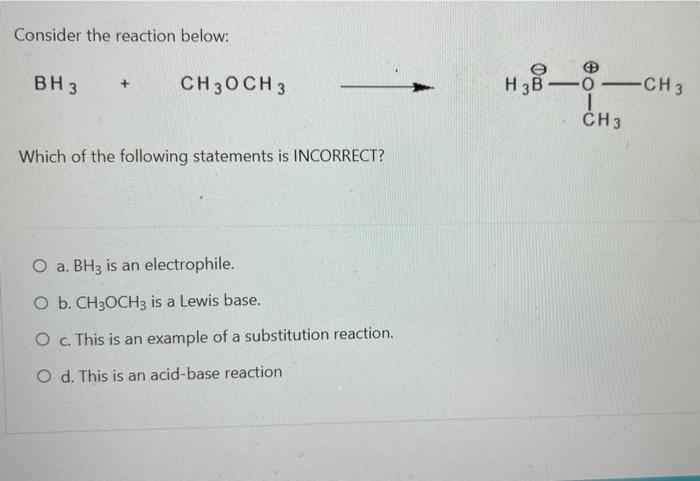
Consider the reaction below: BH 3 + CH3OCH 3 Which of the following statements is INCORRECT?...
Fantastic news! We've Found the answer you've been seeking!
Question:



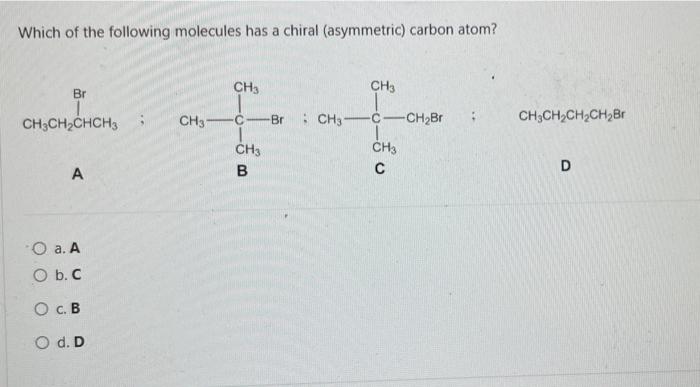

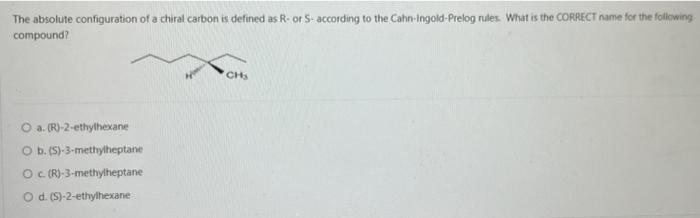
Transcribed Image Text:
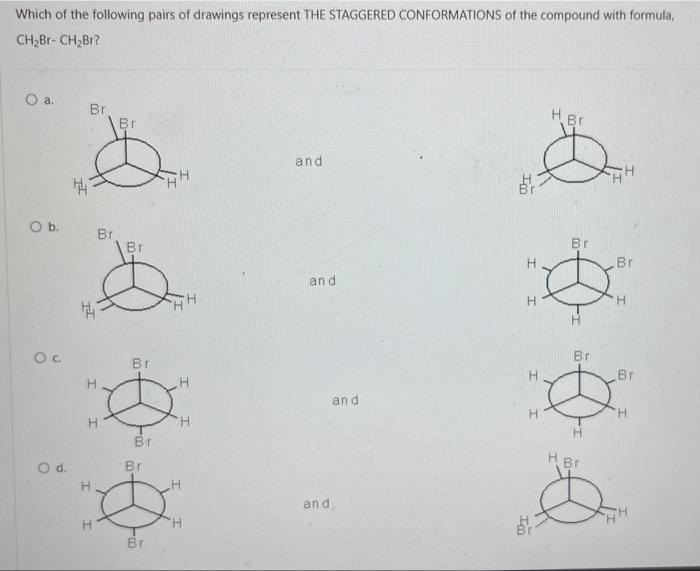
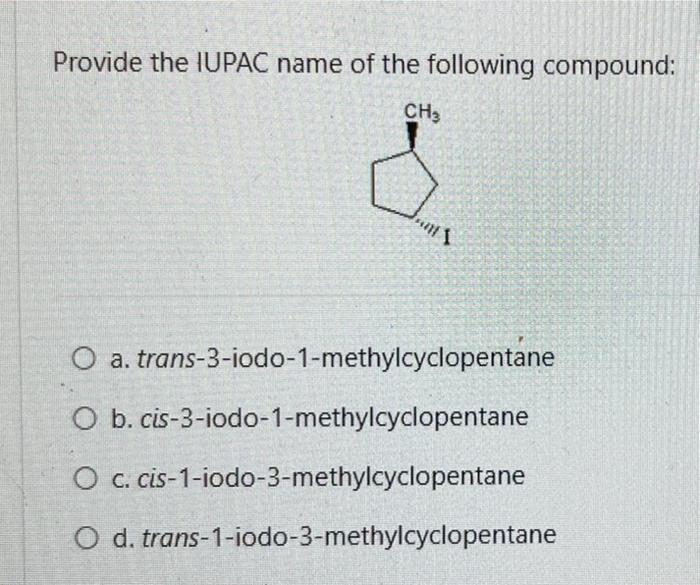
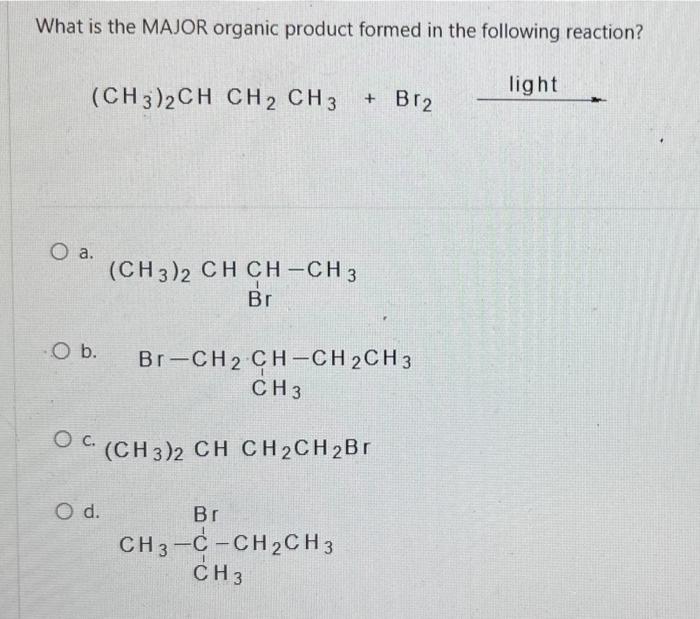
Consider the reaction below: BH 3 + CH3OCH 3 Which of the following statements is INCORRECT? O a. BH3 is an electrophile. O b. CH3OCH3 is a Lewis base. O c. This is an example of a substitution reaction. O d. This is an acid-base reaction H₂8-6. -0-CH3 I CH 3 Which of the following pairs of drawings represent THE STAGGERED CONFORMATIONS of the compound with formula, CH₂Br- CH₂Br? O a. O b. OC O d. 44 Br 14 H H Br H Br Br -00 Br Br -00 Em Br -10 Br H and. and and and Im 100 H H.Br Br Br HBr Br Br H Provide the IUPAC name of the following compound: CH3 mt\ O a. trans-3-iodo-1-methylcyclopentane O b. cis-3-iodo-1-methylcyclopentane O c. cis-1-iodo-3-methylcyclopentane O d. trans-1-iodo-3-methylcyclopentane The reaction of bromine with propane in the presence of high heat or uv light gives primarily 2-bromopropane as the reaction product because O a. secondary carbon free radicals are more stable than primary carbon free radicals O b. bromine reacts with any alkane to produce predominantly a secondary carbon free radical intermediate. O c. propane easily forms free radicals O d. bromine forms reactive free radicals. What is the MAJOR organic product formed in the following reaction? light (CH3)2CH CH2 CH3 + Br2 O a. O b. O C. O d. (CH3)2 CH CH-CH3 Br Br-CH₂ CH-CH2CH3 CH 3 (CH3)2 CH CH2CH2Br Br CH3-C-CH₂CH3 CH3 Which of the following statements are INCORRECT? O a. Free radicals contain an unpaired electron. O b. Carbocations are acidic. O c. Carbanions are strongly acidic. O d. Carbocations are electrophilic. Consider the carbocations shown below and identify the CORRECT statement. CH₂CHCH A CH₂CH-CH-CHCH₂ B (CH₂CH₂ С O a. C is the MOST stable carbocation because C is a tertiary carbocation and A is a secondary carbocation. O b. The MOST stable carbocation is B because B is a primary carbocation, which is more stable than the secondary and tertiary carbocations. O c. Carbocations, A and C, have electron-withdrawing