1) Which one of the following is characteristic of a base? A) produces H30 in water...
Fantastic news! We've Found the answer you've been seeking!
Question:





Transcribed Image Text:
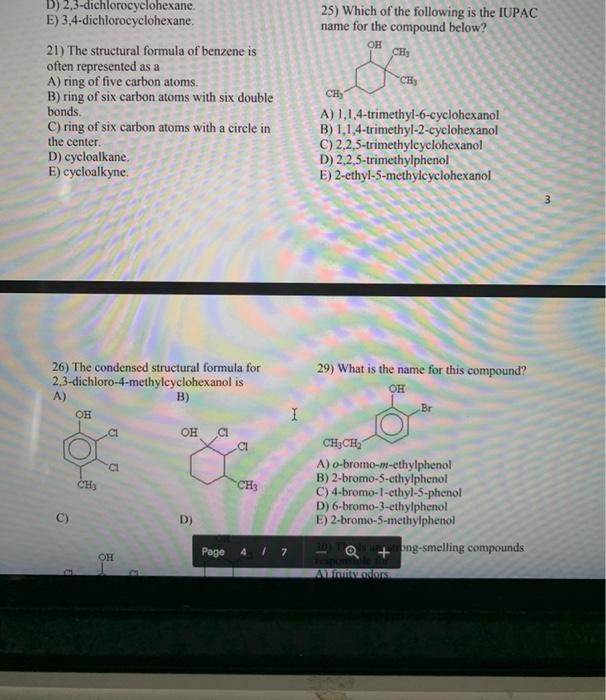
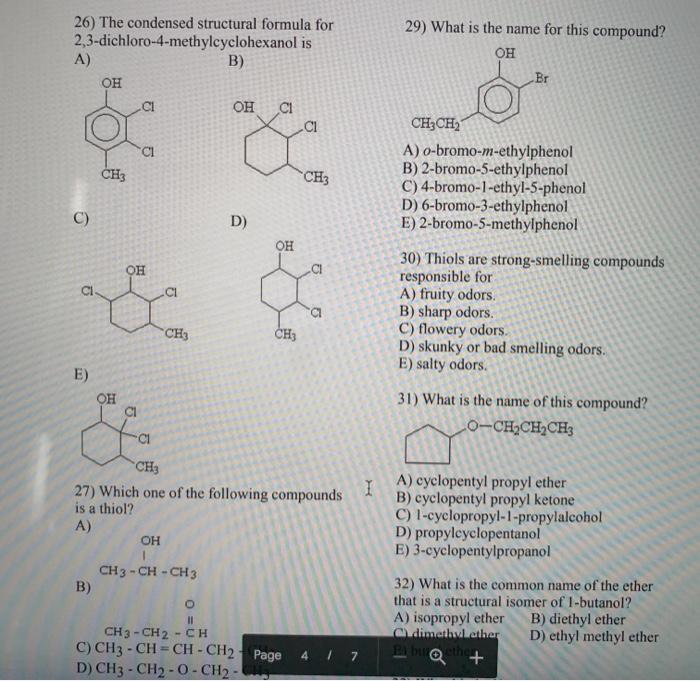
1) Which one of the following is characteristic of a base? A) produces H30¹ in water B) has a sour taste C) has a slippery, soapy feel D) turns blue litmus red E) is insoluble in water 2) Identify the Bronsted-Lowry acid in the following reaction.. H₂O + CO3²- -> HCO³ + OH A) H₂O B) CO²- C) HCO³- D) OH E) H₂CO₂ 3) Which of the following is correctly identified? A) NH3, strong acid B) NaOH, strong base C) HCI, weak acid D) H₂CO3, strong acid E) Ca(OH)2, weak base 4) What is the [OH-] in a solution that has a [H3O+] 2.0 x 10-4 M? A) 2.0 10-10 M B) 5.0 x C) 1.0 x 10-10 M 10-10 M D) 2.0 × 10-4 M E) 5.0 × 10-11 M I 5) Which of the following statements correctly describes the hydronium- hydroxide balance in the given solution? A) In acids, [OH-] is greater than [H3O+]. B) In bases, [OH-] = [H3O+]. C) In neutral solutions, [H3O+] = [O]. D) In bases, [OH-] is greater than [H3O+]. E) In bases, [OH-] is less than [H3O+]. 6) In which of the following are the pH values arranged from the most acidic to the most basic? A) 1, 3, 6, 8, 11, 14 B) 2, 5, 7, 9, 10, 1.1 C) 14, 10, 7, 4, 3, 1 D) 14, 10, 7, 1, 3, 5 E) 7, 10, 14, 4, 3, 11 7) What is the pH of a solution with [H3O+] = 3.0 × 10-3 M? A) 3.0 × 10-3 C) 3.0 E) 9.0 B) 2.52 D) -2.52 8) Which of the following is a neutralization reaction? A) KCI + NaNO3 1 KNO3 + NaCl B) HNO3 + KOH → H₂O + KNO3 C) H₂O + SO3 → H₂SO4 D) 4Na + O₂ 1 2Na₂O E) 2NO2 1 2NO + 0₂ 9) A 25.0 mL sample of H₂SO4 requires 20.0 mL of 2.00 M KOH for complete neutralization. What is the molarity of the acid? H₂SO4 + 2KOH → A) 2.00 M B) 2.50 M C) 0.800 M D) 1.60 M E) 1.25 M 10) Generally, a solution of an organic compound in water will be electrically A) highly conductive. B) highly ionized. C) nonconductive. K₂SO4 + 2H₂O D) insulated. E) charged. 11) Which of the following is NOT typical of most organic compounds? A) high melting point B) poor solubility in water C) low boiling point D) covalent bonding E) high flammability 12) What is the IUPAC name of this allena I 14) Which of the following pairs of compounds are structural isomers? A) CI CH3 CHCH2 CH3 and CH3CH2CHCH3 B) CICH2CH2CH2CH3 and CH3CH2CH2CH₂Cl C) D) Cl E) CH3CHCH3 and CH3CHCH2C1 CH₂C1 CH3 CH3CHCH3 and CH 3 C CH3 CH₂ CH3 and 12) What is the IUPAC name of this alkane? CH3 CH3-C-CH₂-CH-CH3 I CH3 CH₂-CH3 A) 4-ethyl-2,2-dimethylpentane B) 2-ethyl-4,4-dimethylpentane C) 2,2,4-trimethylhexane D) 2-ethyl-2,2-dimethylpentane E) 3,5,5-trimethylhexane 13) What type of compound is CH3CH₂CH₂CI? A) alkane C) cycloalkane E) isomer B) branched alkane D) haloalkane 15) One reason for the very large number of organic compounds (compared to the total number of inorganic compounds) is A) functional groups. B) structural isomerism. C) hydrogen bonding. D) the occurrence of organic compounds in living things. E) the electronegativity of carbon. 