13-88 A mixture of carbon dioxide and nitrogen flows through a converging nozzle. The mixture leaves...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
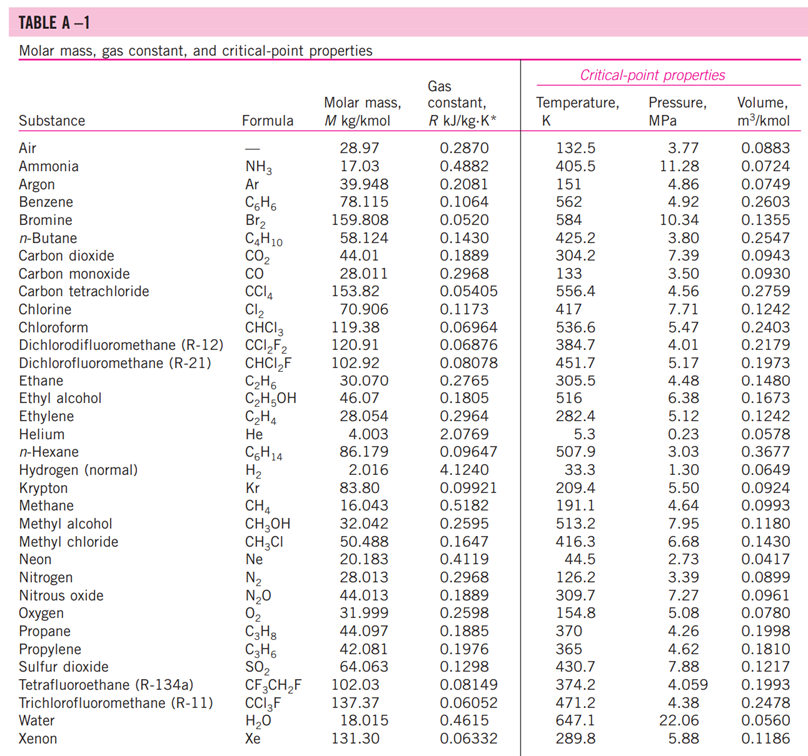
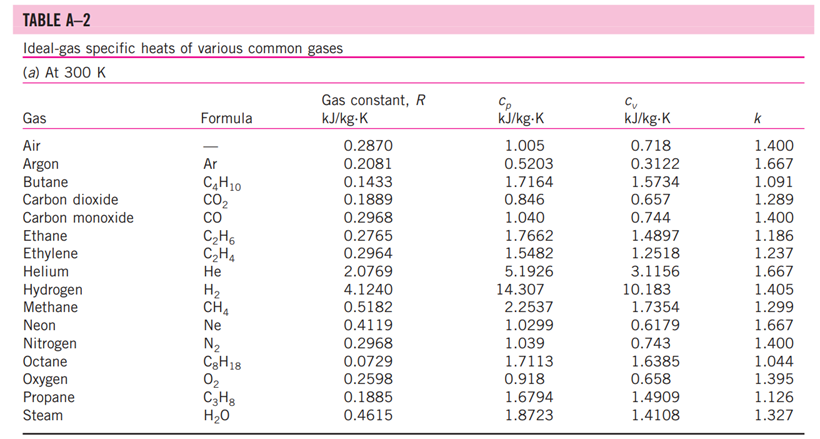
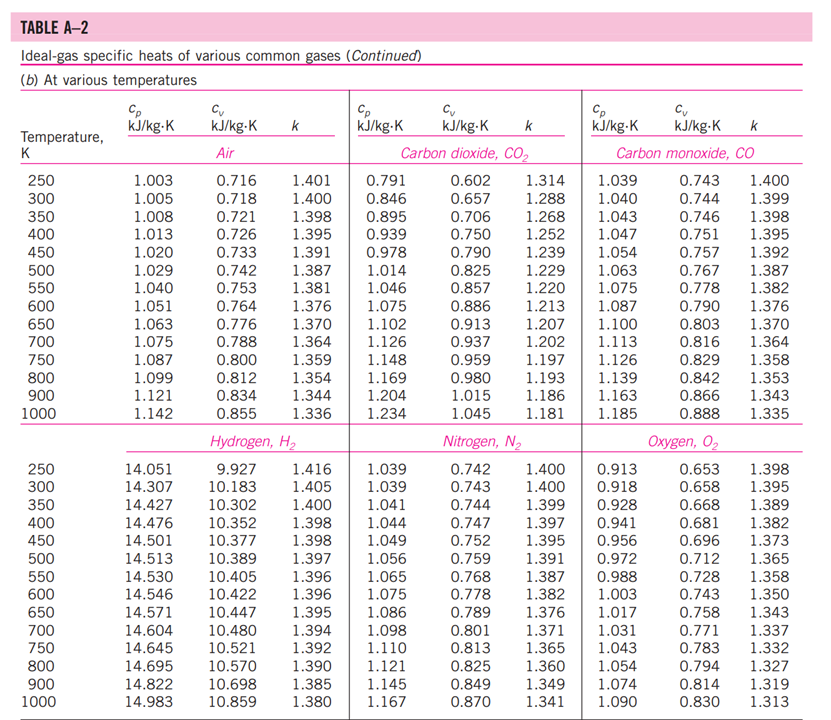
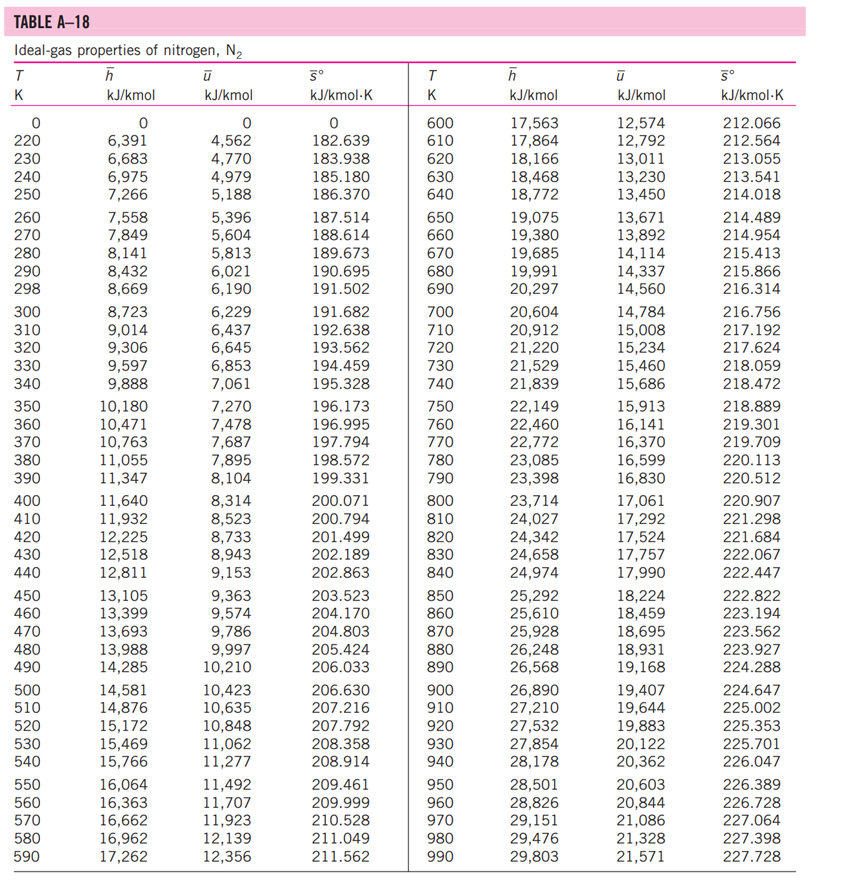
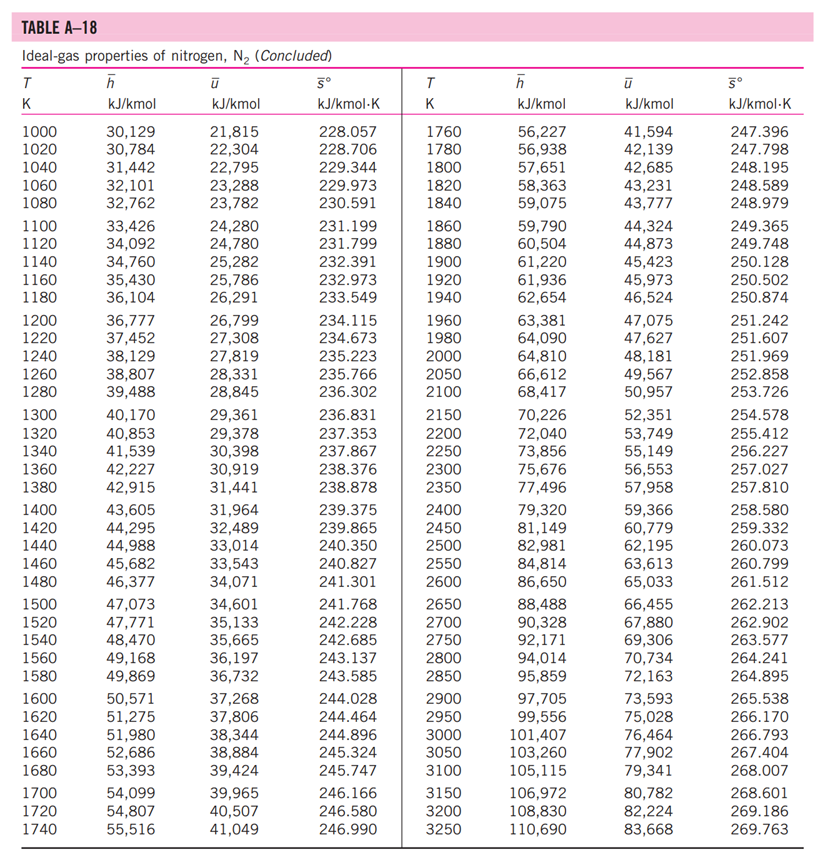
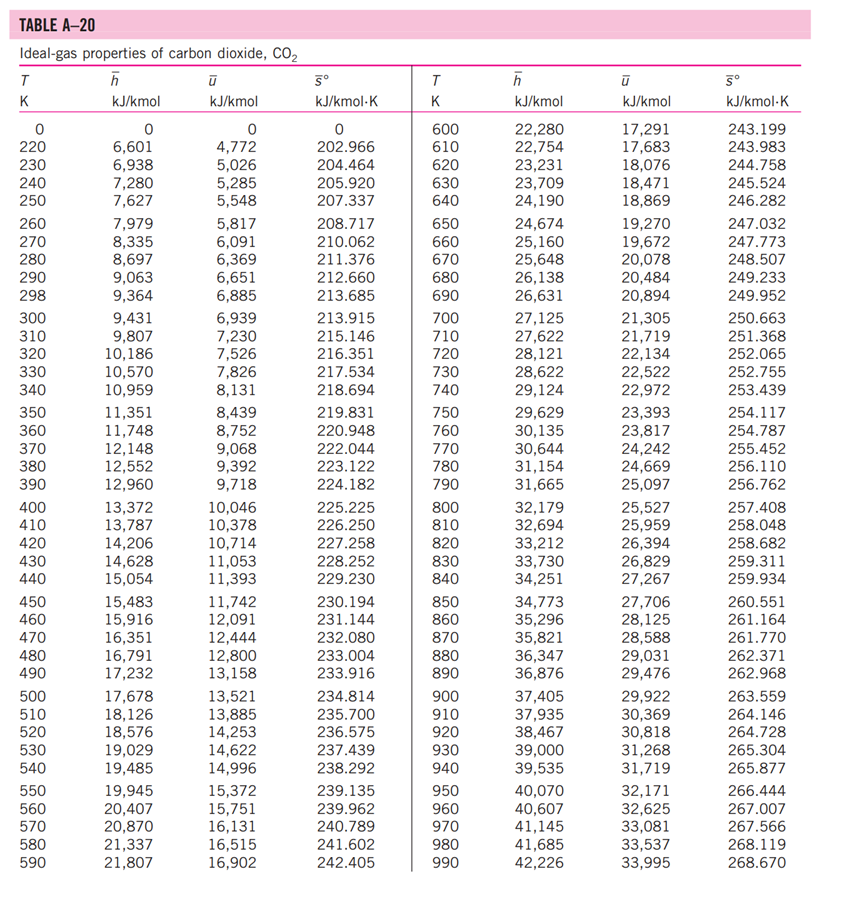
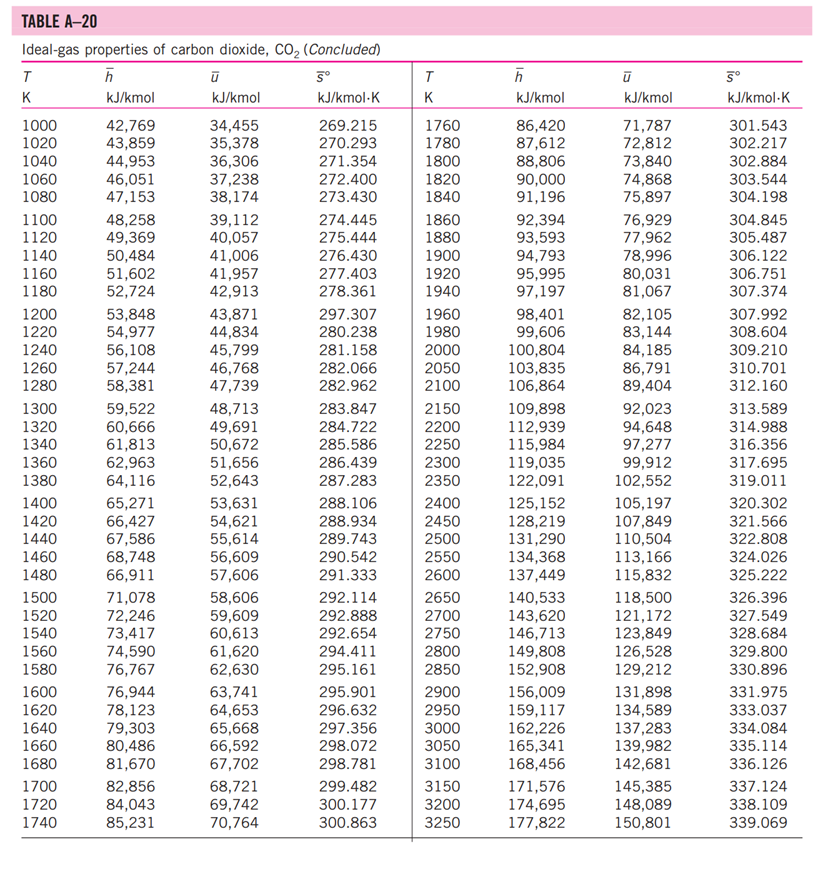
13-88 A mixture of carbon dioxide and nitrogen flows through a converging nozzle. The mixture leaves the nozzle at a temperature of 500 K with a velocity of 360 m/s. If the velocity is equal to the speed of sound at the exit temperature, determine the required makeup of the mixture on a mass basis. TABLE A -1 Molar mass, gas constant, and critical-point properties Substance Air Ammonia Argon Benzene Bromine n-Butane Carbon dioxide Carbon monoxide Carbon tetrachloride Chlorine Chloroform Dichlorodifluoromethane (R-12) Dichlorofluoromethane (R-21) Ethane Ethyl alcohol Ethylene Helium n-Hexane Hydrogen (normal) Krypton Methane Methyl alcohol Methyl chloride Neon Nitrogen Nitrous oxide Oxygen Propane Propylene Sulfur dioxide Tetrafluoroethane (R-134a) Trichlorofluoromethane (R-11) Water Xenon Formula NH3 Ar CH6 Br C4H10 CO CO CCI4 Cl CHCI 3 CCIF CHCIF CH6 CH5OH CH4 He C6H14 H Kr CH4 CHOH CH3CI Ne N NO 0 C3H8 C3H6 SO CF3CHF CCI3F HO Xe Molar mass, M kg/kmol 28.97 17.03 39.948 78.115 159.808 58.124 44.01 28.011 153.82 70.906 119.38 120.91 102.92 30.070 46.07 28.054 4.003 86.179 2.016 83.80 16.043 32.042 50.488 20.183 28.013 44.013 31.999 44.097 42.081 64.063 102.03 137.37 18.015 131.30 Gas constant, R kJ/kg-K* 0.2870 0.4882 0.2081 0.1064 0.0520 0.1430 0.1889 0.2968 0.05405 0.1173 0.06964 0.06876 0.08078 0.2765 0.1805 0.2964 2.0769 0.09647 4.1240 0.09921 0.5182 0.2595 0.1647 0.4119 0.2968 0.1889 0.2598 0.1885 0.1976 0.1298 0.08149 0.06052 0.4615 0.06332 Critical-point properties Temperature, Pressure, MPa K 132.5 405.5 151 562 584 425.2 304.2 133 556.4 417 536.6 384.7 451.7 305.5 516 282.4 5.3 507.9 33.3 209.4 191.1 513.2 416.3 44.5 126.2 309.7 154.8 370 365 430.7 374.2 471.2 647.1 289.8 3.77 11.28 4.86 4.92 10.34 3.80 7.39 3.50 4.56 7.71 5.47 4.01 5.17 4.48 6.38 5.12 0.23 3.03 1.30 5.50 4.64 7.95 6.68 2.73 3.39 7.27 5.08 4.26 4.62 7.88 4.059 4.38 22.06 5.88 Volume, m/kmol 0.0883 0.0724 0.0749 0.2603 0.1355 0.2547 0.0943 0.0930 0.2759 0.1242 0.2403 0.2179 0.1973 0.1480 0.1673 0.1242 0.0578 0.3677 0.0649 0.0924 0.0993 0.1180 0.1430 0.0417 0.0899 0.0961 0.0780 0.1998 0.1810 0.1217 0.1993 0.2478 0.0560 0.1186 TABLE A-2 Ideal-gas specific heats of various common gases (a) At 300 K Gas Air Argon Butane Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon Nitrogen Octane Oxygen Propane Steam Formula | Ar C4H10 CO CO CH6 CH4 He H CH4 Ne N C8H18 0 C3H8 HO Gas constant, R kJ/kg.K 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 0.0729 0.2598 0.1885 0.4615 Cp kJ/kg.K 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 1.7113 0.918 1.6794 1.8723 Cv kJ/kg-K 0.718 0.3122 1.5734 0.657 0.744 1.4897 1.2518 3.1156 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 k 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 1.044 1.395 1.126 1.327 TABLE A-2 Ideal-gas specific heats of various common gases (Continued) (b) At various temperatures Temperature, K 250 300 350 400 450 500 550 600 650 700 750 800 900 1000 250 300 350 400 450 500 550 600 650 700 750 800 900 1000 Cp kJ/kg.K 1.003 1.005 1.008 1.013 1.020 1.029 1.040 1.051 1.063 1.075 1.087 1.099 1.121 1.142 C kJ/kg.K k Air 14.695 14.822 14.983 Cp C kJ/kg-K kJ/kg.K k Carbon dioxide, CO 0.716 1.401 0.791 0.718 1.400 0.846 0.721 1.398 0.895 0.726 1.395 0.939 0.733 1.391 0.978 0.742 0.753 1.381 1.387 1.014 1.046 0.764 1.376 1.075 0.776 1.370 1.102 1.126 0.788 1.364 0.800 1.359 1.148 0.812 1.354 0.834 1.344 0.855 1.336 1.169 1.204 1.234 