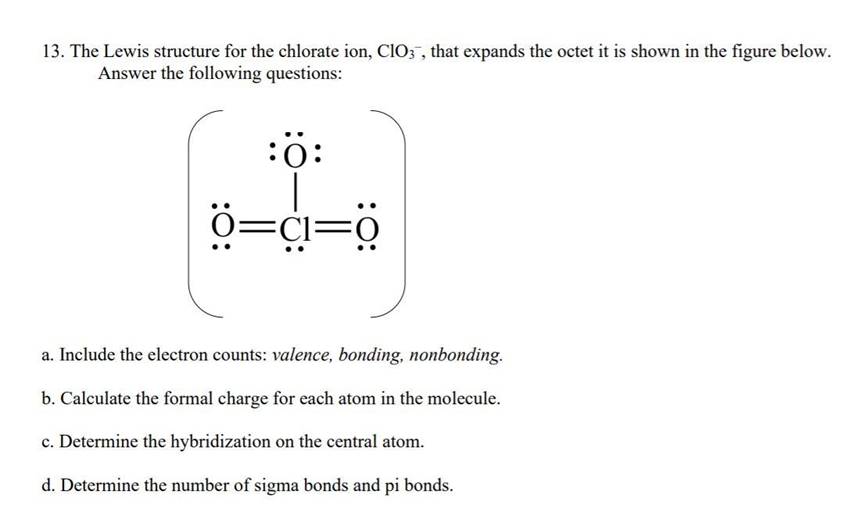
13. The Lewis structure for the chlorate ion, CIO; , that expands the octet it is...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
13. The Lewis structure for the chlorate ion, CIO; , that expands the octet it is shown in the figure below. Answer the following questions: :ö: ö=ci=ö a. Include the electron counts: valence, bonding, nonbonding. b. Calculate the formal charge for each atom in the molecule. c. Determine the hybridization on the central atom. d. Determine the number of sigma bonds and pi bonds. 13. The Lewis structure for the chlorate ion, CIO; , that expands the octet it is shown in the figure below. Answer the following questions: :ö: ö=ci=ö a. Include the electron counts: valence, bonding, nonbonding. b. Calculate the formal charge for each atom in the molecule. c. Determine the hybridization on the central atom. d. Determine the number of sigma bonds and pi bonds.
Expert Answer:
Answer rating: 100% (QA)
Cl172 8 7 O8 2 6 Total valance electron of ClO37631 26 The 3 oxygen ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
The device shown in the figure below is used to estimate the thermal conductivity of a new bio material by measuring the steady-state heat-transfer rate and temperature. The bio material is wrapped...
-
If you have the following information: Sales 400.000. sales returns 40.000, purchases 40.000. purchase expenses 20,000, beginning merchandise 10,000. ending merchandise 25.000, selling expenses...
-
It is shown in Example 25.7 that the potential at a point P a distance a above one end of a uniformly charged rod of length lying along the x axis is Use this result to derive an expression for the y...
-
A house girl added 20g of sodium chloride (NaCl) to 80g of water (atomic masses are Na=23amu, Cl=35.5amu). Calculate a)Percent(w/w) of NaCl b)Mole fraction of NaCl
-
A tire manufacturer recently announced a recall because 2% of its tires are defective. If you just bought a new set of four tires from this manufacturer, what is the probability that at least one of...
-
Fresh Look Paint Store uses a perpetual inventory system. The company had the following transactions in March. Mar. 16 Purchased $15,000 of merchandise from Central Paint Distributors, terms 2/10,...
-
Identify three objects for an ATM system.
-
Firms often enter into transactions that are peripheral to their core operations but generate gains and losses that must be reported on the income statement. A gain labeled peripheral by one firm may...
-
2. If all n elements in the input array are the same, what is the big O notation of Insertion Sort? Why? (20 points) INSERTION-SORT(A) 1 for 2 to A.length key=A[j] //Insert A[] into the sorted 2 3...
-
After a complaint was filed against him, Broker Ike was found guilty of a license law violation. He was fined and had to pay legal fees totaling $8,900. This was paid by the Recovery Fund. What else...
-
Laker Company reported the following January purchases and sales data for its only product. For specific identification, ending inventory consists of 300 units from the January 30 purchase, 5 units...
-
Summarize Cognitive Psychology. Identify 3 ways that Cognitive Psychology influences the practice of psychology today.
-
Compare and contrast cognitive psychology and behaviorism? what are some cognitive psychology and behaviorism potential uses in therapy? with examples. how might you use cognitive psychology (or...
-
What is Cognitive Psychology? What are 3 ways that Cognitive Psychology influences the practice of psychology today?
-
A USD 100 million loan is swapped into a JPY 10 billion loan. The spot exchange rate is JPY/USD 100. The interest rates on dollars and yen are 6% and 2% respectively. The maturity of the contract is...
-
Evaluate how the outcomes changed as the process unfolded in Tennessee v Garner (1985). provide citation.
-
Book Print ferences A & R Problem 1-1 Jack Tasker opened his Auto Repair Shop in November 2023. The balance sheet at November 30, 2023, prepared by an inexperienced part-time bookkeeper, is shown...
-
Find the equation of the plane passing through the points P 5,4,3 ,Q 4,3,1 and R 1,5,4
-
The molecule 2-butene, C4H8, can undergo a geometric change called cis-trans isomerization: As discussed in the "Chemistry and Life" box on the chemistry of vision, such transformations can be...
-
Oxyhemoglobin, with an O2 bound to iron, is a low-spin Fe(II) complex; deoxyhemoglobin, without the O 2 molecule, is a high-spin complex. (a) Assuming that the coordination environment about the...
-
The indicator methyl orange has been added to both of these solutions. Based on the colors, classify each statement as true or false: (a) The pH of solution A is definitely less than 7.00. (b) The pH...
-
For the heritage center described in Problem 32, note that a survey that has determined that annual benefits of \(\$ 3\) each are now received by 12,000 visitors, \(\$ 5\) each by 14,000 visitors,...
-
Consider 1 year of a utility company's financial results. The company pays income tax of \(\$ 9,000,000\), where the tax rate is 40 percent. Debt is \(\$ 80,000,000\) and the interest rate on debt is...
-
A new public school in Knoxville is proposed and will replace one older facility and affect school changes for several middle school students. The following estimates apply: a. Calculate the \(B /...

Study smarter with the SolutionInn App