16) The functional group contained in the compound CH3CH₂COOCH, is a(n) A) thiol. B) carboxylic acid. C) amine, D) ester, E) amide. 2 17) What is the IUPAC name for the following compound? CH3 CI CH3-CH-C - CHÍCH 1 CH3 CH3 A) 4-chloro-4,5-dimethyl-2-hexene B) 3-chloro-1,3,4-trimethyl-1-pentene C) 3-chloro-2,3-dimethyl-4-hexene D) 3-chloro-2,3,5-trimethyl-4-pentene E) 3-chloro-1,3,4,4-tetramethyl-1-butene 18) The reaction for the combustion of heptane is C7H16+ 1102 → 7CO₂ + 8H₂O. How many liters of CO2 at STP are produced from the complete combustion of 2.00 moles of heptane? A) 44.8 L C) 157 L E) 314 L B) 22.4 L D) 246 L 19) The reaction of hydrogen (H₂) and propene using a platinum catalyst is called B) substitution. A) combustion. C) addition. D) neutralization. E) condensation. 20) The reaction of cyclohexene and Cl₂ yields A) 1-chlorocyclohexene. B) 1,2-dichlorocyclohexane. C) 1,3-dichlorocyclohexane. D) 2,3-dichlorocyclohexane. E) 3,4-dichlorocyclohexane. 22) Name the following compound. CH₂CH₂CH₂CH, CH₂CH₂CC CH CH₂CH₂CH, A) 3-butyl-3-propyl-1-pentyne B) 3-butyl-3-propyl-4-pentyne C) 3-ethyl-3-propyl-1-heptyne D) 5-ethyl-5-propyl-6-heptyne E) 3-ethyl-3-butyl-1-hexyne 23) Which set of reactants would give this compound? CI CH3-CH2-CH-CH₂Cl A) CH3CH₂CH₂CH3 + 2HCl B) CH3CH CHCH3 + 2HCl C) CH3CH2CH CH2 + 2HCl D) CH3CH CHCH3 + Cl2 E) CH3CH2CH CH2 + Cl2 I 24) What are the prefixes used to designate substituent positions on disubstituted benzene compounds? A) ortho-, meta-, and para-. B) cis- and trans- C) syn- and anti- D) All of the above. E) None of the above. 25) Which of the following is the IUPAC name for the compound below? D) 2,3-dichlorocyclohexane. E) 3,4-dichlorocyclohexane. 21) The structural formula of benzene is often represented as a A) ring of five carbon atoms. B) ring of six carbon atoms with six double bonds. C) ring of six carbon atoms with a circle in the center. D) cycloalkane. E) cycloalkyne. 26) The condensed structural formula for 2,3-dichloro-4-methylcyclohexanol is A) он CH3 a OH B) OH D) Cl 53 CH₂ Page 4 / 7 25) Which of the following is the IUPAC name for the compound below? ОН CH₂ CH₂ CH₂ A) 1,1,4-trimethyl-6-cyclohexanol B) 1,1,4-trimethyl-2-cyclohexanol C) 2,2,5-trimethylcyclohexanol D) 2,2,5-trimethylphenol E) 2-ethyl-5-methylcyclohexanol 29) What is the name for this compound? OH 5 Br CH₂CH₂ A) o-bromo-m-ethylphenol B) 2-bromo-5-ethylphenol C) 4-bromo-1-ethyl-5-phenol D) 6-bromo-3-ethylphenol E) 2-bromo-5-methylphenol 20TQ+ong-smelling compounds Al fruity odors m 26) The condensed structural formula for 2,3-dichloro-4-methylcyclohexanol is A) C) 3 OH CH3 E) OH B) CI CI OH CI CH3 B) OH CH3 -CH-CH3 OH D) OH CH3 CH3 fa CH3 27) Which one of the following compounds is a thiol? A) a O 11 CH3 - CH2 - CH C) CH3 -CH=CH-CH2 Page 4/7 D) CH3 - CH2 - 0-CH2-L I 29) What is the name for this compound? OH CH3CH₂ A) o-bromo-m-ethylphenol B) 2-bromo-5-ethylphenol C) 4-bromo-1-ethyl-5-phenol D) 6-bromo-3-ethylphenol E) 2-bromo-5-methylphenol Br 30) Thiols are strong-smelling compounds responsible for A) fruity odors. B) sharp odors. C) flowery odors. D) skunky or bad smelling odors. E) salty odors. 