1. Write a balanced equation for the reaction of the active ingredient in Tums with excess...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
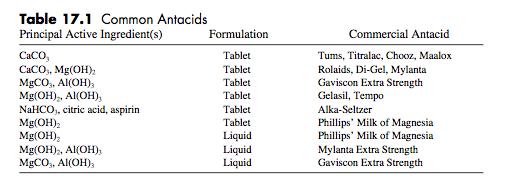
1. Write a balanced equation for the reaction of the active ingredient in Tums with excess acid. See Table 17.1. 2. Identify the two most common anions present in antacids. 3. a. How much time should be allowed for the titrant io drain from the buret wall before a reading is made? 16 b. Experimental Procedure, Part B.2. Bromophenol blue is the indicator used in delecting the endpoint for the antacid analysis in this experiment. What is the expected color change at the endpoint? 4. a. How many moles of stomach acid would be neutralized by one tablet of Tums Ultra 1000 that contains 1000 mg of calcium carbonate? CacO, (ag) +2 H,0(ag) - Ca"(ag) + CO(g) + 3 H,O) b. Assuming the volume of the stomach to be 1.0 L what will be the pH change of the stomach acid resulting from the ingestion of one Tums ultra 1000 tablet that contains 1000 mg of cal- cium carbonate. TUMS Table 17.1 Common Antacids Principal Active Ingredient(s) Formulation Commercial Antacid CaCO, CaCO, Mg(OH); MgCO,, Al(OH), Mg(OH),, Al(OH), NaHCO, citric acid, aspirin Mg(OH): Mg(OH), Mg(OH)2, Al(OH), MGCO, Al(OH), Tablet Tums, Titralac, Chooz, Maalox Rolaids, Di-Gel, Mylanta Gaviscon Extra Strength Gelasil, Tempo Tablet Tablet Tablet Alka-Seltzer Tablet Tablet Phillips' Milk of Magnesia Phillips' Milk of Magnesia Mylanta Extra Strength Gaviscon Extra Strength Liquid Liquid Liquid 1. Write a balanced equation for the reaction of the active ingredient in Tums with excess acid. See Table 17.1. 2. Identify the two most common anions present in antacids. 3. a. How much time should be allowed for the titrant io drain from the buret wall before a reading is made? 16 b. Experimental Procedure, Part B.2. Bromophenol blue is the indicator used in delecting the endpoint for the antacid analysis in this experiment. What is the expected color change at the endpoint? 4. a. How many moles of stomach acid would be neutralized by one tablet of Tums Ultra 1000 that contains 1000 mg of calcium carbonate? CacO, (ag) +2 H,0(ag) - Ca"(ag) + CO(g) + 3 H,O) b. Assuming the volume of the stomach to be 1.0 L what will be the pH change of the stomach acid resulting from the ingestion of one Tums ultra 1000 tablet that contains 1000 mg of cal- cium carbonate. TUMS Table 17.1 Common Antacids Principal Active Ingredient(s) Formulation Commercial Antacid CaCO, CaCO, Mg(OH); MgCO,, Al(OH), Mg(OH),, Al(OH), NaHCO, citric acid, aspirin Mg(OH): Mg(OH), Mg(OH)2, Al(OH), MGCO, Al(OH), Tablet Tums, Titralac, Chooz, Maalox Rolaids, Di-Gel, Mylanta Gaviscon Extra Strength Gelasil, Tempo Tablet Tablet Tablet Alka-Seltzer Tablet Tablet Phillips' Milk of Magnesia Phillips' Milk of Magnesia Mylanta Extra Strength Gaviscon Extra Strength Liquid Liquid Liquid
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Write a balanced equation for the reaction of each of the following compounds with water: (a) SO2(g) (b) Cl2O7(g) (c) Na2O2(s) (d) BaC2(s) (e) RbO2(s) (f) Mg3N2(s) (g) NaH(s).
-
Write a balanced equation for the reaction of -(1)-galactose (use either an acyclic or a cyclic structure, whichever seems more appropriate) with each of the following: a. Hydroxylamine (to form an...