groups which make the carbocations unstable. O d. The MOST stable carbocation is B because the carbocation, B, is stabilized by resonance delocalization which is absent in the other carbocations. Which of the following molecules has a chiral (asymmetric) carbon atom? Br CH3CH₂CHCH3 A O a. A O b. C O c. B O d. D CH31 CH3 CH3 B Br : CH3- - CH3 C CH3 o -CH₂Br CH3CH₂CH₂CH₂Br D Consider the following molecule: HAD CH3CH₂ CH₂CH₂CH3 OH Which statement is INCORRECT? O a. With the lowest (fourth) priority facing away from the viewer, a curved arrow drawn from the first (highest) priority group through the second priority group to third priority group points in a counterclockwise direction. O b. The highest priority group is OH and the lowest priority group is H. O c. The molecule has (S)- configuration. O d. The molecule has (R)- configuration. The absolute configuration of a chiral carbon is defined as R- or S- according to the Cahn-Ingold-Prelog rules. What is the CORRECT name for the following compound? O a. (R)-2-ethylhexane O b. (S)-3-methylheptane O c. (R)-3-methylheptane O d. (S)-2-ethylhexane Consider the reaction below: BH 3 + CH3OCH 3 Which of the following statements is INCORRECT? O a. BH3 is an electrophile. O b. CH3OCH3 is a Lewis base. O c. This is an example of a substitution reaction. O d. This is an acid-base reaction H₂8-6. -0-CH3 I CH 3 Which of the following pairs of drawings represent THE STAGGERED CONFORMATIONS of the compound with formula, CH₂Br- CH₂Br? O a. O b. OC O d. 44 Br 14 H H Br H Br Br -00 Br Br -00 Em Br -10 Br H and. and and and Im 100 H H.Br Br Br HBr Br Br H Provide the IUPAC name of the following compound: CH3 mt\ O a. trans-3-iodo-1-methylcyclopentane O b. cis-3-iodo-1-methylcyclopentane O c. cis-1-iodo-3-methylcyclopentane O d. trans-1-iodo-3-methylcyclopentane The reaction of bromine with propane in the presence of high heat or uv light gives primarily 2-bromopropane as the reaction product because O a. secondary carbon free radicals are more stable than primary carbon free radicals O b. bromine reacts with any alkane to produce predominantly a secondary carbon free radical intermediate. O c. propane easily forms free radicals O d. bromine forms reactive free radicals. What is the MAJOR organic product formed in the following reaction? light (CH3)2CH CH2 CH3 + Br2 O a. O b. O C. O d. (CH3)2 CH