31) What is the name of this compound? O-CH,CH,CH A) cyclopentyl propyl ether B) cyclopentyl propyl ketone C) 1-cyclopropyl-1-propylalcohol D) propylcyclopentanol E) 3-cyclopentylpropanol 32) What is the common name of the ether that is a structural isomer of 1-butanol? A) isopropyl ether Cdimethyl ether Bburether + 350 B) diethyl ether D) ethyl methyl ether UITZ E) CH3 - SH 28) 1,2-ethanediol (ethylene diol) has uses that include A) antifreeze. C) production of synthetic fibers. D) solvent for ink. E) All of the above. B) solvent for paint. 34) In the oxidation of an alcohol to a ketone, there is A) a loss of hydrogen. B) a loss of oxygen. C) a loss of carbon. D) a gain of hydrogen. E) a gain of oxygen. 35) What is the product when the following compound is oxidized? OH CH3-CH2-CH-CH2-CH3 A) diethyl ketone B) 2-pentene C) pentanal D) diethyl ether E) pentane I 33) What kind of bonds do alcohols form between individual molecules? A) oxygen bonds B) hydrogen bonds C) single bonds D) carbon bonds E) ionic bonds 37) What is the name of this compound? O CH 3 C CHCH3 CH3 A) 2-pentanone B) methyl propyl ketone C) 3-methyl-2-butanone D) 2-methyl-3-butanone E) 2-methyl-3-ketone butane 38) The carbonyl group consists of A) a carbon-oxygen-hydrogen structure. B) a carbon-oxygen single bond. C) a carbon-oxygen double bond. D) a carbon-oxygen triple bond. 1) Which one of the following is characteristic of a base? A) produces H30¹ in water B) has a sour taste C) has a slippery, soapy feel D) turns blue litmus red E) is insoluble in water 2) Identify the Bronsted-Lowry acid in the following reaction.. H₂O + CO3²- -> HCO³ + OH A) H₂O B) CO²- C) HCO³- D) OH E) H₂CO₂ 3) Which of the following is correctly identified? A) NH3, strong acid B) NaOH, strong base C) HCI, weak acid D) H₂CO3, strong acid E) Ca(OH)2, weak base 4) What is the [OH-] in a solution that has a [H3O+] 2.0 x 10-4 M? A) 2.0 10-10 M B) 5.0 x C) 1.0 x 10-10 M 10-10 M D) 2.0 × 10-4 M E) 5.0 × 10-11 M I 5) Which of the following statements correctly describes the hydronium- hydroxide balance in the given solution? A) In acids, [OH-] is greater than [H3O+]. B) In bases, [OH-] = [H3O+]. C) In neutral solutions, [H3O+] = [O]. D) In bases, [OH-] is greater than [H3O+]. E) In bases, [OH-] is less than [H3O+]. 6) In which of the following are the pH values arranged from the most acidic to the most basic? A) 1, 3, 6, 8, 11, 14 B) 2, 5, 7, 9, 10, 1.1 C) 14, 10, 7, 4, 3, 1 D) 14, 10, 7, 1, 3, 5 E) 7, 10, 14, 4, 3, 11 7) What is the pH of a solution with [H3O+] = 3.0 × 10-3 M? A) 3.0 × 10-3 C) 3.0 E) 9.0 B) 2.52 D) -2.52 8) Which of the following is a neutralization reaction? A) KCI + NaNO3 1 KNO3 + NaCl B) HNO3 + KOH → H₂O + KNO3 C) H₂O + SO3 → H₂SO4 D) 4Na + O₂ 1 2Na₂O E) 2NO2 1 2NO + 0₂ 9) A 25.0 mL sample of H₂SO4 requires 20.0 mL of 2.00 M KOH for complete neutralization. What is the molarity of the acid? H₂SO4 + 2KOH → A) 2.00 M B) 2.50 M C) 0.800 M D) 1.60 M E) 1.25 M 10) Generally, a solution of an organic compound in water will be electrically A) highly conductive. B) highly ionized. C) nonconductive. K₂SO4 + 2H₂O D) insulated. E) charged. 