Choose an explanation(s) for the formation of a racemic mixture. Both ends of the forming ring...
Fantastic news! We've Found the answer you've been seeking!
Question:

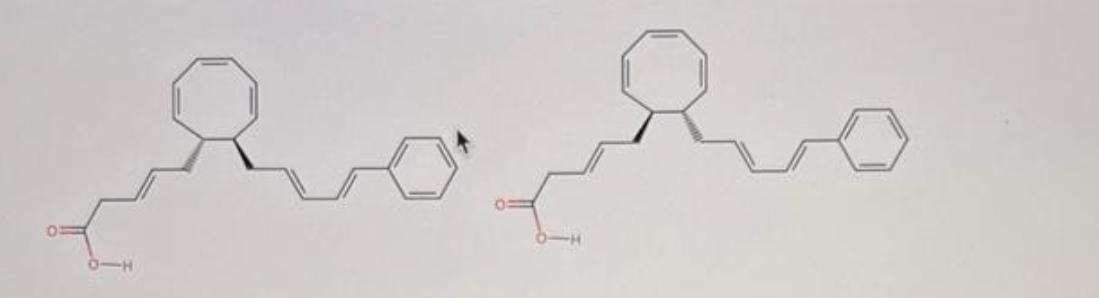
Transcribed Image Text:
Choose an explanation(s) for the formation of a racemic mixture. Both ends of the forming ring can rotate counter-clockwise. The compound has plane of symmetry. The compound undergoes disrotatory ring closure under thermal conditions. The compound undergoes ring closure to form cis configuration. Both ends of the forming ring can rotate clockwise. The compound undergoes conrotatory ring closure under thermal conditions. Choose an explanation(s) for the formation of a racemic mixture. Both ends of the forming ring can rotate counter-clockwise. The compound has plane of symmetry. The compound undergoes disrotatory ring closure under thermal conditions. The compound undergoes ring closure to form cis configuration. Both ends of the forming ring can rotate clockwise. The compound undergoes conrotatory ring closure under thermal conditions.
Expert Answer:
Answer rating: 100% (QA)
Racemic mixtures are often formed when achiral substanc... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The equilibrium constant (KP) for the formation of the air pollutant nitric oxide (NO) in an automobile engine at 530°C is 2.9 Ã 10-11: (a) Calculate the partial pressure of NO under these...
-
A proposed mechanism for the formation of N 2 O 5 (g) from NO 2 (g) and O 3 (g) is Determine the rate law expression for the production of N 2 O 5 (g) given this mechanism. NO,(g) + O3(g)1...
-
The standard free energy change for the formation of H 2 (g) + O 2 (g) from H 2 O is G = + 237.13 kJ. The reactions are Calculate the standard voltage (E) needed to decompose water into its elements...
-
Question 3: Assume that Narine Inc. is considering leasing a car from Proctor Inc. for a period of four years. The fair value of the car today is $36,000. The current market interest rate for...
-
Show that n i=1 n X i is sufficient for . Also show that X-bar is sufficient for .
-
Payments under some retirement plans are based on the average earnings in the last few years of employment. Discuss the potential incentive effects of this policy.
-
Which of the following statements is false? (a) A negative cash flow can occur in a year in which net income is positive. (b) An increase in accounts receivable represents accounts not yet collected...
-
The adjusted trial balance of Big Papi Music Company at June 30, 2012, follows: Requirements 1. Journalize Big Papis closing entries. 2. Prepare Big Papis single-step income statement for the year....
-
How much should investors pay for a 6%, semiannual coupon bond of Ford Motor Company? The bond has $1,000 par value and 19 years remaining to maturity. Investor's required return is 13%. The bond...
-
The Case Study - "Lemonade: Delighting Insurance Customers with AI and Behavioral Economics". 1. How do you explain the phenomenal growth experienced by Lemonade? 2. Does Lemonade create a unique...
-
The driver of a car travelling with speed 30 m/sec towards a hill sounds a horn of frequency 600 Hz. If the velocity of sound in air is 330 m/s, the frequency of reflected sound as heard by driver is...
-
What do you see as the greatest specific cost and benefit of globalization?
-
The giant Asian pond turtle is endangered. It is also highly sought after for medicinal and food purposes. Would you hunt and kill one of these turtles for $20? For $10,000? At what price would you...
-
What is naturalism? Is naturalism associated exclusively with an interpretive approach to research? Explain why or why not.
-
What criteria do you most often use when faced with an ethical dilemma? Which of the theories described in this chapter is the closest to your personal theory? Do you think any of the new ideas you...
-
Explain why GDP is not a good measure of social welfare.
-
Assignment Setting Provide a monetary policy recommendation based on a description of the state of the economy provided below and to explain how their policy recommendation will affect the economy....
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
Is the addition of Br2 to an alkene such as trans-2-pentene a stereoselective reaction? Is it a stereospecific reaction? Is it an enantioselective reaction?
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. CH CH3 CH CH H2S04 CH,CH CHCH CH ,504 CH CH-CCH CH3 OH ' CH3O CH3OH 1. TsCl/pyridine 2. NaCEN CH3 OH (CH2CH2CH2hCuli CI
-
a. Name the kind of sigmatropic rearrangement that occurs in each of the following reactions. b. Using arrows show the electron rearrangement that takes place in each of the reactions. 1. 2. 3. 4....
-
A sample contains radioactive atoms of two types, A and B. Initially there are five times as many A atoms as there are B atoms. Two hours later, the numbers of the two atoms are equal. The half-life...
-
The technique known as potassiumargon dating is used to date volcanic rock and ash, and thus establish dates for nearby fossils, like this 1.8 -millionyear-old hominid skull. The potassium isotope...
-
You are assisting in an anthropology lab over the summer by carrying out \({ }^{14} \mathrm{C}\) dating. \(\mathrm{A}\) graduate student found a bone he believes to be 20,000 years old. You extract...

Study smarter with the SolutionInn App