Hydrogen, H 14.051 9.927 1.416 1.039 14.307 10.183 1.405 1.039 14.427 10.302 1.400 1.041 14.476 10.352 1.398 1.044 14.501 10.377 1.398 1.049 14.513 10.389 1.397 1.056 14.530 10.405 1.396 1.065 14.546 10.422 1.396 1.075 14.571 10.447 1.395 1.086 14.604 10.480 1.394 1.098 14.645 10.521 1.392 1.110 10.570 1.390 1.121 10.698 1.145 1.385 10.859 1.380 1.167 Cp kJ/kg-K Cu kJ/kg-K k Carbon monoxide, CO Nitrogen, N 0.742 1.400 0.743 0.602 1.314 1.039 0.657 1.288 1.040 0.706 1.268 1.043 0.750 1.252 1.047 0.790 1.239 1.054 0.825 1.229 1.063 0.857 1.220 1.075 0.886 1.213 1.087 0.913 1.207 1.100 0.937 1.202 1.113 1.197 1.126 0.959 0.980 1.193 1.139 1.015 1.186 1.163 1.181 1.045 1.185 0.913 1.400 0.918 0.744 1.399 0.928 0.747 1.397 0.941 0.752 1.395 0.956 0.759 0.972 1.391 0.768 1.387 0.988 0.778 1.382 1.003 1.376 1.017 1.371 1.031 1.365 1.043 0.789 0.801 0.813 0.825 1.360 1.054 0.849 1.349 1.074 0.870 1.341 1.090 0.743 1.400 0.744 1.399 1.398 1.395 1.392 0.746 0.751 0.757 0.767 1.387 0.778 1.382 0.790 1.376 0.803 1.370 0.816 1.364 0.829 1.358 0.842 1.353 0.866 1.343 0.888 1.335 Oxygen, 0 0.653 0.658 1.398 1.395 0.668 1.389 0.681 1.382 0.696 1.373 1.365 1.358 1.350 1.343 1.337 1.332 0.712 0.728 0.743 0.758 0.771 0.783 0.794 0.814 0.830 1.327 1.319 1.313 TABLE A-18 Ideal-gas properties of nitrogen, N h kJ/kmol kJ/kmol 0 6,391 6,683 6,975 7,266 T K 0 220 230 240 250 260 270 280 290 298 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 450 460 470 480 490 500 510 520 530 540 550 560 570 580 590 7,558 7,849 8,141 8,432 8,669 8,723 9,014 9,306 9,597 9,888 10,180 10,471 10,763 11,055 11,347 11,640 11,932 12,225 12,518 12,811 13,105 13,399 13,693 13,988 14,285 14,581 14,876 15,172 15,469 15,766 16,064 16,363 16,662 16,962 17,262 0 4,562 4,770 4,979 5,188 5,396 5,604 5,813 6,021 6,190 6,229 6,437 6,645 6,853 7,061 7,270 7,478 7,687 7,895 8,104 8,314 8,523 8,733 8,943 9,153 9,363 9,574 9,786 9,997 10,210 10,423 10,635 10,848 11,062 11,277 11,492 11,707 11,923 12,139 12,356 S kJ/kmol-K 0 182.639 183.938 185.180 186.370 187.514 188.614 189.673 190.695 191.502 191.682 192.638 193.562 194.459 195.328 196.173 196.995 197.794 198.572 199.331 200.071 200.794 201.499 202.189 202.863 203.523 204.170 204.803 205.424 206.033 206.630 207.216 207.792 208.358 208.914 209.461 209.999 210.528 211.049 211.562 T K 600 610 620 630 640 650 660 670 680 690 700 710 720 730 740 750 760 770 780 790 800 810 820 830 840 850 860 870 880 890 900 910 920 930 940 950 960 970 980 990 h kJ/kmol 17,563 17,864 18,166 18,468 18,772 19,075 19,380 19,685 19,991 20,297 20,604 20,912 21,220 21,529 21,839 22,149 22,460 22,772 23,085 23,398 23,714 24,027 24,342 24,658 24,974 25,292 25,610 25,928 26,248 26,568 26,890 27,210 27,532 27,854 28,178 28,501 28,826 29,151 29,476 29,803 kJ/kmol 12,574 12,792 13,011 13,230 13,450 13,671 13,892 14,114 14,337 14,560 14,784 15,008 15,234 15,460 15,686 15,913 16,141 16,370 16,599 16,830 17,061 17,292 17,524 17,757 17,990 18,224 18,459 18,695 18,931 19,168 19,407 19,644 19,883 20,122 20,362 20,603 20,844 21,086 21,328 21,571 5 kJ/kmol-K 212.066 212.564 213.055 213.541 214.018 214.489 214.954 215.413 215.866 216.314 216.756 217.192 217.624 218.059 218.472 218.889 219.301 219.709 220.113 220.512 220.907 221.298 221.684 222.067 222.447 222.822 223.194 223.562 223.927 224.288 224.647 225.002 225.353 225.701 226.047 226.389 226.728 227.064 227.398 227.728 TABLE A-18 Ideal-gas properties of nitrogen, N (Concluded) h kJ/kmol kJ/kmol 30,129 21,815 30,784 22,304 31,442 22,795 23,288 23,782 T K 1000 1020 1040 1060 1080 1100 1120 1140 1160 1180 1200 1220 1240 1260 1280 1300 1320 1340 1360 1380 1400 1420 1440 1460 1480 1500 1520 1540 1560 1580 1600 1620 1640 1660 1680 1700 1720 1740 32,101 32,762 33,426 34,092 34,760 35,430 36,104 36,777 37,452 38,129 38,807 39,488 40,170 40,853 41,539 42,227 42,915 43,605 44,295 44,988 45,682 46,377 47,073 47,771 48,470 