11) Which of the following is NOT typical of most organic compounds? A) high melting point B) poor solubility in water C) low boiling point D) covalent bonding E) high flammability 12) What is the IUPAC name of this allena I 14) Which of the following pairs of compounds are structural isomers? A) CI CH3 CHCH2 CH3 and CH3CH2CHCH3 B) CICH2CH2CH2CH3 and CH3CH2CH2CH₂Cl C) D) Cl E) CH3CHCH3 and CH3CHCH2C1 CH₂C1 CH3 CH3CHCH3 and CH 3 C CH3 CH₂ CH3 and 12) What is the IUPAC name of this alkane? CH3 CH3-C-CH₂-CH-CH3 I CH3 CH₂-CH3 A) 4-ethyl-2,2-dimethylpentane B) 2-ethyl-4,4-dimethylpentane C) 2,2,4-trimethylhexane D) 2-ethyl-2,2-dimethylpentane E) 3,5,5-trimethylhexane 13) What type of compound is CH3CH₂CH₂CI? A) alkane C) cycloalkane E) isomer B) branched alkane D) haloalkane 15) One reason for the very large number of organic compounds (compared to the total number of inorganic compounds) is A) functional groups. B) structural isomerism. C) hydrogen bonding. D) the occurrence of organic compounds in living things. E) the electronegativity of carbon. 16) The functional group contained in the compound CH3CH₂COOCH, is a(n) A) thiol. B) carboxylic acid. C) amine, D) ester, E) amide. 2 17) What is the IUPAC name for the following compound? CH3 CI CH3-CH-C - CHÍCH 1 CH3 CH3 A) 4-chloro-4,5-dimethyl-2-hexene B) 3-chloro-1,3,4-trimethyl-1-pentene C) 3-chloro-2,3-dimethyl-4-hexene D) 3-chloro-2,3,5-trimethyl-4-pentene E) 3-chloro-1,3,4,4-tetramethyl-1-butene 18) The reaction for the combustion of heptane is C7H16+ 1102 → 7CO₂ + 8H₂O. How many liters of CO2 at STP are produced from the complete combustion of 2.00 moles of heptane? A) 44.8 L C) 157 L E) 314 L B) 22.4 L D) 246 L 19) The reaction of hydrogen (H₂) and propene using a platinum catalyst is called B) substitution. A) combustion. C) addition. D) neutralization. E) condensation. 20) The reaction of cyclohexene and Cl₂ yields A) 1-chlorocyclohexene. B) 1,2-dichlorocyclohexane. C) 1,3-dichlorocyclohexane. D) 2,3-dichlorocyclohexane. E) 3,4-dichlorocyclohexane. 22) Name the following compound. CH₂CH₂CH₂CH, CH₂CH₂CC CH CH₂CH₂CH, A) 3-butyl-3-propyl-1-pentyne B) 3-butyl-3-propyl-4-pentyne C) 3-ethyl-3-propyl-1-heptyne D) 5-ethyl-5-propyl-6-heptyne E) 3-ethyl-3-butyl-1-hexyne 23) Which set of reactants would give this compound? CI CH3-CH2-CH-CH₂Cl A) CH3CH₂CH₂CH3 + 2HCl B) CH3CH CHCH3 + 2HCl C) CH3CH2CH CH2 + 2HCl D) CH3CH CHCH3 + Cl2 E) CH3CH2CH CH2 + Cl2 I 24) What are the prefixes used to designate substituent positions on disubstituted benzene compounds? A) ortho-, meta-, and para-. B) cis- and trans- C) syn- and anti- D) All of the above. E) None of the above. 