1.Calculate the formula mass of each compound. Keep at least one decimal place in atomic masses...
Fantastic news! We've Found the answer you've been seeking!
Question:

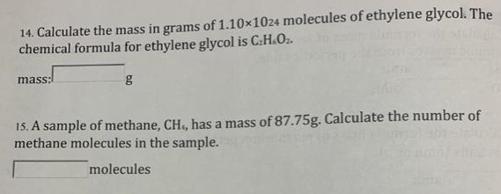
Transcribed Image Text:
1.Calculate the formula mass of each compound. Keep at least one decimal place in atomic masses from the periodic table. H²S: amu HFO²:[ 2. Calculate the formula mass of magnesium perbromate, Mg(Br0+)2, in atomic mass units (amu or u). amu amu 3. Calculate the percent composition by mass of each element in LICIO4. % Li= % Cl= % 0= 4. How many grams of Br are in 285g CaBrz? 33.3g Ag= 33.3g Cr203 % % % g 8. Suppose you have two samples that are equal in weight, 33.3g Ag and 33.3g Cr203. Calculate the number of moles of each substance. mol mol 9.How many grams of potassium bromite (KBrO.) are there in 0.495mol? 0.495 mol= g 10. Convert 1.25x1024 atoms of carbon to moles of carbon. 1.25x1024 atoms= mol C 11. What is the number of formula units in a 1.00mol sample of CaO? formula units 14. Calculate the mass in grams of 1.10×1024 molecules of ethylene glycol. The chemical formula for ethylene glycol is C.H.O₂. mass: g 15. A sample of methane, CH., has a mass of 87.75g. Calculate the number of methane molecules in the sample. molecules 1.Calculate the formula mass of each compound. Keep at least one decimal place in atomic masses from the periodic table. H²S: amu HFO²:[ 2. Calculate the formula mass of magnesium perbromate, Mg(Br0+)2, in atomic mass units (amu or u). amu amu 3. Calculate the percent composition by mass of each element in LICIO4. % Li= % Cl= % 0= 4. How many grams of Br are in 285g CaBrz? 33.3g Ag= 33.3g Cr203 % % % g 8. Suppose you have two samples that are equal in weight, 33.3g Ag and 33.3g Cr203. Calculate the number of moles of each substance. mol mol 9.How many grams of potassium bromite (KBrO.) are there in 0.495mol? 0.495 mol= g 10. Convert 1.25x1024 atoms of carbon to moles of carbon. 1.25x1024 atoms= mol C 11. What is the number of formula units in a 1.00mol sample of CaO? formula units 14. Calculate the mass in grams of 1.10×1024 molecules of ethylene glycol. The chemical formula for ethylene glycol is C.H.O₂. mass: g 15. A sample of methane, CH., has a mass of 87.75g. Calculate the number of methane molecules in the sample. molecules
Expert Answer:
Answer rating: 100% (QA)
Ill solve the questions one by one First lets start with question 1 Calculate the formula mass of each compound Keep at least one decimal in atomic masses from the periodic table a HS b HS c HFO To ca... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Suppose you have two samples that are equal in weight, 37.8 g Sr and 37.8 g Al2O3 . Calculate the number of moles of each substance. 37.8 g Sr= 37.8 g Al2O3=
-
Calculate the formula mass of the following molecules to three significant figures. P CI (a) (b)
-
Calculate the formula mass of the following molecules to three significant figures. N' ( (b) (a)
-
If the current spot rate is 2.10 (HC/FC) and the government undertakes policies to depreciate the currency 16%, what is the new spot rate? Note: 2.10 * 1.16 is not the right formula. Based on this,...
-
Rally Furniture started and finished Job 310 during August. The companys records show that the following direct materials were requisitioned for Job 310: Lumber: 51 units at $ 9 per unit Padding: 15...
-
The file P08_06.xlsx contains data on repetitive task times for each of two workers. John has been doing this task for months, whereas Fred has just started. Each time listed is the time (in seconds)...
-
The Glendora Company has 200,000 shares of cumulative, five percent, \(\$ 100\) par value preferred stock outstanding. Last year the company failed to pay its regular dividend, but the board of...
-
Using capitalized cost determine which type of road surface is preferred on a particular section of highway Use 12% interest rate. A B Initial cost $700,000 $500,000 Annual maintenance Periodic...
-
https://amazonfctours.com/virtualtours(Links to an external site.) Write one-page report on what you learned? How do they use technology in the warehouse?
-
A rotating step shaft is loaded as shown, where the forces F A and FB are constant at 600 lbf and 300 lbf, respectively, and the torque T alternates from 0 to 1800 lbf in. The shaft is to be...
-
Mr. Aranjuez bought a flat screen TV on credit. They ask for a down payment of $5,300.00 and you have a balance of $21,200.00 left to cover with 12 payments of $2,200.00 each. What is the annual...
-
Two students are named to present their work from a class of 12 students: 4 from electrical department, 5 from telecommunication department and 3 from computer department. Let X denote the number of...
-
A small town in S. America had been plagued, thousands of people were dying of yellow fever. The task was to find the cause and eradicate it. Two approaches used were: Microscopic research on the...
-
How do effective change management strategies mitigate resistance and foster buy-in among stakeholders, ensuring smooth transitions and minimizing disruptions to established workflows and routines ?
-
i. Suggest at least THREE (3) kinds of analytics and machine learning algorithms that Champo Carpets can use to solve their problems, and in general, for value creation. ii. Explain the data strategy...
-
In your program, add comments that describe in English what your program does. Begin by writing your header and main program. Declare your variables, define their type, and use them. Make a list of...
-
Competitive advantages of tata group based on global level following these: Cost Leadership Low-cost structure Reduce associated risks and capital requirements in foreign markets. Higher bargaining...
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
The voltage needed to produce a current of 5 A in a resistance of 40 is a. 0.125 V b. 5 V c. 8 V d. 200 V
-
How could you distinguish calcite crystals from quartz crystals?
-
Do ions in solution need activation energy to react with one another? If not, why not?
-
The accounting profession has been investigated by Congress over a number of years. However, none of the investigations or recommendations for change seems to have made a difference in that fraud...
-
The following two questions directly relate to ethics standards in the AICPA Code of Professional Conduct: a. Is it ever improper for a CPA/CPA firm to accept a commission from a client for...
-
Have you ever agreed to do something when you werent sure that you had the skills to accomplish the job? Why did you agree to do it? Can you draw an analogy between your motivations and the ethical...

Study smarter with the SolutionInn App


