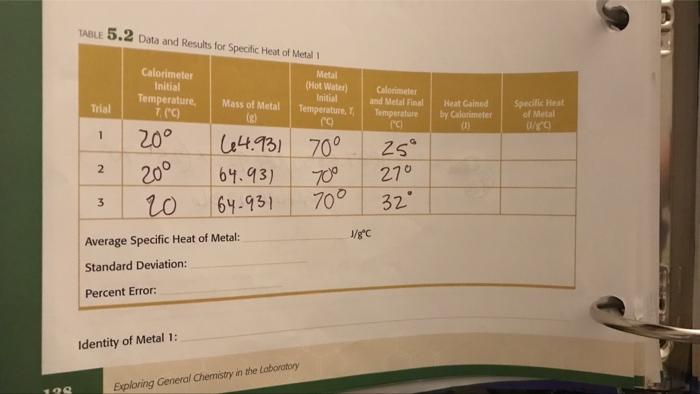
199 TABLE 5.2 Data and Results for Specific Heat of Metal 1 Metal (Hot Water) Initial...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
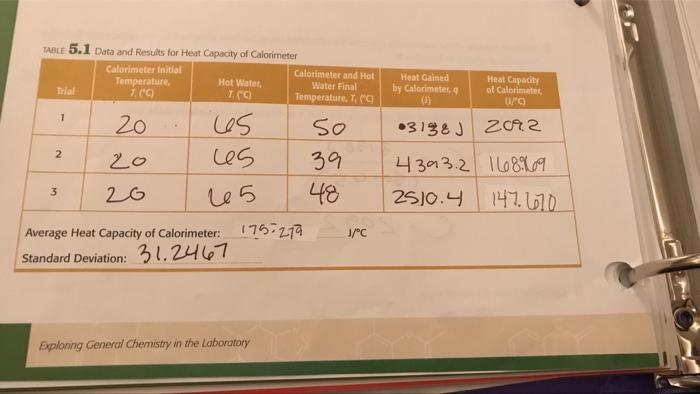
199 TABLE 5.2 Data and Results for Specific Heat of Metal 1 Metal (Hot Water) Initial Temperature, T E 64.931 70° 70° Trial 1 2 3 Calorimeter Initial Temperature, 7. (C) 20° 20⁰ 20 Mass of Metal (2) Identity of Metal 1: 64.93) 64-931 Average Specific Heat of Metal: Standard Deviation: Percent Error: Exploring General Chemistry in the Laboratory Calorimeter and Metal Final Temperature (c) J/gºC 25° 270 32° Heat Gained by Calorimeter B Specific Heat of Metal 0/80 TABLE 5.1 Data and Results for Heat Capacity of Calorimeter Calorimeter Initial Temperature, 7. (C) 20 Trial 1 2 3 20 20 Hot Water, TCC) los 65 25 Average Heat Capacity of Calorimeter: Standard Deviation: 31.2467 Calorimeter and Hot Water Final Temperature. T, ("C 50 39 48 175-279 Exploring General Chemistry in the Laboratory J/°C Heat Gained by Calorimeter, q (1) 03138 J Heat Capacity of Calorimeter, 0/0) 2092 4393.2 168969 2510.4 147.670 199 TABLE 5.2 Data and Results for Specific Heat of Metal 1 Metal (Hot Water) Initial Temperature, T E 64.931 70° 70° Trial 1 2 3 Calorimeter Initial Temperature, 7. (C) 20° 20⁰ 20 Mass of Metal (2) Identity of Metal 1: 64.93) 64-931 Average Specific Heat of Metal: Standard Deviation: Percent Error: Exploring General Chemistry in the Laboratory Calorimeter and Metal Final Temperature (c) J/gºC 25° 270 32° Heat Gained by Calorimeter B Specific Heat of Metal 0/80 TABLE 5.1 Data and Results for Heat Capacity of Calorimeter Calorimeter Initial Temperature, 7. (C) 20 Trial 1 2 3 20 20 Hot Water, TCC) los 65 25 Average Heat Capacity of Calorimeter: Standard Deviation: 31.2467 Calorimeter and Hot Water Final Temperature. T, ("C 50 39 48 175-279 Exploring General Chemistry in the Laboratory J/°C Heat Gained by Calorimeter, q (1) 03138 J Heat Capacity of Calorimeter, 0/0) 2092 4393.2 168969 2510.4 147.670
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Water of mass m = 20 g is enclosed in a thermally insulated cylinder at the temperature of 0 C under a weightless piston whose area is S = 410 cm2 . The outside pressure is equal to standard...
