13. The decreasing order of acidity of following phenol derivatives is HC-OH (Q) CHOH (P)...
Fantastic news! We've Found the answer you've been seeking!
Question:
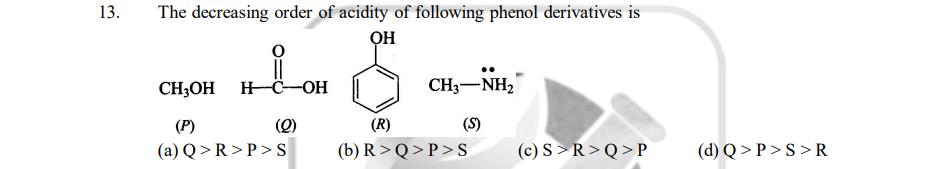
Transcribed Image Text:
13. The decreasing order of acidity of following phenol derivatives is ОН HC-OH (Q) CH₂OH (P) (a) Q>R>P>S CH–NH, (S) (R) (b) R>Q>P>S (c) S>R>Q> P (d) Q>P>S>R 13. The decreasing order of acidity of following phenol derivatives is ОН HC-OH (Q) CH₂OH (P) (a) Q>R>P>S CH–NH, (S) (R) (b) R>Q>P>S (c) S>R>Q> P (d) Q>P>S>R
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
A ball is released from rest at the left of the metal track shown here. Assume it has only enough friction to roll, but not to lessen its speed. Rank these quantities from greatest to least at each...
-
In this assignment you will use the Income Statement and Balance Sheet information of Triple B Company to meet the following requirements: Part 1: Use the worksheet titled Horizontal Analysis to...
-
14. The decreasing order of acidity of following phenol derivatives is OH &&&& CH OH CH3 CH3 (R) (P) (a) S>Q>P>R (b) S>Q>R>P (Q) OH (S) (c) P>Q>R>S (d) R>S>Q> P
-
Find the magnitude and direction of the electric field strength at the point P due to the point charges at A and B as shown in the Figure 1. (k = 9 x 10 Nm/C) +2 C 10 cm A 10 cm Figure 1 B -8 C (5...
-
Orange County Engine Parts Inc. (OCEP) produces three productspistons, valves, and camsfor the heavy equipment industry. OCEP has a very simple production process and product line and uses a single...
-
Refer to the data for Minden Company in Problem 817. The company is considering making the following changes to the assumptions underlying its master budget: In Problem 817 Minden Company is a...
-
A significant issue when a contract is breached is whether the cause of action is strictly in breach of contract or may be expanded to negligence and strict liability. The significance comes into...
-
The City of Lynn operates its municipal airport. The trial balance of the Airport Fund as of January 1, 20X0, was as follows: The following transactions took place during the year: 1. Revenues...
-
You are the COO managing the operations of a robo-advisor and you need to ensure that all the stock holdings in the portfolios under management are kept up to date. Given the following inputs in...
-
Figure P3.14 shows three physical situations; show the possible process in a Pv diagram. Po m, "p R-410a (a) (b) (c) FIGURE P3.14
-
My Son in law is a huge Cubs fan. He took his three boys to see a game. He had to save up all year long because he knew prices would be crazy. A hot dog was something like $6.50 so he spent around...
-
Differentiate Si yasinxcosx
-
What considerations should be taken into account when developing a comprehensive patch testing methodology, balancing the need for thorough validation with the imperative to minimize service...
-
A cleaning business employs and pays individuals as contractors. How should the business report payments that have been made to these contractors to the ATO?When is the report due and what methods of...
-
in a 'push' for promotional strategy, information flow occurs in the following order: a . . producer, wholesaler, retailer, consumer b . . producer, retailer, agent, consumer c . . consumer,...
-
Write a brief description of loan management and credit analysis and discuss the background information associated with the problems that need to be solved. Identify project goals and the underlying...
-
Lithium, Inc. is considering two mutually exclusive projects, A and B. Project A costs $95,000 and is expected to generate $65,000 in year one and $75,000 in year two. Project B costs $120,000 and is...
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
Write formulas for all of the isomers of each of the following. Designate pairs of enantiomers and achiral compounds where they exist. (a) 1-Bromo-2-chlorocyclohexane (b) 1-Bromo-3-chlorocyclohexane...
-
In addition to fructose (Practice Problem 22.12) and sorbose (Problem 22.24), there are two other 2-ketohexoses, psicose and tagatose. D-Psicose yields the same phenylosazone as D-allose (or...
-
Compound B (C8H12O2) shows a strong carbonyl absorption in its IR spectrum. The broadband proton-decoupled 13C NMR spectrum of B has only three signals, at 19 (CH3), 71 (C), and 216 (C). Propose a...
-
A second-order dynamic system is modeled as \[9 \ddot{x}+6 \dot{x}+\frac{10}{9} x=14 \delta(t), \quad x(0)=0, \quad \dot{x}(0)=-\frac{1}{4}\] a. Find the response \(x(t)\) in closed form. b. Plot the...
-
Consider a first-order system with time constant \(\tau\) and zero initial condition. Find the system's unit-step response for \(\tau=\frac{1}{3}\) and \(\frac{2}{3}\), plot the two curves versus \(0...
-
a. Identify the damping type and find the free response. b. Plot the free response by using the initial command. \(\ddot{x}+3 \dot{x}+4 x=0, \quad x(0)=\frac{2}{5}, \quad \dot{x}(0)=0\)
Computational Chemotaxis Models Neurodegenerative Disease 1st Edition - ISBN: 9813207450 - Free Book

Study smarter with the SolutionInn App



