Question: Hahn Company uses a job-order costing system. Its plantwide predetermined overhead rate uses direct labor-hours as the allocation base. The company pays its direct
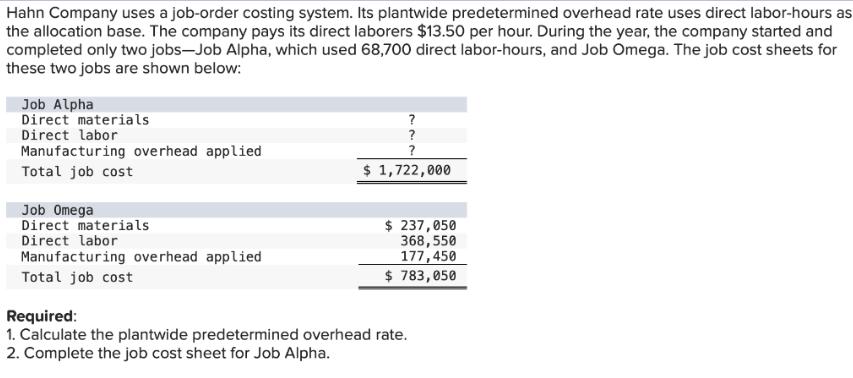
Hahn Company uses a job-order costing system. Its plantwide predetermined overhead rate uses direct labor-hours as the allocation base. The company pays its direct laborers $13.50 per hour. During the year, the company started and completed only two jobs-Job Alpha, which used 68,700 direct labor-hours, and Job Omega. The job cost sheets for these two jobs are shown below: Job Alpha Direct materials Direct labor Manufacturing overhead applied Total job cost Job Omega Direct materials Direct labor Manufacturing overhead applied Total job cost ? ? ? $ 1,722,000 $ 237,050 368,550 177,450 $ 783,050 Required: 1. Calculate the plantwide predetermined overhead rate. 2. Complete the job cost sheet for Job Alpha.
Step by Step Solution
3.45 Rating (168 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


