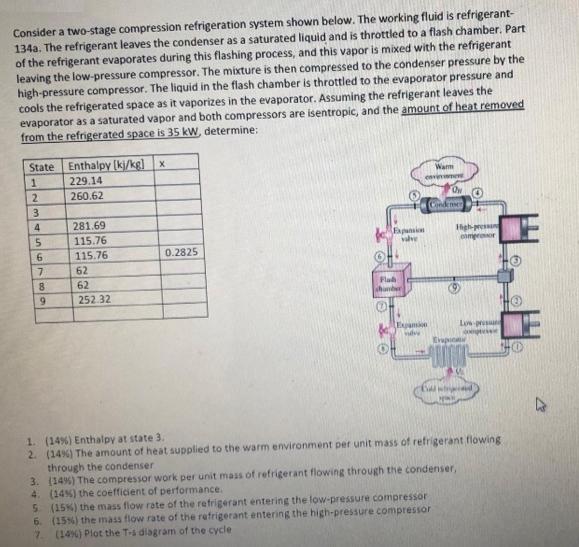
Consider a two-stage compression refrigeration system shown below. The working fluid is refrigerant- 134a. The refrigerant...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider a two-stage compression refrigeration system shown below. The working fluid is refrigerant- 134a. The refrigerant leaves the condenser as a saturated liquid and is throttled to a flash chamber. Part of the refrigerant evaporates during this flashing process, and this vapor is mixed with the refrigerant leaving the low-pressure compressor. The mixture is then compressed to the condenser pressure by the high-pressure compressor. The liquid in the flash chamber is throttled to the evaporator pressure and cools the refrigerated space as it vaporizes in the evaporator. Assuming the refrigerant leaves the evaporator as a saturated vapor and both compressors are isentropic, and the amount of heat removed from the refrigerated space is 35 kW, determine: State Enthalpy (kj/kg] x 229.14 Wam 260.62 Cindenser 281.69 Fapansin Igh-pressan Comeroor 115.76 6. 115.76 0.2825 62 8. 62 Flah chunber 9. 252.32 Expamion Low-prus 1. (14%) Enthalpy at state 3. 2. (14%) The amount of heat supplied to the warm environment per unit mass of refrigerant flowing through the condenser 3. (14%) The compressor work per unit mass of refrigerant flowing through the condenser, 4. (14%) the coefficient of performance. (15%) the mass flow rate of the refrigerant entering the low-pressure compressor 6. (15%) the mass flow rate of the refrigerant entering the high-pressure compressor 7 (14%) Plot the T-i diagram of the cycle 2345 Consider a two-stage compression refrigeration system shown below. The working fluid is refrigerant- 134a. The refrigerant leaves the condenser as a saturated liquid and is throttled to a flash chamber. Part of the refrigerant evaporates during this flashing process, and this vapor is mixed with the refrigerant leaving the low-pressure compressor. The mixture is then compressed to the condenser pressure by the high-pressure compressor. The liquid in the flash chamber is throttled to the evaporator pressure and cools the refrigerated space as it vaporizes in the evaporator. Assuming the refrigerant leaves the evaporator as a saturated vapor and both compressors are isentropic, and the amount of heat removed from the refrigerated space is 35 kW, determine: State Enthalpy (kj/kg] x 229.14 Wam 260.62 Cindenser 281.69 Fapansin Igh-pressan Comeroor 115.76 6. 115.76 0.2825 62 8. 62 Flah chunber 9. 252.32 Expamion Low-prus 1. (14%) Enthalpy at state 3. 2. (14%) The amount of heat supplied to the warm environment per unit mass of refrigerant flowing through the condenser 3. (14%) The compressor work per unit mass of refrigerant flowing through the condenser, 4. (14%) the coefficient of performance. (15%) the mass flow rate of the refrigerant entering the low-pressure compressor 6. (15%) the mass flow rate of the refrigerant entering the high-pressure compressor 7 (14%) Plot the T-i diagram of the cycle 2345
Expert Answer:

Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these mechanical engineering questions
-
Refrigerant 134a enters an adiabatic compressor as a saturated vapor at 70F. It leaves the compressor at 160 psia and 120F. If the mass flow rate of the refrigerant is 10 lbm/s, determine the...
