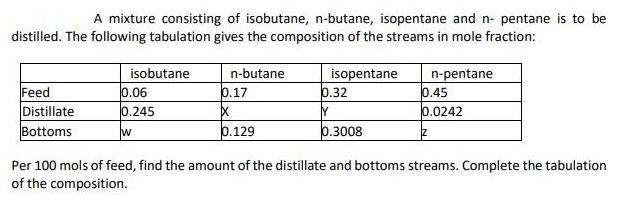
A mixture consisting of isobutane, n-butane, isopentane and n- pentane is to be distilled. The following...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A mixture consisting of isobutane, n-butane, isopentane and n- pentane is to be distilled. The following tabulation gives the composition of the streams in mole fraction: isobutane n-butane isopentane b.32 n-pentane 0.45 Feed Distillate Bottoms 0.06 0.245 0.17 Y 0.0242 w 0.129 0.3008 Per 100 mols of feed, find the amount of the distillate and bottoms streams. Complete the tabulation of the composition. A mixture consisting of isobutane, n-butane, isopentane and n- pentane is to be distilled. The following tabulation gives the composition of the streams in mole fraction: isobutane n-butane isopentane b.32 n-pentane 0.45 Feed Distillate Bottoms 0.06 0.245 0.17 Y 0.0242 w 0.129 0.3008 Per 100 mols of feed, find the amount of the distillate and bottoms streams. Complete the tabulation of the composition.
Expert Answer:
Answer rating: 100% (QA)
Let B amount of bottom product Let D amount of distillate Let X mole fractio... View the full answer

Posted Date:
Students also viewed these chemical engineering questions
-
Find the adiabatic exponent for a mixture consisting of v1 moles of a monatomic gas and v2 moles of gas of rigid diatomic molecules.
-
A mixture consisting of 1-mol-% ethane, 5-mol-% propane. 44-mol-% n-butane, and 50-mol-% isobutanc is brought to a condition of 70(F) at pressure P. If the molar fraction of the system that is vapor...
-
A mixture consisting of 0.18 kmol of methane and 0.274 kmol of butane occupies a volume of 0.3 m3 at a temperature of 240oC. Using the IG model, determine the pressure exerted by the mixture.
-
In Problems 1158, perform the indicated operation, and write each expression in the standard form a + bi. 2 + 3i 1-i
-
The technical report Ozone Season Emissions by State (U.S. Environmental Protection Agency, 2002) gave the following nitrous oxide emissions (in thousands of tons) for the 48 states in the...
-
Stine Company uses a job order cost system. On May 1, the company has a balance in Work in Process Inventory of $3,500 and two jobs in process: Job No. 429 $2,000, and Job No. 430 $1,500. During May,...
-
A 20-ounce bottle of Dasani water typically costs about \($1.99\) at a convenience store. At an airport shop, that same bottle of Dasani water could cost \($2.89\) or more. The costs of operating...
-
Organic Life Co. is an HMO for businesses in the Portland area. The following account balances appear on the balance sheet of Organic Life Co: Common stock (250,000 shares authorized), $125 par,...
-
Microsoft Corporation makes Xbox video game consoles. For Microsoft's financial year YYYY, please make use of the following provided information regarding the inventory of those Xbox consoles:...
-
Modify the sentence-generator program of Case Study so that it inputs its vocabulary from a set of text files at startup. The filenames are nouns.txt, verbs. txt, articles.txt, and prepositions.txt....
-
1 . Please explain sustainability reporting 2 . What is the major challenge ( s ) a company faces in preparing the sustainability reporting made public? 3 . What result ( s ) or conclusion ( s ) may...
-
Calculate the percentage of the tax borne by the demander and supplier in each of the following cases: a. ED = 0.3, ES = 1.2 b. ED = 3, ES = 2 c. ED = 0.5, ES = 1 d. ED = 0.5, ES = 0.5 e. Summarize...
-
Economists have estimated the following transportation elasticities. For each pair, explain possible reasons why the elasticities differ. a. Elasticity of demand for buses is 0.23 during peak hours...
-
Imagine that you are the owner of a small business and are searching on the Web for al company that can supply the products or services needed by your business. Find several business-to-business...
-
The Office of the Comptroller of the Currency is responsible for many of the regulations affecting bank operations. Go to www.occ.treas.gov/. Click on "Law and Regulations" in the far-right column...
-
An analysis of costs of Sullivan Manufacturing Company gives the following information. You are required to determine (a) Break-even sales volume (b) Profit at the budgeted sales of 18,50,000. Cost...
-
Question 4 - 10 are based on the figure below: A 33 5.6 22 4.8 too X 4. For vector B, what are the signs of its x and y components? A. Postive, negative B. Negative, positive C. Positive, positive D....
-
Find a least expensive route, in monthly lease charges, between the pairs of computer centers in Exercise 11 using the lease charges given in Figure 2. a) Boston and Los Angeles b) New York and San...
-
True or False: Build-Operate-Transfer (BOT) makes use of a public-private partnership.
-
Do an Internet search on "build operate transfer." Identify a BOT project, preferably from your home continent (e.g., AF, AS, EU, Pacific, NA, SA), and copy a small portion of what you find (a page...
-
Using an Internet-based search on 'build operate transfer," find an additional definition from a source other than used in Section 14.2. Copy and paste it, as well as any graphics, examples,...

Study smarter with the SolutionInn App

