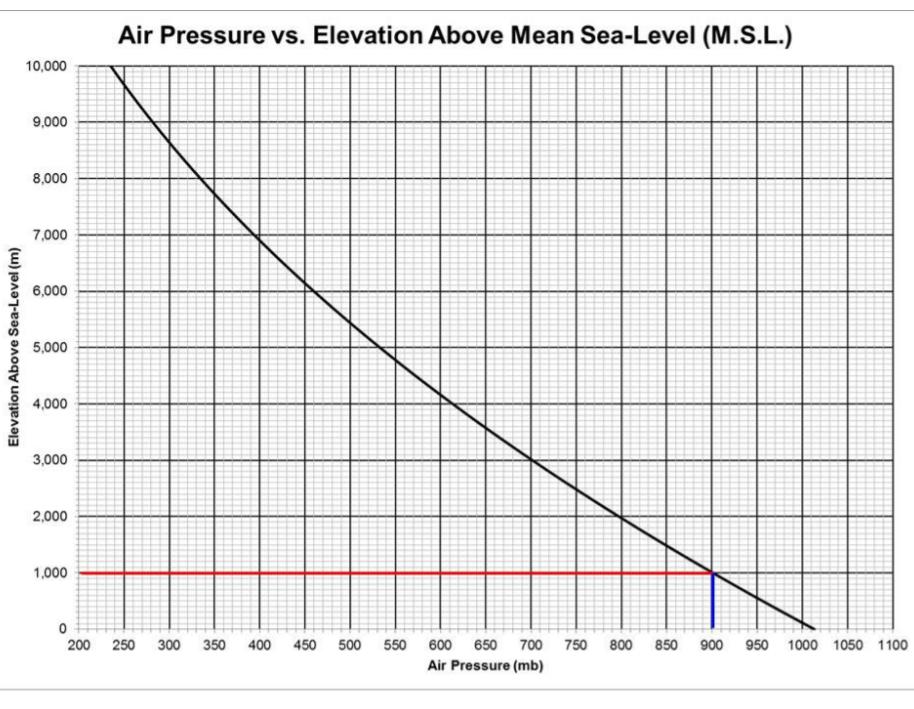
11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July. 11. The elevation of Mount Whitney (California) is 14,505 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea-Level (M.S.L.), determine the air pressure at the top of Mt. Whitney. DO NOT USE ANY UNITS; JUST SUBMIT THE NUMBER OF MILLIBARS. 12. If the temperature at 52,800 feet (the stratosphere) was -40°F, and instead of the Average Lapse Rate being 3.5°F per 1,000 feet of elevation change, what would be the temperature of the air if it were somehow brought down to Fresno, in °F, and the ALR was 5..5°F per 1,000 feet of elevation change? Use Equation (1), but with 5.5 instead of 3.5. 13. The elevation of a campsite at Shaver Lake is 5,577 feet above sea-level. Using the attached figure of Air Pressure vs. Elevation above Mean Sea- Level (M.S.L.), determine the air pressure at the campsite. DO NOT USE ANY UNITS: JUST SUBMIT THE NUMBER OF MILLIBARS. 15. If the temperature in Fresno is 103°F what would be the temperature of a campsite at Shaver Lake that is at 5,577 feet above sea-level? Elevation Above Sea-Level (m) 10,000 9,000 8,000 7,000 6,000 5,000 4,000 3,000 2,000 1,000 0 200 Air Pressure vs. Elevation Above Mean Sea-Level (M.S.L.) 250 300 350 400 450 500 550 600 650 700 750 800 850 900 950 1000 1050 1100 Air Pressure (mb) Method 1: Convert the air pressure at 1,000 m to PSI by dividing 900 mb/1013.25 mb and multiplying the result by 14.7 PSI to get 13.05699482, or 13.1 PSI. To convert from feet to meters, multiply feet by 0.3048 To convert from meters to feet, divide meters by 0.3048 To convert from °F to °C, °C = (°F - 32)/1.8 To convert from °C to °F, °F = °C x 1.8 +32 T Elev2 = T Elev1 + ((Elev1 - Elev2) / 1,000) * Average Lapse RateEQ (1) (where: ELEV1 is the elevation where you are located, T Elev1 is the temperature where you are located, Elev2 is the elevation of the other location, and T Elev2 is the temperature at the other location). (Note: It is understood that all elevations are relative to sea-level, and that elevations above sea-level are positive, and that elevations below sea-level are negative) For the following questions, students may assume that Fresno has an elevation of 328 feet above sea-level, and that the time period is any day in July.
Expert Answer:
Answer rating: 100% (QA)
11 We find that elevation is given in feet while the height in th... View the full answer