49,168 49,869 50,571 51,275 51,980 52,686 53,393 54,099 54,807 55,516 24,280 24,780 25,282 25,786 26,291 26,799 27,308 27,819 28,331 28,845 29,361 29,378 30,398 30,919 31,441 31,964 32,489 33,014 33,543 34,071 34,601 35,133 35,665 36,197 36,732 37,268 37,806 38,344 38,884 39,424 39,965 40,507 41,049 5 kJ/kmol-K 228.057 228.706 229.344 229.973 230.591 231.199 231.799 232.391 232.973 233.549 234.115 234.673 235.223 235.766 236.302 236.831 237.353 237.867 238.376 238.878 239.375 239.865 240.350 240.827 241.301 241.768 242.228 242.685 243.137 243.585 244.028 244.464 244.896 245.324 245.747 246.166 246.580 246.990 T K 1760 1780 1800 1820 1840 1860 1880 1900 1920 1940 1960 1980 2000 2050 2100 2150 2200 2250 2300 2350 2400 2450 2500 2550 2600 2650 2700 2750 2800 2850 2900 2950 3000 3050 3100 3150 3200 3250 h kJ/kmol 56,227 56,938 57,651 58,363 59,075 59,790 60,504 61,220 61,936 62,654 63,381 64,090 64,810 66,612 68,417 70,226 72,040 73,856 75,676 77,496 79,320 81,149 82,981 84,814 86,650 88,488 90,328 92,171 94,014 95,859 97,705 99,556 101,407 103,260 105,115 106,972 108,830 110,690 kJ/kmol 41,594 42,139 42,685 43,231 43,777 44,324 44,873 45,423 45,973 46,524 47,075 47,627 48,181 49,567 50,957 52,351 53,749 55,149 56,553 57,958 59,366 60,779 62,195 63,613 65,033 66,455 67,880 69,306 70,734 72,163 73,593 75,028 76,464 77,902 79,341 80,782 82,224 83,668 5 kJ/kmol-K 247.396 247.798 248.195 248.589 248.979 249.365 249.748 250.128 250.502 250.874 251.242 251.607 251.969 252.858 253.726 254.578 255.412 256.227 257.027 257.810 258.580 259.332 260.073 260.799 261.512 262.213 262.902 263.577 264.241 264.895 265.538 266.170 266.793 267.404 268.007 268.601 269.186 269.763 TABLE A-20 Ideal-gas properties of carbon dioxide, CO h kJ/kmol 0 6,601 6,938 7,280 7,627 T K 0 220 230 240 250 260 270 280 290 298 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 450 460 470 480 490 500 510 520 530 540 550 560 570 580 590 7,979 8,335 8,697 9,063 9,364 9,431 9,807 10,186 10,570 10,959 11,351 11,748 12,148 12,552 12,960 13,372 13,787 14,206 14,628 15,054 15,483 15,916 16,351 16,791 17,232 17,678 18,126 18,576 19,029 19,485 19,945 20,407 20,870 21,337 21,807 kJ/kmol 0 4,772 5,026 5,285 5,548 5,817 6,091 6,369 6,651 6,885 6,939 7,230 7,526 7,826 8,131 8,439 8,752 9,068 9,392 9,718 10,046 10,378 10,714 11,053 11,393 11,742 12,091 12,444 12,800 13,158 13,521 13,885 14,253 14,622 14,996 15,372 15,751 16,131 16,515 16,902 5 kJ/kmol-K 202.966 204.464 205.920 207.337 208.717 210.062 211.376 212.660 213.685 213.915 215.146 216.351 217.534 218.694 219.831 220.948 222.044 223.122 224.182 225.225 226.250 227.258 228.252 229.230 230.194 231.144 232.080 233.004 233.916 234.814 235.700 236.575 237.439 238.292 239.135 239.962 240.789 241.602 242.405 T K 600 610 620 630 640 650 660 670 680 690 700 710 720 730 740 750 760 770 780 790 800 810 820 830 840 850 860 870 880 890 900 910 920 930 940 950 960 970 980 990 h kJ/kmol 22,280 22,754 23,231 23,709 24,190 24,674 25,160 25,648 26,138 26,631 27,125 27,622 28,121 28,622 29,124 29,629 30,135 30,644 31,154 31,665 32,179 32,694 33,212 33,730 34,251 34,773 35,296 35,821 36,347 36,876 37,405 37,935 38,467 39,000 39,535 40,070 40,607 41,145 41,685 42,226 kJ/kmol 17,291 17,683 18,076 18,471 18,869 19,270 19,672 20,078 20,484 20,894 21,305 21,719 22,134 22,522 22,972 23,393 23,817 24,242 24,669 25,097 25,527 25,959 26,394 26,829 27,267 27,706 28,125 28,588 29,031 29,476 29,922 30,369 30,818 31,268 31,719 32,171 32,625 33,081 33,537 33,995 5 kJ/kmol-K 243.199 243.983 244.758 245.524 246.282 247.032 247.773 248.507 249.233 249.952 250.663 251.368 252.065 252.755 253.439 254.117 254.787 255.452 256.110 256.762 257.408 258.048 258.682 259.311 259.934 260.551 261.164 261.770 262.371 262.968 263.559 264.146 264.728 265.304 265.877 266.444 267.007 267.566 268.119 268.670 TABLE A-20 Ideal-gas properties of carbon dioxide, CO (Concluded) h T K 1000 1020 1040 1060 1080 1100 1120 1140 1160 1180 1200 1220 1240 1260 1280 1300 1320 1340 1360 1380 1400 1420 1440 1460 1480 1500 1520 1540 1560 1580 1600 1620 1640 1660 1680 1700 1720 1740 kJ/kmol 42,769 43,859 44,953 46,051 47,153 48,258 49,369 50,484 51,602 52,724 53,848 54,977 56,108 57,244 58,381 59,522 60,666 61,813 62,963 64,116 65,271 66,427 67,586 68,748 66,911 71,078 72,246 73,417 74,590 76,767 76,944 78,123 79,303 80,486 81,670 82,856 84,043 85,231 kJ/kmol 34,455 35,378 36,306 37,238 38,174 39,112 40,057 41,006 41,957 42,913 43,871 44,834 45,799 46,768 47,739 48,713 49,691 50,672 51,656 52,643 53,631 54,621 55,614 56,609 57,606 58,606 59,609 60,613 61,620 62,630 63,741 64,653 65,668 66,592 67,702 68,721 69,742 70,764 5 kJ/kmol-K 269.215 270.293 271.354 272.400 273.430 274.445 275.444 276.430 277.403 278.361 297.307 280.238 281.158 282.066 282.962 283.847 284.722 285.586 286.439 287.283 288.106 288.934 289.743 290.542 291.333 292.114 292.888 292.654 294.411 295.161 295.901 296.632 297.356 298.072 298.781 299.482 300.177 300.863 T K 1760 1780 1800 1820 1840 1860 1880 1900 1920 1940 1960 1980 2000 2050 2100 2150 2200 2250 2300 2350 2400 2450 2500 2550 2600 2650 2700 2750 2800 2850 2900 2950 3000 3050 3100 3150 3200 3250 h kJ/kmol 86,420 87,612 88,806 90,000 91,196 92,394 93,593 94,793 95,995 97, 197 98,401 99,606 100,804 103,835 106,864 109,898 112,939 115,984 119,035 122,091 125,152 128,219 131,290 134,368 137,449 140,533 143,620 146,713 149,808 152,908 156,009 159,117 162,226 165,341 168,456 171,576 174,695 177,822 kJ/kmol 71,787 72,812 73,840 74,868 75,897 76,929 77,962 78,996 80,031 81,067 82,105 83,144 84,185 86,791 89,404 92,023 94,648 97,277 99,912 102,552 105,197 107,849 110,504 113,166 115,832 118,500 121,172 123,849 126,528 129,212 131,898 134,589 137,283 139,982 142,681 145,385 148,089 150,801 5 kJ/kmol-K 301.543 302.217 302.884 303.544 304.198 304.845 305.487 306.122 306.751 307.374 307.992 308.604 309.210 310.701 312.160 313.589 314.988 316.356 317.695 319.011 320.302 321.566 322.808 324.026 325.222 326.396 327.549 328.684 329.800 330.896 331.975 333.037 334.084 335.114 336.126 337.124 338.109 339.069 13-88 A mixture of carbon dioxide and nitrogen flows through a converging nozzle. The mixture leaves the nozzle at a temperature of 500 K with a velocity of 360 m/s. If the velocity is equal to the speed of sound at the exit temperature, determine the required makeup of the mixture on a mass basis. TABLE A -1 Molar mass, gas constant, and critical-point properties Substance Air Ammonia Argon Benzene Bromine n-Butane Carbon dioxide Carbon monoxide Carbon tetrachloride Chlorine Chloroform Dichlorodifluoromethane (R-12) Dichlorofluoromethane (R-21) Ethane Ethyl alcohol Ethylene Helium n-Hexane Hydrogen (normal) Krypton Methane Methyl alcohol Methyl chloride Neon Nitrogen Nitrous oxide Oxygen Propane Propylene Sulfur dioxide Tetrafluoroethane (R-134a) Trichlorofluoromethane (R-11) Water Xenon Formula NH3 Ar CH6 Br C4H10 CO CO CCI4 Cl CHCI 3 CCIF CHCIF CH6 CH5OH CH4 He C6H14 H Kr CH4 CHOH CH3CI Ne N NO 0 C3H8 C3H6 SO CF3CHF CCI3F HO Xe Molar mass, M kg/kmol 28.97 17.03 39.948 78.115 159.808 58.124 44.01 28.011 153.82 70.906 119.38 120.91 102.92 30.070 46.07 28.054 4.003 86.179 2.016 83.80 16.043 32.042 50.488 20.183 28.013 44.013 31.999 44.097 42.081 64.063 102.03 137.37 18.015 131.30 Gas constant, R kJ/kg-K* 0.2870 0.4882 0.2081 0.1064 0.0520 0.1430 0.1889 0.2968 0.05405 0.1173 0.06964 0.06876 0.08078 0.2765 0.1805 0.2964 2.0769 0.09647 4.1240 0.09921 0.5182 0.2595 0.1647 0.4119 0.2968 0.1889 0.2598 0.1885 0.1976 0.1298 0.08149 0.06052 0.4615 0.06332 Critical-point properties Temperature, Pressure, MPa K 132.5 405.5 151 562 584 425.2 304.2 133 556.4 417 536.6 384.7 451.7 305.5 516 282.4 5.3 507.9 33.3 209.4 191.1 513.2 416.3 44.5 126.2 309.7 154.8 370 365 430.7 374.2 471.2 647.1 