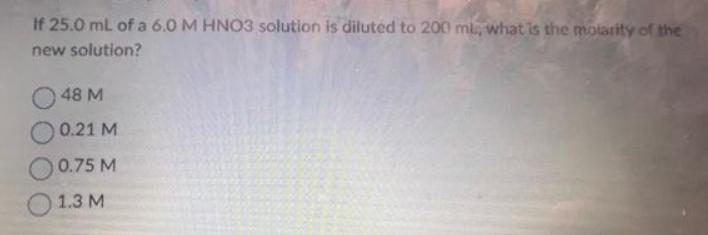
Transcribed Image Text:
If 25.0 mL of a 6.0 M HNO3 solution is diluted to 200 ml, what is the molarity of the new solution? 48 M 0.21 M 0.75 M 1.3 M If 25.0 mL of a 6.0 M HNO3 solution is diluted to 200 ml, what is the molarity of the new solution? 48 M 0.21 M 0.75 M 1.3 M
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Answer ... View the full answer

Posted Date:
Students also viewed these mathematics questions
-
If 12.00 L of a 6.00 M HNO3 solution needs to be diluted to 0.750 M, what will be its final volume?
-
QUESTION 21 3 points Save Answ You would like to determi QUESTION 21 3 points Save Answ You would like to determine the knowledge of geography of people aged 17 or older in the Capital Region. You...
-
If 1.00 mL of a 2.25 M H2SO4 solution needs to be diluted to 1.00 M, what will be its final volume?
-
Prove the following statement: If a transformer having a series impedance Ze is connected as an autotransformer, its per-unit series impedance Z as an autotransformer will be SE Zoq NSE + NC Note...
-
International Data Systems information on revenue and costs is only relevant up to a sales volume of 105,000 units. After 105,000 units, the market becomes saturated and the price per unit falls from...
-
The records at the end of January 2015 for Young Company showed the following for a particular kind of merchandise: Inventory, December 31, 2014, at FIFO: 19 Units @ $16 = $304 Inventory, December...
-
In what ways can the quest for information security be compared to striving for total quality?
-
Presented below are two independent situations. (a) On March 3, Cornwell Appliances sells $680,000 of its receivables to Marsh Factors Inc. Marsh Factors assesses a finance charge of 3% of the amount...
-
Formulate an opinion on the death penalty in SouthAfrica Explain why the death penalty is unconstitutional in accordance with the S V Makwanyane 1993 (3) SA 391 (CC) case Define the law of criminal...
-
A rotary lawnmower blade rotates at 3500 rev/min. The steel blade has a uniform cross section 1/8 in thick by 1 ¼ in wide, and has a ½-in-diameter hole in the center as shown in the...
-
1.What can be done to maximize safety in a dock area? 2.How are signs color-coded to identify specific dangers? 3.Explain the classifications of fire and which fire extinguishers are used for each...
-
Marks plc and Spencer plc both base their dividend policy on the Lintner model. Both firms have a target payout rate of 4 0 % . Marks plc s adjustment rate is 0 . 2 and Spencer plc s adjustment...
-
3. Micrographia, a publication illustrating insects, sponges, as well as plant cells, was published by a. Robert Hooke. b. Antony van Leeuwenhoek. c. Hans Janssen. d. John Needham. 4. Low-power...
-
If the electoral system is replaced with a proportional representation system, what kind of effects will it have on Canadian politics and government?
-
Van Frank Telecommunications has a patent on a cellular transmission process. The company has amortized the patent on a straight - line basis since 2 0 2 0 , when it was acquired at a cost of $ 9...
-
If the speed of sound is determined by an experiment and found to be 3 5 5 m / s instead of the usual 3 4 5 m / s , what factor ( s ) beside experimental error could account for this increase?
-
2. Ramaryatt Inn is a hotel chain with properties in numerous markets. In their New York City property, they are currently allowed to book up to 230 rooms, which is 50% of their total capacity. They...
-
Under what conditions is the following SQL statement valid?
-
Which of the following is potentially a barrier to entry into a product market? a. patent protection on the design of the product b. economies of scale in the product market c. government licensing...
-
How do we know whether the monopolist is making a profit?
-
How does the monopolist decide what output to produce?

Study smarter with the SolutionInn App