CH-CH3 Br Br-CH₂ CH-CH2CH3 CH 3 (CH3)2 CH CH2CH2Br Br CH3-C-CH₂CH3 CH3 Which of the following statements are INCORRECT? O a. Free radicals contain an unpaired electron. O b. Carbocations are acidic. O c. Carbanions are strongly acidic. O d. Carbocations are electrophilic. Consider the carbocations shown below and identify the CORRECT statement. CH₂CHCH A CH₂CH-CH-CHCH₂ B (CH₂CH₂ С O a. C is the MOST stable carbocation because C is a tertiary carbocation and A is a secondary carbocation. O b. The MOST stable carbocation is B because B is a primary carbocation, which is more stable than the secondary and tertiary carbocations. O c. Carbocations, A and C, have electron-withdrawing groups which make the carbocations unstable. O d. The MOST stable carbocation is B because the carbocation, B, is stabilized by resonance delocalization which is absent in the other carbocations. Which of the following molecules has a chiral (asymmetric) carbon atom? Br CH3CH₂CHCH3 A O a. A O b. C O c. B O d. D CH31 CH3 CH3 B Br : CH3- - CH3 C CH3 o -CH₂Br CH3CH₂CH₂CH₂Br D Consider the following molecule: HAD CH3CH₂ CH₂CH₂CH3 OH Which statement is INCORRECT? O a. With the lowest (fourth) priority facing away from the viewer, a curved arrow drawn from the first (highest) priority group through the second priority group to third priority group points in a counterclockwise direction. O b. The highest priority group is OH and the lowest priority group is H. O c. The molecule has (S)- configuration. O d. The molecule has (R)- configuration. The absolute configuration of a chiral carbon is defined as R- or S- according to the Cahn-Ingold-Prelog rules. What is the CORRECT name for the following compound? O a. (R)-2-ethylhexane O b. (S)-3-methylheptane O c. (R)-3-methylheptane O d. (S)-2-ethylhexane
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Lets break down each option and give a detailed explanation for each option ONE BH3 is an ... View the full answer

Related Book For 

An Introduction to Statistical Methods and Data Analysis
ISBN: 978-1305269477
7th edition
Authors: R. Lyman Ott, Micheal T. Longnecker
Posted Date:
Students also viewed these chemistry questions
-
20 21 22 23 24 11 14 15 7 8 9 10 13 4 6 2 3 5 ABC Co. manufactures widgets and has the following operating information for the past year: A A 12 B B Product Line Product Line Budgeted Unit Sales...