-
The specific heat of copper metal was determined by putting a piece of the metal weighing 35.4 g in hot water. The quantity of heat absorbed by the metal was calculated to be 47.0 J from the...
-
For A = 3i + j k, B = i + 2j + 5k, and C = 2j 3k, find C (A B).
-
Let U = {1, 2, 3, 4,p, 10} A = {1, 3, 5, 7} B = {3, 7, 9, 10} C = {1, 7, 10} Determine the following. A - B
-
Suppose the United States decides to subsidize the export of U.S. agricultural products, but it does not increase taxes or decrease any other government spending to offset this expenditure. Using a...
-
Create and test an HTML document that describes an ordered list with the following contents: The highest level should be the names of your parents, with your mother first. Under each parent, you must...
-
On September 14, 2010, C & T Machinery, Inc., sold $2,300 of inventory (cost is $1,350) on account to one of its customers. The terms were 1/10, n/30, FOB destination. On September 16, C & T...
-
Rhett purchased a 13% zero-coupon bond with a 15-year maturity and a $20,000 par value 15 years ago. The bond matures tomorrow. How much will Rhett receive in total from this investment, assuming all...
-
XY Ltd currently extends a credit period of 30 days to its debtors. It intends to change the credit period t0 3/15 net 45 in order to increase sales. The proposed change in credit terms will have the...
-
Using Fig. 4.102 , design a problem that will help other students better understand Thevenin equivalent circuits. Find the Thevenin equivalent at terminals a-b of the circuit in Fig. 4.102. R3 R, R2...
-
Technology for the public sector for now and beyond: Governments and the Megatrend Enabling Technology A major focal point on the digital agenda of governments besides infrastructure is data, open...
-
Hasan receives an invoice for $103.50 dated September 30, with terms 5/10, 2/30, n/60. He decides to pay it in full on October 25. What is the total amount paid to settle the account? ( Answer to the...
-
1. What are some of the most common forms of unethical behavior in our workforce today? How could leadership in organizations help to minimize this ethical misconduct? Explain and support your...
-
1. What kinds of industries or companies do you think would benefit most from using activity-based costing and why? 2. In designing or modifying an accounting system to capture appropriate costs for...
-
A bullet whose mass is 5.88g moves at a speed of 677m/s. a. What is the kinetic energy, in joules, of the bullet? b. What would be the kinetic energy, in joules, if the bullet were to instead move at...
-
Jose Ruiz manages a construction rm's equipment repair... Jose Ruiz manages a construction firm's equipment repair department. His department is a cost center. Costs for a recent period follow. Jose...
-
1 . Calculate the acceleration of a car, if its speed changes from 0 0 m / / s ( at rest ) to 3 0 m / / s in 6 seconds. 2. A rock falls of a cliff. What is the acceleration due to gravity if the...
-
Modify the counter from Exercise 5.44 such that the counter will either increment by 4 or load a new 32-bit value, D, on each clock edge, depending on a control signal Load. When Load = 1, the...
-
Bev and Ken Hair have been married for 3 years. They live at 3567 River Street, Springfield, MO 63126. Ken is a full-time student at Southwest Missouri State University (SMSU) and Bev works as an...
-
Mark owns his home and has a $250,000 mortgage related to his purchase of the residence. When his daughter went to college in the fall of 2012, he borrowed $20,000 through a home equity loan on his...
-
Dr. George E. Beeper is a single taxpayer. He lives at 45 Mountain View Dr., Apt. 321, Spokane, WA 99210. Dr. Beeper's Social Security number is 775-88-9531. Dr. Beeper works for the Pine Medical...
-
Let $S^{2}$ denote a sphere of unit radius, centered at the origin of $\mathbb{R}^{3}$. What is the group of transformations that leave it invariant? Next, let $S_{(1)}^{2}$ be a sphere of unit...
-
Prove that the order of the symmetric group of a set of $N$ elements, $S_{N}$, is $N$ !.
-
Let $S_{(2)}^{2}$ denote a sphere of unit radius, centered at the origin of threedimensional space, with two noncoincident points on its surface. What is the group of transformations that leave...

Study smarter with the SolutionInn App


