14) Which of the following compounds is optically active: a. trans-1-bromo-3-chlorocyclobutane b. cis-1,4-dimethylcyclohexane c. cis-1,3-dimethylcyclohexane d....
Fantastic news! We've Found the answer you've been seeking!
Question:


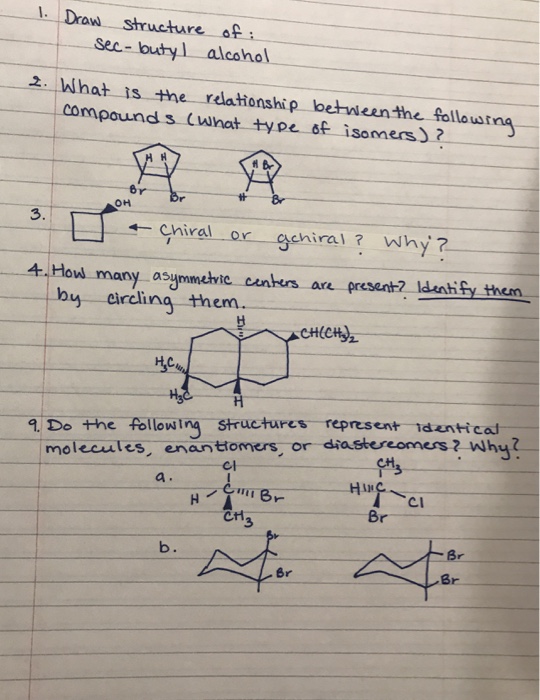
Transcribed Image Text:
14) Which of the following compounds is optically active: a. trans-1-bromo-3-chlorocyclobutane b. cis-1,4-dimethylcyclohexane c. cis-1,3-dimethylcyclohexane d. trans-1,3-dimethylcyclohexane CHS MC CH3 CHE CH3 CH3 Br 6) Which of the following compounds (choose only one) is never chiral? (4 points) 2,3-dibromobutane; 1,3-dibromobutane; 1,2-dichlorobutane; 1,4-dibromobutane; and 1-bromo-2-chlorobutane. 1. Draw structure of: Sec-butyl alcohol 2. What is the relationship between the following compounds (what type of isomers)? 3. HH Br OH Chiral or gchiral? Why? 4. How many asymmetric centers are present? Identify them by circling them. CH(CH₂)₂ a. Br H A H₂C H₂C 9. Do the following structures represent identical molecules, enantiomers, or diastereomers? Why? CI CH3 I C Br A CH3 b. Br H- Br HINC Br CI Br Br 14) Which of the following compounds is optically active: a. trans-1-bromo-3-chlorocyclobutane b. cis-1,4-dimethylcyclohexane c. cis-1,3-dimethylcyclohexane d. trans-1,3-dimethylcyclohexane CHS MC CH3 CHE CH3 CH3 Br 6) Which of the following compounds (choose only one) is never chiral? (4 points) 2,3-dibromobutane; 1,3-dibromobutane; 1,2-dichlorobutane; 1,4-dibromobutane; and 1-bromo-2-chlorobutane. 1. Draw structure of: Sec-butyl alcohol 2. What is the relationship between the following compounds (what type of isomers)? 3. HH Br OH Chiral or gchiral? Why? 4. How many asymmetric centers are present? Identify them by circling them. CH(CH₂)₂ a. Br H A H₂C H₂C 9. Do the following structures represent identical molecules, enantiomers, or diastereomers? Why? CI CH3 I C Br A CH3 b. Br H- Br HINC Br CI Br Br
Expert Answer:
Answer rating: 100% (QA)
14 D option is correct Explanation Only compound D does not have a p... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following compounds is easier to decarboxylate? NCOH
-
Which of the following compounds is more easily decarboxylated? CH2CH2 or
-
Which of the following compounds is more likely to exhibit activity as a tranquilizer? CH3 CH CH O or CH C
-
The largest government expenditure for Japan & the US is... a) military / defense b) transfer payments c) education / healthcare d) infrastructure
-
a. Based on historical data and assuming less-than-full employment, periods of sharp acceleration in the growth rate of the money supply tend to be associated initially with: i. Periods of economic...
-
Speculate as to why Canon is so unusual in its degree of independence from Japans domestic market, compared to most other Japanese firms.
-
Find the = 0.01 critical value for the chi-square statistic with 5 degrees of freedom.
-
Digiread Technologies Ltd. (DTL) is a privately held distributor of e-readers and tablets. The Calgary-based company is well established and is currently managed by its founder, Erin Jacobs. The...
-
A 25-year maturity mortgage-backed bond is issued. The bond has a par value of $10,000 and promises to pay an 8 percent annual coupon. At issue, bond market investors require a 12 percent interest...
-
Kitchen World Inc. is a Canadian controlled private corporation (CCPC) that operates a retail business selling cooking utensils, knives and small appliances for both professional chefs and home...
-
Aria Acoustics, Inc. (AAI), projects unit sales for a new seven-octave voice emulation implant as follows: Year Unit Sales 1 73,000 2 86,000 3 105,000 4 97,000 5 67,000 Additional information: ...
-
Determine the present value of the following single amounts. Note: Use tables, Excel, or a financial calculator. Round your final answers to nearest whole dollar amount. (FV of $1. PV of $1. FVA of...
-
1. Issued common stock for $29,800 cash. 2. Obtained a bank loan for $8,700 by issuing a note payable. 3. Paid $13,600 cash to buy equipment. 4. Paid $1,500 cash for April office rent. 5. Paid $1,800...
-
Max Laboratories Inc. has been operating for over thirty years producing medications and food for pets and farm animals. Due to new growth opportunities they are interested in your expert opinion on...
-
Can you elaborate on the role of goal setting theory and expectancy theory in shaping performance management processes, examining how clear, challenging goals and performance incentives can motivate...
-
What is FP's total current taxes payable and current tax expense/(benefit) for 2020? What is FP's DTA/DTL (federal and state) and deferred tax expense/(benefit) for 2020? What is FP's total income...
-
6. Find the bearing stress on the side of the bolts at each plate thickness. The applied force is 10.2 kN. (Marked) F F 10 mm 10 mm 12-mm dia. typical 11 11 11 11 (a) Top view (h) Side view 50 mm 15...
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
Determine the partial negative charge on the oxygen atom in a C=O bond. The bond length is 1.22 * and the bond dipole moment is 2.30 D.
-
What m zvalue is most likely for the base peak in the mass spectrum of 3-methylpentane?
-
What would be the results of the experiment in Problem 39 if radioactive AMP were added to the incubation mixture instead of radioactive pyrophosphate?
-
The auditor's report may be addressed to any of the following except the client's a. Stockholders. b. Board of directors. c. Chief executive officer. d. Partners.
-
The standard audit report includes all of the following except a (an) a. Opinion paragraph. b. Scope paragraph. c. Explanatory paragraph. d. Opening paragraph.
-
The existence of audit risk is recognized by the statement in the auditor's standard report that the a. Auditor is responsible for expressing an opinion on the financial statements, which are the...

Study smarter with the SolutionInn App