25) Which of the following is the IUPAC name for the compound below? D) 2,3-dichlorocyclohexane. E) 3,4-dichlorocyclohexane. 21) The structural formula of benzene is often represented as a A) ring of five carbon atoms. B) ring of six carbon atoms with six double bonds. C) ring of six carbon atoms with a circle in the center. D) cycloalkane. E) cycloalkyne. 26) The condensed structural formula for 2,3-dichloro-4-methylcyclohexanol is A) он CH3 a OH B) OH D) Cl 53 CH₂ Page 4 / 7 25) Which of the following is the IUPAC name for the compound below? ОН CH₂ CH₂ CH₂ A) 1,1,4-trimethyl-6-cyclohexanol B) 1,1,4-trimethyl-2-cyclohexanol C) 2,2,5-trimethylcyclohexanol D) 2,2,5-trimethylphenol E) 2-ethyl-5-methylcyclohexanol 29) What is the name for this compound? OH 5 Br CH₂CH₂ A) o-bromo-m-ethylphenol B) 2-bromo-5-ethylphenol C) 4-bromo-1-ethyl-5-phenol D) 6-bromo-3-ethylphenol E) 2-bromo-5-methylphenol 20TQ+ong-smelling compounds Al fruity odors m 26) The condensed structural formula for 2,3-dichloro-4-methylcyclohexanol is A) C) 3 OH CH3 E) OH B) CI CI OH CI CH3 B) OH CH3 -CH-CH3 OH D) OH CH3 CH3 fa CH3 27) Which one of the following compounds is a thiol? A) a O 11 CH3 - CH2 - CH C) CH3 -CH=CH-CH2 Page 4/7 D) CH3 - CH2 - 0-CH2-L I 29) What is the name for this compound? OH CH3CH₂ A) o-bromo-m-ethylphenol B) 2-bromo-5-ethylphenol C) 4-bromo-1-ethyl-5-phenol D) 6-bromo-3-ethylphenol E) 2-bromo-5-methylphenol Br 30) Thiols are strong-smelling compounds responsible for A) fruity odors. B) sharp odors. C) flowery odors. D) skunky or bad smelling odors. E) salty odors. 31) What is the name of this compound? O-CH,CH,CH A) cyclopentyl propyl ether B) cyclopentyl propyl ketone C) 1-cyclopropyl-1-propylalcohol D) propylcyclopentanol E) 3-cyclopentylpropanol 32) What is the common name of the ether that is a structural isomer of 1-butanol? A) isopropyl ether Cdimethyl ether Bburether + 350 B) diethyl ether D) ethyl methyl ether UITZ E) CH3 - SH 28) 1,2-ethanediol (ethylene diol) has uses that include A) antifreeze. C) production of synthetic fibers. D) solvent for ink. E) All of the above. B) solvent for paint. 34) In the oxidation of an alcohol to a ketone, there is A) a loss of hydrogen. B) a loss of oxygen. C) a loss of carbon. D) a gain of hydrogen. E) a gain of oxygen. 35) What is the product when the following compound is oxidized? OH CH3-CH2-CH-CH2-CH3 A) diethyl ketone B) 2-pentene C) pentanal D) diethyl ether E) pentane I 33) What kind of bonds do alcohols form between individual molecules? A) oxygen bonds B) hydrogen bonds C) single bonds D) carbon bonds E) ionic bonds 37) What is the name of this compound? O CH 3 C CHCH3 CH3 A) 2-pentanone B) methyl propyl ketone C) 3-methyl-2-butanone D) 2-methyl-3-butanone E) 2-methyl-3-ketone butane 38) The carbonyl group consists of A) a carbon-oxygen-hydrogen structure. B) a carbon-oxygen single bond. C) a carbon-oxygen double bond. D) a carbon-oxygen triple bond.
