1. Use Equation 12 to calculate the volume of copper stock solution required to create a...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
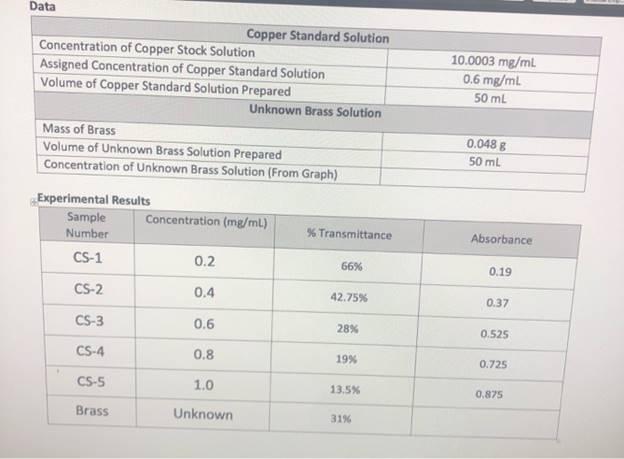
1. Use Equation 12 to calculate the volume of copper stock solution required to create a copper standard solution with the concentration assigned by your instructor (See data table above). 2. Calculate the mass of copper in the unknown brass solution by multiplying the concentration of copper in the unknown brass solution (mg/mL) by the volume of brass solution prepared (mL). Mass of Cu = (Conc. of Cu) x (Volume of Brass Solution) 3. Calculate the percent weight of copper in the unknown brass solution. %wt Cu =Mass of Copper/Mass of Brass x 100 Discussion State the results of the experiment. Which aspects of the experiment likely impacted your ability to determine an accurate percent weight of copper in the brass sample? If you elected to exclude the results from any of the standards when constructing your calibration curve, please explain your rationale. Data Copper Standard Solution Concentration of Copper Stock Solution Assigned Concentration of Copper Standard Solution Volume of Copper Standard Solution Prepared 10.0003 mg/ml 0.6 mg/mL 50 mL Unknown Brass Solution Mass of Brass Volume of Unknown Brass Solution Prepared Concentration of Unknown Brass Solution (From Graph) 0.048 8 50 ml Experimental Results Sample Number Concentration (mg/mL) Transmittance Absorbance CS-1 0.2 66% 0.19 CS-2 0.4 42.75% 0.37 CS-3 0.6 28% 0.525 CS-4 0.8 19% 0.725 CS-5 1.0 13.5% 0.875 Brass Unknown 31% 1. Use Equation 12 to calculate the volume of copper stock solution required to create a copper standard solution with the concentration assigned by your instructor (See data table above). 2. Calculate the mass of copper in the unknown brass solution by multiplying the concentration of copper in the unknown brass solution (mg/mL) by the volume of brass solution prepared (mL). Mass of Cu = (Conc. of Cu) x (Volume of Brass Solution) 3. Calculate the percent weight of copper in the unknown brass solution. %wt Cu =Mass of Copper/Mass of Brass x 100 Discussion State the results of the experiment. Which aspects of the experiment likely impacted your ability to determine an accurate percent weight of copper in the brass sample? If you elected to exclude the results from any of the standards when constructing your calibration curve, please explain your rationale. Data Copper Standard Solution Concentration of Copper Stock Solution Assigned Concentration of Copper Standard Solution Volume of Copper Standard Solution Prepared 10.0003 mg/ml 0.6 mg/mL 50 mL Unknown Brass Solution Mass of Brass Volume of Unknown Brass Solution Prepared Concentration of Unknown Brass Solution (From Graph) 0.048 8 50 ml Experimental Results Sample Number Concentration (mg/mL) Transmittance Absorbance CS-1 0.2 66% 0.19 CS-2 0.4 42.75% 0.37 CS-3 0.6 28% 0.525 CS-4 0.8 19% 0.725 CS-5 1.0 13.5% 0.875 Brass Unknown 31%
Expert Answer:
Answer rating: 100% (QA)
Explanation Q1 Using equation M1V1stock M2V2standard 100003 Vstock ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Use Eq. (10) to calculate the volume of the unit ball as a surface integral over the unit sphere?
-
Calculate the volume of CO2 at 10.0C and 746 mmHg pressure obtained by treating 25.0 g of Na2CO3 with an excess of hydrochloric acid.
-
Calculate the volume of a 0.156 M CuSO4 solution that would react with 7.89g of zinc.
-
Suppose that we have a linear program that is not in standard form. We could produce the dual by first converting it to standard form, and then taking the dual. It would be more convenient, however,...
-
Green Lighting Supply plans inventory levels (at cost) at the end of each month as follows: May, $271,000; June, $226,000; July, $209,000; and August, $241,000. Sales are expected to be June,...
-
Cullumber Inc. made a $34000 sale on account with the following terms: 2/15, n/30. if the company uses the net method to record sales made on credit, what is/are the debit(s) in the journal entry to...
-
Explain how and to what extent combinatorial problems can be solved using resolution.
-
Bryce Power Tool Company Kevin Ertle is the director of information technology (IT) for the Bryce Power Tool Company. A decision was made recently to upgrade Bryces legacy systems to a comprehensive...
-
The Merlin rocket engines developed by SpaceX can produce 7.41 x 105 N of instantaneous thrust with an exhaust speed of 3.05 x 103 m/s in vacuum. What mass of fuel does the engine burn each second to...
-
Cinder and PQ are associated [ITA 256(1)(a)] and connected [ITA 186(4)] for tax purposes. Cinder Inc. is a Canadian-controlled private corporation based in your province. The company operates a...
-
The board has agreed to consider building a student lounge on campus, but your team will need to convince them at the board meeting. Work with your group to come up with a detailed plan that includes...
-
What are two lessons-learned that you have gleaned from a recent project in your organisation? Was a lessonslearned workshop carried out? What Action Plans were generated to improve project...
-
Interview a fellow project manager in your class and ask them what kind of closure procedures they have previously used to complete a project. Also ask them to provide (a minimum of two) examples of...
-
Ask a colleague in the room about the status of a current project they are involved with. Ask them some easy and difficult questions and take note of any non-verbal cues that may reveal their inner...
-
Take a postcard-sized envelope, or a half-sheet of A4 paper, and create an elevator pitch (EP) for your group project. Share your pitch with a project team member to gain feedback and then refine the...
-
Reflect back on a recent project you have been involved in which necessitated contractual negotiations. How did the negotiations consider the four essential elements of a contract?
-
In a company, knowledge workers are ______. employees who contribute through manual labor, employees who contribute to a company by sharing what they know, managers who depend on employees who...
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
Which of the following diagrams represent diatomic molecules, polyatomic molecules, molecules that are not compounds, molecules that are compounds, or an elemental form of the substance? (b) (c)
-
Suggest a physical method that would enable you to separate hydrogen gas from neon gas.
-
In steelmaking, nonmetallic impurities such as P, S, and Si are removed as the corresponding oxides. The inside of the furnace is usually lined with CaCO3 and MgCO3, which decompose at high...
-
What are some of the advantages of equity financing?
-
You have been asked to consult for an entrepreneur who is assembling investors for a new professional sport league. A critical decision for the league will be whether to organize under a...
-
Is the yield curve typically upward or downward sloping? Why?

Study smarter with the SolutionInn App