289.8 3.77 11.28 4.86 4.92 10.34 3.80 7.39 3.50 4.56 7.71 5.47 4.01 5.17 4.48 6.38 5.12 0.23 3.03 1.30 5.50 4.64 7.95 6.68 2.73 3.39 7.27 5.08 4.26 4.62 7.88 4.059 4.38 22.06 5.88 Volume, m/kmol 0.0883 0.0724 0.0749 0.2603 0.1355 0.2547 0.0943 0.0930 0.2759 0.1242 0.2403 0.2179 0.1973 0.1480 0.1673 0.1242 0.0578 0.3677 0.0649 0.0924 0.0993 0.1180 0.1430 0.0417 0.0899 0.0961 0.0780 0.1998 0.1810 0.1217 0.1993 0.2478 0.0560 0.1186 TABLE A-2 Ideal-gas specific heats of various common gases (a) At 300 K Gas Air Argon Butane Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon Nitrogen Octane Oxygen Propane Steam Formula | Ar C4H10 CO CO CH6 CH4 He H CH4 Ne N C8H18 0 C3H8 HO Gas constant, R kJ/kg.K 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 0.0729 0.2598 0.1885 0.4615 Cp kJ/kg.K 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 1.7113 0.918 1.6794 1.8723 Cv kJ/kg-K 0.718 0.3122 1.5734 0.657 0.744 1.4897 1.2518 3.1156 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 k 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 1.044 1.395 1.126 1.327 TABLE A-2 Ideal-gas specific heats of various common gases (Continued) (b) At various temperatures Temperature, K 250 300 350 400 450 500 550 600 650 700 750 800 900 1000 250 300 350 400 450 500 550 600 650 700 750 800 900 1000 Cp kJ/kg.K 1.003 1.005 1.008 1.013 1.020 1.029 1.040 1.051 1.063 1.075 1.087 1.099 1.121 1.142 C kJ/kg.K k Air 14.695 14.822 14.983 Cp C kJ/kg-K kJ/kg.K k Carbon dioxide, CO 0.716 1.401 0.791 0.718 1.400 0.846 0.721 1.398 0.895 0.726 1.395 0.939 0.733 1.391 0.978 0.742 0.753 1.381 1.387 1.014 1.046 0.764 1.376 1.075 0.776 1.370 1.102 1.126 0.788 1.364 0.800 1.359 1.148 0.812 1.354 0.834 1.344 0.855 1.336 1.169 1.204 1.234 Hydrogen, H 14.051 9.927 1.416 1.039 14.307 10.183 1.405 1.039 14.427 10.302 1.400 1.041 14.476 10.352 1.398 1.044 14.501 10.377 1.398 1.049 14.513 10.389 1.397 1.056 14.530 10.405 1.396 1.065 14.546 10.422 1.396 1.075 14.571 10.447 1.395 1.086 14.604 10.480 1.394 1.098 14.645 10.521 1.392 1.110 10.570 1.390 1.121 10.698 1.145 1.385 10.859 1.380 1.167 Cp kJ/kg-K Cu kJ/kg-K k Carbon monoxide, CO Nitrogen, N 0.742 1.400 0.743 0.602 1.314 1.039 0.657 1.288 1.040 0.706 1.268 1.043 0.750 1.252 1.047 0.790 1.239 1.054 0.825 1.229 1.063 0.857 1.220 1.075 0.886 1.213 1.087 0.913 1.207 1.100 0.937 1.202 1.113 1.197 1.126 0.959 0.980 1.193 1.139 1.015 1.186 1.163 1.181 1.045 1.185 0.913 1.400 0.918 0.744 1.399 0.928 0.747 1.397 0.941 0.752 1.395 0.956 0.759 0.972 1.391 0.768 1.387 0.988 0.778 1.382 1.003 1.376 1.017 1.371 1.031 1.365 1.043 0.789 0.801 0.813 0.825 1.360 1.054 0.849 1.349 1.074 0.870 1.341 1.090 0.743 1.400 0.744 1.399 1.398 1.395 1.392 0.746 0.751 0.757 0.767 1.387 0.778 1.382 0.790 1.376 0.803 1.370 0.816 1.364 0.829 1.358 0.842 1.353 0.866 1.343 0.888 1.335 Oxygen, 0 0.653 0.658 1.398 1.395 0.668 1.389 0.681 1.382 0.696 1.373 1.365 1.358 1.350 1.343 1.337 1.332 0.712 0.728 0.743 0.758 0.771 0.783 0.794 0.814 0.830 1.327 1.319 1.313 TABLE A-18 Ideal-gas properties of nitrogen, N h kJ/kmol kJ/kmol 0 6,391 6,683 6,975 7,266 T K 0 220 230 240 250 260 270 280 290 298 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 450 460 470 480 490 500 510 520 530 540 550 560 570 580 590 7,558 7,849 8,141 8,432 8,669 8,723 9,014 9,306 9,597 9,888 10,180 10,471 10,763 11,055 11,347 11,640 11,932 12,225 12,518 12,811 13,105 13,399 13,693 13,988 14,285 14,581 14,876 15,172 15,469 15,766 16,064 16,363 16,662 16,962 17,262 0 4,562 4,770 4,979 5,188 5,396 5,604 5,813 6,021 6,190 6,229 6,437 6,645 6,853 7,061 7,270 7,478 7,687 7,895 8,104 8,314 8,523 8,733 8,943 9,153 9,363 9,574 9,786 9,997 10,210 10,423 10,635 10,848 11,062 11,277 11,492 11,707 11,923 12,139 12,356 S kJ/kmol-K 0 182.639 183.938 185.180 186.370 187.514 188.614 189.673 190.695 191.502 191.682 192.638 193.562 194.459 195.328 196.173 196.995 197.794 198.572 199.331 