Expert Answer:

Related Book For 

Accounting concepts and applications
ISBN: 978-0538745482
11th Edition
Authors: Albrecht Stice, Stice Swain
Posted Date:
Students also viewed these chemistry questions
-
Which one of the following is correct? a. Management accounting reports are usually available to the public. b. Management accounting is legislated and governed by regulatory agencies. c. Financial...
-
Which one of the following is incorrect? a. Management accounting is not as important as financial accounting for the competitive success of a company. b. Governments do not require a company to...
-
Which one of the following is correct? a. Short-run planning includes capital budgeting and operational budgeting. b. Long-run planning includes production and process prioritizing. c. Long-run...
-
1. For each of the following, compute the present value (Do not round intermediate calculations and round your final answers to 2 decimal places. (e.g., 32.16)): 2. For each of the following, compute...
-
The following information was taken from the annual report of ROM. The account balances are as of December 31, 2011. Cash.. $ 1,220 Accounts receivable.. 3,112 Merchandise inventory.. 966 Prepaid...
-
Steel mills in three cities produce the following amounts of steel: Location Weekly Production (tons) A. ............Bethlehem 180 B. ..........Birmingham 260 C. .............Gary 310 740 These mills...
-
An investment is guaranteed to have a unique value of IRR if which of the following is true? a. Alternating positive and negative cash flows b. An initial negative cash flow followed by all positive...
-
Last year (2013), Richter Condos installed a mechanized elevator for its tenants. The owner of the company, Ron Richter, recently returned from an industry equipment exhibition where he watched a...
-
What is the binary pattern represented by the following Differential Manchester encoding:
-
Ashton and Melody Webb are a married couple in their mid-20s. Ashton has a good start as an electrical engineer and Melody works as a sales representative. Since their marriage four years ago, Ashton...
-
Suppose that loan demand is given by Loans Demanded=d0+d1Y-d2 r Loans Demanded=2025+0.05Y-0.01r Also, suppose that the loan supply is Loans Supplied=s0+s1 (r-rrf ) Loans Supplied=75+1000(r-rrf ) a)....
-
Most atoms happen to be about 1 Angstrom or 108 cm in radius, and typical nuclei are about 1013 cm in radius (actually, larger nuclei are somewhat larger, but we will ignore this complication)....
-
Capillary electrophoresis ( CE ) is notable for extremely narrow peak widths. What features of CE allow for these narrow peak widths? What are the advantages of narrow peaks?
-
Benfer Corporation's budgeA company's beginning income tax liability plus its budgeted income tax minus its budgeted income tax for the period equals its budgeted ending income tax liability ted...
-
How do different types of gene mutations influence genetic variation, disease susceptibility, and evolutionary processes? Provide examples
-
What are Coca-Cola's competitive advantages?
-
Suppose that the diffusion coefficient of a substance is D = f(c). Show that c(x, t) satisfies the equation at ac = D + g x (c) . where g = f'(c).
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
Why is it necessary to physically count inventory when the perpetual inventory method is being used?
-
On April 1, 2012, Cajun Company paid $210,000 in cash to purchase land, a building, and equipment. The appraised fair market values of the assets were as follows: land, $70,000; building, $120,000;...
-
Using the information from PE 12-50, make the necessary journal entry to record the purchase of this held-to-maturity security.
-
On January 1, 2015, Meredith Corporation purchased 25% of the ordinary shares of Pirates Company for 200,000. During the year, Pirates earned net income of 80,000 and paid dividends of 20,000....
-
Cairo Corporation has government bonds classified as held-for-collection at December 31, 2015. These bonds have a par value of \($800\),000, an amortized cost of \($800\),000, and a fair value of...
-
Komissarov Company has a debt investment in the bonds issued by Keune Inc. The bonds were purchased at par for 400,000 and, at the end of 2015, have a remaining life of 3 years with annual interest...

Study smarter with the SolutionInn App