-
Find the 50th percentile for the data above. Use the rule given inthe text/notes. 20 21 21 21 22 22 24 25 26 29 29 32 32 32 33 33 34 35 36 37 37 38 39 40 41 41 43 43 45 46 50 52 52 53 57 58 59 59 60...
-
x 20 25 27 27 23 29 30 26 y 20 24 25 29 20 29 32 29
-
The two tugboats each exert a constant force F on the boat such that these forces are always directed perpendicular to the boats centerline. If the boat has a mass m and a radius of gyration k G...
-
In our Nodhead example, true depreciation was decelerated. That is not always the case. For instance, Table 12.6 shows how on average the market value of a Boeing 737 has varied with its age 27 and...
-
Prince Charles Island Company has expected sales of $6,000 in September, $10,000 in October, $16,000 in November, and $12,000 in December. Cash sales are 20 percent and credit sales are 80 percent of...
-
Granger Technology Solutions, Inc., completed the following transactions during January 2011, its first month of operations: Requirement 1. Record the preceding transactions in the journal of Granger...
-
Subsidiary Company S had the following stockholders equity on January 1, 2014, prior to issuing 20,000 additional new shares to non-controlling shareholders: Common stock ($1par), 100,000 shares...
-
Describe how standard costs are determined by a manufacturing company.Describe 2 types of standard cost variances that are used by management to assess both the efficiency and effectiveness of...
-
The model with all useable variables is? Round to 2 decimal places If an answer is 0 (Zero) enter 0.00 4 Price 25099.34 + A A/ * SqFt + Thatched Roof + 46 a
-
. Quartz Sdn Bhd is specializing in producing alarm clock. The company is currently producing and selling one type of alarm clock known as Princess Frozen Clock. The costs and revenues of Princess...
-
Find an analytic stuxction whose reas parte. SinLy Coshly- COS2x.
-
3. ZR Corporation's stock has a beta coefficient equal to 0.8 and a required rate of return equal to 11 percent. If the expected rate of return on the market is 12.5 percent, what is the risk-free...
-
Atlee Inc. wants to determine the average collection period ratio for the last fiscal from the information provided: Accounts Receivables (Jan 01, 2020): $43,000 / Accounts Receivables (Dec 31,...
-
Decrypt the Cipher Text "THTIPPNTOYENCGIRGRRSEYAIS" using the key=456213 with Columnar Transposition Technique This is a Cryptography and network security subject?
-
Sethuraman Inc has the following periods of high and low costs and units produced during the period. Sethuraman Costs and Units Produced Month of Activity Units Produced Total Cost August 40,000...
-
a) Derive or state the gate-voltage relationship of the MOS capacity. b) In inversion we find two charge types on the semiconductor side, describe their origin. Which ones are fixed, which ones are...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
The public safety department at a large urban university was concerned about criminal activities involving nonstudents stealing bicycles and laptops from students. The campus police designed a study...
-
A chain of small convenience food stores performs a regression analysis to explain variation in sales volume among 16 stores. The variables in the study are as follows: Sales: Average daily...
-
Refer to Exercise 10.40. Suppose the recruitment director is preparing a presentation for upper management to recommend new hiring practices of the firm. a. Provide a graph of the data in Exercise...
-
What is the File Drawer Problem?
-
What is evidence-based medicine?
-
What is a forest plot?

Study smarter with the SolutionInn App


