17) If I wish to travel 88 mph (miles per hour) in a DeLorean. How fast...
Fantastic news! We've Found the answer you've been seeking!
Question:
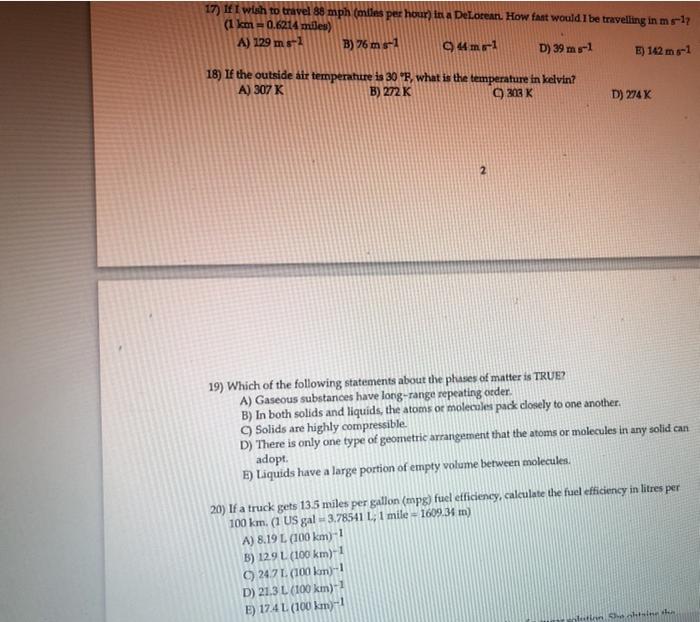
Transcribed Image Text:
17) If I wish to travel 88 mph (miles per hour) in a DeLorean. How fast would I be travelling in m s-17 (1 km-0.6214 miles) A) 129 m 8-1 B) 76 m s-1 C44ma-1 D) 39 m 8-1 E) 142 m 8-1 18) If the outside air temperature is 30 °F, what is the temperature in kelvin? A) 307 K B) 272 K C) 303 K 2 19) Which of the following statements about the phases of matter is TRUE? A) Gaseous substances have long-range repeating order. D) 274 K B) In both solids and liquids, the atoms or molecules pack closely to one another. C) Solids are highly compressible. D) There is only one type of geometric arrangement that the atoms or molecules in any solid can adopt. E) Liquids have a large portion of empty volume between molecules. B) 129 L (100 km)-1 C) 24.7 1. (100 km)-1 D) 21.3 L (100 km)-1 E) 17.4L (100 km)-1 20) If a truck gets 13.5 miles per gallon (mpg) fuel efficiency, calculate the fuel efficiency in litres per 100 km. (1 US gal = 3.78541 L, 1 mile = 1609.34 m) A) 8.19 L (100 km)-1 plution Sha chising th 17) If I wish to travel 88 mph (miles per hour) in a DeLorean. How fast would I be travelling in m s-17 (1 km-0.6214 miles) A) 129 m 8-1 B) 76 m s-1 C44ma-1 D) 39 m 8-1 E) 142 m 8-1 18) If the outside air temperature is 30 °F, what is the temperature in kelvin? A) 307 K B) 272 K C) 303 K 2 19) Which of the following statements about the phases of matter is TRUE? A) Gaseous substances have long-range repeating order. D) 274 K B) In both solids and liquids, the atoms or molecules pack closely to one another. C) Solids are highly compressible. D) There is only one type of geometric arrangement that the atoms or molecules in any solid can adopt. E) Liquids have a large portion of empty volume between molecules. B) 129 L (100 km)-1 C) 24.7 1. (100 km)-1 D) 21.3 L (100 km)-1 E) 17.4L (100 km)-1 20) If a truck gets 13.5 miles per gallon (mpg) fuel efficiency, calculate the fuel efficiency in litres per 100 km. (1 US gal = 3.78541 L, 1 mile = 1609.34 m) A) 8.19 L (100 km)-1 plution Sha chising th
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below 17 A 129 m s1 Explanation 88 mph ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements about the project budgeting process is true? The weekly budget is formed by only using the early start times of each activity. The weekly budget is formed by only...
-
Which of the following statements about the online newspaper audience is not true? A) Online newspaper readership is growing at more than 10% a year. B) The majority of newspaper readers read the...
-
Which of the following statements about the minimum variance portfolio of all risky securities are valid? (Assume short sales are allowed.) Explain. a. Its variance must be lower than those of all...
-
Glencoe First National Bank operated for years under the assumption that profitability can be increased by increasing dollar volumes. Historically, First Nationals efforts were directed toward...
-
It has been about 20 years since Julia graduated with a major in aeronautical engineering, and she has been quite successful in her career and her personal finances. Accordingly she wants to sell her...
-
Three long, parallel wires are arranged as shown in Fig. 5-43 (P5.18 (a)). Determine the force per unit length acting on the wire carrying I3. O 4 = 10A 2m I3 = 10A 2m- 2m 1 = 10A Figure P5.18: (a)...
-
When the Concepcions went to AT&T to get a phone, they were confused as to why they were paying money for the phone when it was advertised as being free. They had to pay sales tax on the full normal...
-
Compute FIFO, LIFO, Average Cost?Periodic Presented below is information related to radios for the Couples Company for the month of July. (a) Assuming that the periodic inventory method is used,...
-
Tl-208 decays through a beta emission and has a half-life of 3.05 minutes. Besides the beta emission, there are several gamma rays associated with the decay and these gamma emissions occur at...
-
Recently Santa Fe, New Mexico, was named one of the safest places to live in the United States. Since then, housing development projects have been springing up all around Santa Fe. Six housing...
-
The 20-point signal r(n) is given by x(n) = sin 2n 0
-
The Smiths want to qualify for a mortgage loan with a large local bank. The bank mortgage loan officer says that they must have a Housing Ratio #1 of less than or equal to 28% for the loan to made....
-
If a business, for example, requires tenants to make deposits that could mistakenly be recorded as revenue, which liability account should get examined further for potential fraud?
-
Explain how the identified hazards were identified as being the hazards that must be addressed in an Airport Emergency Plan (AEP)
-
How should Walgreens enter international markets with the use of LICENSING as its choice of entry strategy? Explain why LICENSING could be the best entry strategy for Walgreens to enter the...
-
Caroline has $3,000 in taxable interest, $6,000 in taxable dividends, and $1,000 in tax-exempt interest. She has paid a total of $1,000 for investment-related seminars. What is the amount she can...
-
Calculate the depletion expense for 2019. Calculate the total depreciation expense for 2019. Calculate the interest expense for 2019. On January 1, 2018, Micah Joy Mining Corp. acquired property...
-
Which property determines whether a control is available to the user during run time? a. Available b. Enabled c. Unavailable d. Disabled
-
What percentage of the CIs include the parameter p?
-
What is the p-value corresponding to your answer to Problem 8.16? Nutrition The mean 1 sd of ln [calcium intake (mg)] among 25 females, 12 to 14 years of age, below the poverty level is 6.56 0.64....
-
What type of study is this? Hypertension Suppose that 200 obese (BMI 25) children and 500 normal-weight (BMI <25) children are identified in a school-based screening for hypertension. Eighteen of the...
-
How might planning in a not-for-profit organization such as the World Wildlife Fund differ from planning in a for-profit organization such as Airbnb?
-
Provide examples of the sources of data a residential solar panel company might gather when engaging in environmental scanning. Exhibit 8-6 may be helpful when answering this question.
-
What advantages and disadvantages does Johnson Controls OpenBlue platform have over companies looking to develop solutions on their own?

Study smarter with the SolutionInn App