-
Refrigerant 134a enters a compressor with a mass flow rate of 5 kg/s and a negligible velocity. The refrigerant enters the compressor as a saturated vapor at 10°C and leaves the compressor at...
-
Given the following class, which statement is correct? A. The class does not contain any security issues. B. The class contains exactly one security issue. C. The class contains exactly two security...
-
Continue with the scenario of question 11-the new process in the United States and the resulting change in the international equilibrium price ratio. Focus now on effects in the rest of the world. a....
-
What price would you expect to pay for the Kenny Corp. bond? What is the bond's current yield? To calculate the number of years until maturity, assume that it is currently January 15, 2016. All of...
-
Why do we need different levels of testing? Cant we just test the finished software?
-
Alberton Co. uses a standard costing system to account for its production of toys. Plastic is added at the start of production; labor and overhead are incurred at equal rates throughout the process....
-
Melissa recently paid $660 for round-trip airfare to San Francisco to attend a business conference for three days. Melissa also paid the following expenses: $650 fee to register for the conference,...
-
Robert Brown applied for admission to the University of Kansas School of Law. Brown answered "no" to questions on the application asking if he had a criminal history and acknowledged that a false...
-
What is the total conversion cost if a company has $ 1 0 , 0 0 0 1 0 , 0 0 0 in the beginning work - in - process inventory and the cost incurred for the period is $ 3 0 , 0 0 0 ? 3 0 , 0 0 0 ? a ....
-
Discuss the role of the cell cycle in stem cell self-renewal, differentiation, and tissue regeneration, including the coordination of cell cycle progression with developmental signaling pathways,...
-
Discuss feminist ideology a) What is feminism as an ideology? How do feminists explain and understand the root cause of gender inequality and what do they propose to do? Include a discussion of...
-
How do external cues, such as growth factors, nutrient availability, and environmental stressors, integrate with intracellular signaling pathways to modulate cell cycle progression, including the...
-
An injury insurance policy pays out $6000 if a person is seriously injured with permanent disability, $1000 if a person is seriously injured without permanent disability and $0 otherwise. The clients...
-
How does the cell cycle, a highly regulated process governing the sequential progression of cells through interphase, mitosis, and cytokinesis, ensure faithful duplication of genetic material,...
-
Using the following facts, calculate the total program, management and general and fundraising expenses that should be reflected on the Organization's statement of activities for the following...
-
Evaluate the line integral, where C is the given curve. C x 2 dx + y 2 dy, C consists of the arc of the circle x 2 + y 2 = 4 from (2, 0) to (0, 2) followed by the line segment from (0, 2) to (4, 3)
-
A psychrometer is used to measure a dry-bulb temperature of 22 8C and a wet-bulb temperature of 15 8C in a 95 kPa atmosphere. Determine the relative humidity and the humidity ratio for this air. What...
-
A torque of 50 Nm is required to rotate the paddle wheel shown in Fig. 6.47 at 60 rad/s. If it rotates for 2 minutes during which time 200 kJ of heat is transferred to the air from a reservoir,...
-
Four kilograms of saturated steam are being condensed at a constant pressure of 120 kPa in the cylinder of Fig. 6.51 by transferring heat to the 25° C surroundings. Calculate the entropy...
-
The first 5 years of a revenue requirements analysis for a public utility is presented below. The fraction of total capitalization that is debt is 35 percent. Answer the following questions. a. What...
-
True or False: When several independent, indivisible investments are available, form the investment portfolio so that the present worth of the portfolio is maximized.
-
Consolidated Rural Electric Coop (CREC) provides reliable electrical service to a large rural community. "Reading" the meters is very troublesome and costly due to the distance between service...

Study smarter with the SolutionInn App