200.071 200.794 201.499 202.189 202.863 203.523 204.170 204.803 205.424 206.033 206.630 207.216 207.792 208.358 208.914 209.461 209.999 210.528 211.049 211.562 T K 600 610 620 630 640 650 660 670 680 690 700 710 720 730 740 750 760 770 780 790 800 810 820 830 840 850 860 870 880 890 900 910 920 930 940 950 960 970 980 990 h kJ/kmol 17,563 17,864 18,166 18,468 18,772 19,075 19,380 19,685 19,991 20,297 20,604 20,912 21,220 21,529 21,839 22,149 22,460 22,772 23,085 23,398 23,714 24,027 24,342 24,658 24,974 25,292 25,610 25,928 26,248 26,568 26,890 27,210 27,532 27,854 28,178 28,501 28,826 29,151 29,476 29,803 kJ/kmol 12,574 12,792 13,011 13,230 13,450 13,671 13,892 14,114 14,337 14,560 14,784 15,008 15,234 15,460 15,686 15,913 16,141 16,370 16,599 16,830 17,061 17,292 17,524 17,757 17,990 18,224 18,459 18,695 18,931 19,168 19,407 19,644 19,883 20,122 20,362 20,603 20,844 21,086 21,328 21,571 5 kJ/kmol-K 212.066 212.564 213.055 213.541 214.018 214.489 214.954 215.413 215.866 216.314 216.756 217.192 217.624 218.059 218.472 218.889 219.301 219.709 220.113 220.512 220.907 221.298 221.684 222.067 222.447 222.822 223.194 223.562 223.927 224.288 224.647 225.002 225.353 225.701 226.047 226.389 226.728 227.064 227.398 227.728 TABLE A-18 Ideal-gas properties of nitrogen, N (Concluded) h kJ/kmol kJ/kmol 30,129 21,815 30,784 22,304 31,442 22,795 23,288 23,782 T K 1000 1020 1040 1060 1080 1100 1120 1140 1160 1180 1200 1220 1240 1260 1280 1300 1320 1340 1360 1380 1400 1420 1440 1460 1480 1500 1520 1540 1560 1580 1600 1620 1640 1660 1680 1700 1720 1740 32,101 32,762 33,426 34,092 34,760 35,430 36,104 36,777 37,452 38,129 38,807 39,488 40,170 40,853 41,539 42,227 42,915 43,605 44,295 44,988 45,682 46,377 47,073 47,771 48,470 49,168 49,869 50,571 51,275 51,980 52,686 53,393 54,099 54,807 55,516 24,280 24,780 25,282 25,786 26,291 26,799 27,308 27,819 28,331 28,845 29,361 29,378 30,398 30,919 31,441 31,964 32,489 33,014 33,543 34,071 34,601 35,133 35,665 36,197 36,732 37,268 37,806 38,344 38,884 39,424 39,965 40,507 41,049 5 kJ/kmol-K 228.057 228.706 229.344 229.973 230.591 231.199 231.799 232.391 232.973 233.549 234.115 234.673 235.223 235.766 236.302 236.831 237.353 237.867 238.376 238.878 239.375 239.865 240.350 240.827 241.301 241.768 242.228 242.685 243.137 243.585 244.028 244.464 244.896 245.324 245.747 246.166 246.580 246.990 T K 1760 1780 1800 1820 1840 1860 1880 1900 1920 1940 1960 1980 2000 2050 2100 2150 2200 2250 2300 2350 2400 2450 2500 2550 2600 2650 2700 2750 2800 2850 2900 2950 3000 3050 3100 3150 3200 3250 h kJ/kmol 56,227 56,938 57,651 58,363 59,075 59,790 60,504 61,220 61,936 62,654 63,381 64,090 64,810 66,612 68,417 70,226 72,040 73,856 75,676 77,496 79,320 81,149 82,981 84,814 86,650 88,488 90,328 92,171 94,014 95,859 97,705 99,556 101,407 103,260 105,115 106,972 108,830 110,690 kJ/kmol 41,594 42,139 42,685 43,231 43,777 44,324 44,873 45,423 45,973 46,524 47,075 47,627 48,181 49,567 50,957 52,351 53,749 55,149 56,553 57,958 59,366 60,779 62,195 63,613 65,033 66,455 67,880 69,306 70,734 72,163 73,593 75,028 76,464 77,902 79,341 80,782 82,224 83,668 5 kJ/kmol-K 247.396 247.798 248.195 248.589 248.979 249.365 249.748 250.128 250.502 250.874 251.242 251.607 251.969 252.858 253.726 254.578 255.412 256.227 257.027 257.810 258.580 259.332 260.073 260.799 261.512 262.213 262.902 263.577 264.241 264.895 265.538 266.170 266.793 267.404 268.007 268.601 269.186 269.763 TABLE A-20 Ideal-gas properties of carbon dioxide, CO h kJ/kmol 0 6,601 6,938 7,280 7,627 T K 0 220 230 240 250 260 270 280 290 298 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 450 460 470 480 490 500 510 520 530 540 550 560 570 580 590 7,979 8,335 8,697 9,063 9,364 9,431 9,807 10,186 10,570 10,959 11,351 11,748 12,148 12,552 12,960 13,372 13,787 14,206 14,628 15,054 15,483 15,916 16,351 16,791 17,232 17,678 18,126 18,576 19,029 19,485 19,945 20,407 