Related Book For 

A Survey of Mathematics with Applications
ISBN: 978-0134112107
10th edition
Authors: Allen R. Angel, Christine D. Abbott, Dennis Runde
Posted Date:
Students also viewed these law questions
-
The elevation E, in meters, above sea level at which the boiling point of water is t degrees Celsius is given by the function E(t) = 1000(100 - t) + 580(100 - t) 2. At what elevation is the boiling...
-
The summit of Mount Everest is 8850 m above sea level. (a) How much energy would a 90 kg climber expend against the gravitational force on him in climbing to the summit from sea level? (b) How many...
-
The elevation of a mountain above sea level at (x, y) is 3000e-(x2+2y2)/100 meters. The positive x-axis points east and the positive y-axis points north. A climber is directly above (10, 10). If the...
-
Prepare a statement of cash flows in proper form using the inflows and outflows from questions 4-15. Assume net income (earnings after taxes) from the 2018 income statement was $10,628, and $5,000 in...
-
What is the difference between doing design with CRC cards and designing with sequence diagrams?
-
The Rockies Division operates as a profit center. It reports the following for the year ending December 31, 2020. Prepare a responsibility report for the Rockies Division at December 31, 2020. Actual...
-
Which of the following is necessary for fraud to occur? a. Perceived pressure b. Perceived opportunity C. Rationalization d. All three are necessary.
-
At the end of 2016, Sentry Company reported a deferred tax liability of $ 6,120 based on an income tax rate of 30%. On January 2, 2017, Congress changed the income tax rate to 35%. Required: 1....
-
What elements made the ALDUB and Think Different Campaign of Apple viral using the lens of the Rules of Epidemics * The Law of the Few * The Stickiness Factor *The Power of Context
-
Peru Furniture City, Inc., sold $88,000 of furniture in May to customers who used their American Express Credit Cards. Such sales are subject to a 5% discount by American Express (a nonbank credit...
-
During the COVID-19 pandemic, policymakers were concerned about the availability and delivery of personal protective equipment (PPE), e.g. face- masks, to Norway. A policymaker suggests that we...
-
A 0.47-kg block of wood hangs from the ceiling by a string, and a .07-kg wad of putty is thrown straight upward, striking the bottom of the block with a speed of 5.6 m/s. The wad of putty sticks to...
-
Present Value (PV) $240.38 $238.10 $235.85 $231.14 $222.22 $213.70 Cash Flow (CF) $250 $250 $250 $250 J $250 $250 Interest Rate (i) 4% 5% 6% 4% 4% 4% Term (n) 14 14 14 24 34 4 777 1. Given the graph,...
-
A customer goes short 8 silver contracts. His trade is executed at 4 1 6 . 5 cents per ounce ( contract size = 5 , 0 0 0 troy ounces, minimum tick is . 5 cents / oz . ) . Initial margin is $ 1 , 2 0...
-
In a three - level marketing channel, how many entities ( organization / individual ) are connected?
-
The dividend policy of companies varies to meet the circumstances of each firm and it's shareholders. Explain at least three (3) factors that a firm should consider in determining its dividend policy...
-
Research Algorithm Analysis with Big-O Summarize what you learn by yourself and analyze one algorithm or simple program (your code) with Big-O. Find at least two or more cases of each Big-O...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Change the percent to a decimal number. 0.00045%
-
Use lattice multiplication to determine the product. 4 x 327
-
Evaluate the expression. 8 P 4
-
List the reports available for reporting cash flows.
-
What additional steps are necessary to create a statement of cash flows for a period other than the one ending on the computers current system date?
-
Identify three different periods of time that QuickBooks Accountant provides for a statement of cash flows.

Study smarter with the SolutionInn App


