1ONA(s) + 2KNO, (s) K,0(3) + 5Na,0(s) +N, (g) A common laboratory-scale reaction can also...
Fantastic news! We've Found the answer you've been seeking!
Question:
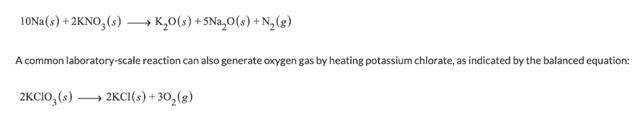
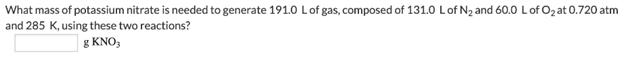

Transcribed Image Text:
1ONA(s) + 2KNO, (s) – K,0(3) + 5Na,0(s) +N, (g) A common laboratory-scale reaction can also generate oxygen gas by heating potassium chlorate, as indicated by the balanced equation: 2KCIO,() → 2KCI(s) + 30,(g) What mass of potassium nitrate is needed to generate 191.0 Lof gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? g KNO, What mass of potassium chlorate is needed to generate 191.0 L of gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? 8 KCIO, 1ONA(s) + 2KNO, (s) – K,0(3) + 5Na,0(s) +N, (g) A common laboratory-scale reaction can also generate oxygen gas by heating potassium chlorate, as indicated by the balanced equation: 2KCIO,() → 2KCI(s) + 30,(g) What mass of potassium nitrate is needed to generate 191.0 Lof gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? g KNO, What mass of potassium chlorate is needed to generate 191.0 L of gas, composed of 131.0 Lof N2 and 60.0 Lof O2 at 0.720 atm and 285 K, using these two reactions? 8 KCIO,
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Oxygen can be prepared by heating potassium chlorate, KClO3, with manganese dioxide as a catalyst. The reaction is How many moles of O2 would be obtained from 1.300 g KClO3? If this amount of O2 were...
-
The reaction N2 + O2 2NO occurs in internal combustion engines. Determine the equilibrium mole fraction of NO when the pressure is 101 kPa and the temperature is 1800 K.
-
A quantity of N2 gas originally held at 5.25 atm pressure in a 1.00-L container at 26oC is transferred to a 12.5-L container at 20oC .A quantity of O2 gas originally at 5.25 atm and 26oC in a 5.00-L...
-
For rigid-body motion, the strains will vanish. Under these conditions, integrate the straindisplacement relations (7.6.1) to show that the most general form of a rigid-body motion displacement field...
-
Repeat the preceding exercise using the sample data from the Menthol cigarettes listed in Data Set 10 from Appendix B.
-
Reconsider Problem 10.22. Use the lessons learned from the CCW case study to address the following questions. a. What might be causing the significant shifts in total sales from month to month that...
-
Consider heat transfer over a flat plate again but now include an additional term due to viscous heating. Show that the similarity method is applicable to this problem as well, and derive the...
-
Mansfield Corporation had 2013 sales of $100 million. The balance sheet items that vary directly with sales and the profit margin are as follows: Percent Cash ..............5% Accounts receivable...
-
Suggest a setup to support both file access as well as block access for a SAN with a suitable diagram also give detailed explanation of how this setup can be used.please explain in detail
-
How can I find the exact p-value for each coefficient (p>t )column if the number of observations = 52, df numerator is 3, df denominator is 48, t static is there in the third column. I need to know...
-
Fordman Companys break-even point is 8,500 units. Variable cost per unit is$140 and total fixed costs are $297,500 per year. What price does Fordman charge?
-
What are the constitutional rights of a corporation?
-
Describe the contrasting approaches brands take to brand architecture. Select one multi-brand organization and analyse its approach.
-
Research has emerged that views the targeting of particular segments as overdone. Discuss the merits and weaknesses of the sophisticated mass marketing approach of appealing to as large a slice of...
-
Soapy Inc. and Suddies Inc., the only laundry detergent producers, collude and agree to share the market equally. If neither firm cheats, each makes $1 million. If one firm cheats, it makes $1.5...
-
How does the students marginal product change? How does the value of marginal product of labor change? The market price of fish falls to 66 a pound, but the packers wage rate remains at $12.50 an...
-
Ciphers in which pairs of numbers replace each letter are called bilateral or dinome ciphers. A simple way to construct such ciphers is to put the letters of the alphabet into a rectangle and number...
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
There are 2.619 1022 atoms in 1.000 g of sodium. Assume that sodium atoms are spheres of radius 1.86 and that they are lined up side by side. How many miles in length is the line of sodium atoms?
-
A solution is 0.35 M Sr(OH)2. What are the concentrations of H3O+ and OH in this solution?
-
Two compounds have the same composition: 85.62% C and 14.38% H. a. Obtain the empirical formula corresponding to this composition. b. One of the compounds has a molecular mass of 28.03 amu; the...
-
Sketch and explain a simple diagram to match your exhibit.
-
a. Write out and explain the equation for volume breakeven. b. What is the difference between accounting breakeven and economic breakeven?
-
a. What is profit analysis, also known as cost-volume-profit (CVP) analysis? b. Why is profit analysis so useful to healthcare managers? c. What is a profit and loss (P&L) statement?

Study smarter with the SolutionInn App