20,870 21,337 21,807 kJ/kmol 0 4,772 5,026 5,285 5,548 5,817 6,091 6,369 6,651 6,885 6,939 7,230 7,526 7,826 8,131 8,439 8,752 9,068 9,392 9,718 10,046 10,378 10,714 11,053 11,393 11,742 12,091 12,444 12,800 13,158 13,521 13,885 14,253 14,622 14,996 15,372 15,751 16,131 16,515 16,902 5 kJ/kmol-K 202.966 204.464 205.920 207.337 208.717 210.062 211.376 212.660 213.685 213.915 215.146 216.351 217.534 218.694 219.831 220.948 222.044 223.122 224.182 225.225 226.250 227.258 228.252 229.230 230.194 231.144 232.080 233.004 233.916 234.814 235.700 236.575 237.439 238.292 239.135 239.962 240.789 241.602 242.405 T K 600 610 620 630 640 650 660 670 680 690 700 710 720 730 740 750 760 770 780 790 800 810 820 830 840 850 860 870 880 890 900 910 920 930 940 950 960 970 980 990 h kJ/kmol 22,280 22,754 23,231 23,709 24,190 24,674 25,160 25,648 26,138 26,631 27,125 27,622 28,121 28,622 29,124 29,629 30,135 30,644 31,154 31,665 32,179 32,694 33,212 33,730 34,251 34,773 35,296 35,821 36,347 36,876 37,405 37,935 38,467 39,000 39,535 40,070 40,607 41,145 41,685 42,226 kJ/kmol 17,291 17,683 18,076 18,471 18,869 19,270 19,672 20,078 20,484 20,894 21,305 21,719 22,134 22,522 22,972 23,393 23,817 24,242 24,669 25,097 25,527 25,959 26,394 26,829 27,267 27,706 28,125 28,588 29,031 29,476 29,922 30,369 30,818 31,268 31,719 32,171 32,625 33,081 33,537 33,995 5 kJ/kmol-K 243.199 243.983 244.758 245.524 246.282 247.032 247.773 248.507 249.233 249.952 250.663 251.368 252.065 252.755 253.439 254.117 254.787 255.452 256.110 256.762 257.408 258.048 258.682 259.311 259.934 260.551 261.164 261.770 262.371 262.968 263.559 264.146 264.728 265.304 265.877 266.444 267.007 267.566 268.119 268.670 TABLE A-20 Ideal-gas properties of carbon dioxide, CO (Concluded) h T K 1000 1020 1040 1060 1080 1100 1120 1140 1160 1180 1200 1220 1240 1260 1280 1300 1320 1340 1360 1380 1400 1420 1440 1460 1480 1500 1520 1540 1560 1580 1600 1620 1640 1660 1680 1700 1720 1740 kJ/kmol 42,769 43,859 44,953 46,051 47,153 48,258 49,369 50,484 51,602 52,724 53,848 54,977 56,108 57,244 58,381 59,522 60,666 61,813 62,963 64,116 65,271 66,427 67,586 68,748 66,911 71,078 72,246 73,417 74,590 76,767 76,944 78,123 79,303 80,486 81,670 82,856 84,043 85,231 kJ/kmol 34,455 35,378 36,306 37,238 38,174 39,112 40,057 41,006 41,957 42,913 43,871 44,834 45,799 46,768 47,739 48,713 49,691 50,672 51,656 52,643 53,631 54,621 55,614 56,609 57,606 58,606 59,609 60,613 61,620 62,630 63,741 64,653 65,668 66,592 67,702 68,721 69,742 70,764 5 kJ/kmol-K 269.215 270.293 271.354 272.400 273.430 274.445 275.444 276.430 277.403 278.361 297.307 280.238 281.158 282.066 282.962 283.847 284.722 285.586 286.439 287.283 288.106 288.934 289.743 290.542 291.333 292.114 292.888 292.654 294.411 295.161 295.901 296.632 297.356 298.072 298.781 299.482 300.177 300.863 T K 1760 1780 1800 1820 1840 1860 1880 1900 1920 1940 1960 1980 2000 2050 2100 2150 2200 2250 2300 2350 2400 2450 2500 2550 2600 2650 2700 2750 2800 2850 2900 2950 3000 3050 3100 3150 3200 3250 h kJ/kmol 86,420 87,612 88,806 90,000 91,196 92,394 93,593 94,793 95,995 97, 197 98,401 99,606 100,804 103,835 106,864 109,898 112,939 115,984 119,035 122,091 125,152 128,219 131,290 134,368 137,449 140,533 143,620 146,713 149,808 152,908 156,009 159,117 162,226 165,341 168,456 171,576 174,695 177,822 kJ/kmol 71,787 72,812 73,840 74,868 75,897 76,929 77,962 78,996 80,031 81,067 82,105 83,144 84,185 86,791 89,404 92,023 94,648 97,277 99,912 102,552 105,197 107,849 110,504 113,166 115,832 118,500 121,172 123,849 126,528 129,212 131,898 134,589 137,283 139,982 142,681 145,385 148,089 150,801 5 kJ/kmol-K 301.543 302.217 302.884 303.544 304.198 304.845 305.487 306.122 306.751 307.374 307.992 308.604 309.210 310.701 312.160 313.589 314.988 316.356 317.695 319.011 320.302 321.566 322.808 324.026 325.222 326.396 327.549 328.684 329.800 330.896 331.975 333.037 334.084 335.114 336.126 337.124 338.109 339.069