-
Write down a balanced equation for the reaction of bromine with cyclohexene. Is the product of the reaction saturated or unsaturated? Explain your answer by describing what type of reaction is...
-
Consider the graph of a function g(x). g(x) 5 f 3 2 1 2 3 4 5 6 Find the point c at which the function has a jump discontinuity but is right-continuous. C = What value should be assigned to g(c) to...
-
The bear increases speed and the hiker steadily slows down until the bear catches the hiker. A bear sets off in pursuit of a hiker. Graph the position of a bear and hiker as functions of time from...
-
Business schools have commonly observed that when job opportunities are down, those interested in business seek to enter MBA programs. To test this hypothesis, we decided to look at the number of...
-
Can a duty to care be established by statute or contract? Discuss your answer.
-
Suppose that during 2013, the market for Stanmore?s special-purpose machines grew by 3%. In Exercise 12- 22, Stanmore Corporation makes a special- purpose machine, D4H, used in the textile industry....
-
The following information on maintenance and repair costs and revenues for the last two years is available from the accounting records at Arnie's Arcade & Video Palace. Arnie has asked you to help...
-
On January 1, 20X1, partners Art, Bru, and Chou, who share profits and losses in the ratio of 5:3:2, respectively, decide to liquidate their partnership. The partnership trial balance at this date...
-
Roger spends $60 per month on beer and $60 per month on cigarettes. He is going to quit smoking and cut his beer expense in half by making his own beer at the local U-Brew. At the end of every month...
-
The Greenpeace Report issued in 2020, UNPACKED: How supermarkets can cut plastic packaging in half by 2025, suggested that: GOVERNMENT SHOULD: Set legally binding targets in the Environment Bill to...
-
The result of including goodwill by valuing the non-controlling shares at their market price using Method 2 is to value the non-controlling shares on a different basis to valuing an equity investment...
-
Call options on a stock are available with strike prices of \(\$ 15, \$ 17 \frac{1}{2}\), and \(\$ 20\), and expiration dates in 3 months. Their prices are \(\$ 4, \$ 2\), and \(\$ \frac{1}{2}\),...
-
An investor enters into a short forward contract to sell 100,000 British pounds for US dollars at an exchange rate of 1.4000 US dollars per pound. How much does the investor gain or lose if the...
-
A US Treasury bond pays a \(7 \%\) coupon on January 7 and July 7 . How much interest accrues per \(\$ 100\) of principal to the bondholder between July 7, 2011, and August 9, 2011? How would your...
-
from Tonina and cos cash flow statements Total cash outflows= $352,500 Total cash inflows= $532,500 Opening balance= $135,000 Calculate the closing balance (2 marks) Define the term cash flow...
-
The water in tank A is at 270 F with quality of 10% and mass 1 lbm. It is connected to a piston/cylinder holding constant pressure of 40 psia initially with 1 lbm water at 700 F. The valve is opened,...
-
(a) How many coulombs are required to plate a layer of chromium metal 0.25 mm thick on an auto bumper with a total area of 0.32 m2 from a solution containing CrO42-? The density of chromium metal is...
-
Organic compounds containing C-O and C-1 bonds are more reactive than simple alkane hydrocarbons. Considering the comparative values of C - H, C - C, C--O and C -- 1 bond enthalpies (Table 8.4), why...
-
Identify each of the following elements as a metal, nonmetal, or metalloid: (a) Gallium, (b) Molybdenum, (c) Tellurium, (d) Arsenic, (e) Xenon, (f) Ruthenium.
-
As the new controller, reply to the following comment made by your plant manager: When I employ a proper accounting software, which can process all my daily accounting records and provide me with all...
-
How is financial accounting different from management accounting?
-
Hannah Gilpin is the controller of Blakemore Auto Glass, a division of Eastern Glass and Window. Her division has been under pressure to improve its divisional operating income. Currently, divisions...

Study smarter with the SolutionInn App