Expert Answer:

Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Posted Date:
Students also viewed these mechanical engineering questions
-
A mixture of carbon dioxide and nitrogen flows through a converging nozzle. The mixture leaves the nozzle at a temperature of 500 K with a velocity of 360 m/s. If the velocity is equal to the speed...
-
A dry mixture of carbon dioxide and nitrogen (30% carbon dioxide and 70% nitrogen by volume) is initially at 90 F and 14.7 psia. Moisture is then added to the mixture until it becomes saturated at 90...
-
Define the necessary and sufficient condition for two transactions to be serialisable. Give an example of a non-serialisable execution of a pair of transactions. [3 marks] (c) Define the necessary...
-
Name the two t tests used in hypothesis testing for one sample and for two independent samples.
-
Indicate the components of the balance sheet and income statement that will change as a consequence of the following transactions: CA NCA CL NCL OEREV EXP RE 1. Factory equipment purchased for cash...
-
Defendants Jack and Claire Lein owned and lived on Willow Creek Farm from 1980 through 2004. The farm manager, Stewart, and his girlfriend, plaintiff Tambra Curtis, also lived on the farm during this...
-
a. Armstrong County established a County Office Building Construction Fund to account for a project that was expected to take less than one year to complete. The Countys fiscal year ends on June 30....
-
what the values of NoSQL are from retail business perspective? Do you agree? Why?
-
4(a) Calculate the Laplace transform of the periodic function shown below. f(t)A [5] 1. 0 1 2 3 4 5 t 4(b) For the circuit shown below, determine vo(t) for t > 0. Given v(0) = 3 V, Rf = 80 k2, R1 =...
-
P1 contributed securities worth $50,000 that he purchased six months ago for $35,000 in ABC Partnership in exchange for a 50 percent profits and capital interest. Determine ABC's inside basis and...
-
What is the big problem that you are trying to solve with a digital transformation (this is connected to the drivers of digital transformation you identified in week 2)? Who is most affected by this...
-
Temperature differences on the Rankine scale are identical to differences on the Fahrenheit scale, but absolute zero is given as 0R. (a) Find a relationship converting the temperatures T of the...
-
How can the design team ensure that the database schema for the e-learning platform facilitates effective course enrollment, progress tracking, and assessment management for both instructors and...
-
What you believe are important elements of organizational behavior and how these elements can challenge the effectiveness of the correctional field?
-
Your company's new portable phone/music player/PDA/bottle washer, the RunMan, will compete against the established market leader, the iNod, in a saturated market. (Thus, for each device you sell, one...
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
Repeat Problem 4.92 for a surface temperature of 150 C, with all other data remaining unchanged. Problem 4.92 A surface is maintained at 100 C and is enclosed by very large surrounding surfaces at 80...
-
During the operation of a steam power plant, the steam flow rate is 227,000 kg/hr with turbine inlet conditions of 4 MPa and 800 K and turbine exhaust (condenser inlet) conditions of 8 kPa and 90%...
-
Natural gas (methane) is mixed with air in a molar ratio of 1:6 (1 kmol of methane with 6 kmol of air). Determine the volume fractions of each component and the apparent molecular weight (kg/kmol) of...
-
Briefly describe what is meant by DEI efforts.
-
Explain each of the four examples of a bona fide occupational qualification.
-
What important precedents were set by the Griggs v. Duke Power Company case? The Albemarle v. Moody case?

Study smarter with the SolutionInn App


