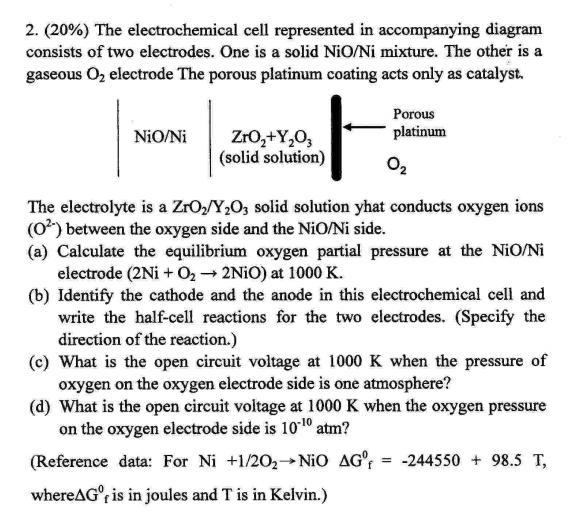
2. (20%) The electrochemical cell represented in accompanying diagram consists of two electrodes. One is a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. (20%) The electrochemical cell represented in accompanying diagram consists of two electrodes. One is a solid NiO/Ni mixture. The other is a gaseous Oz electrode The porous platinum coating acts only as catalyst. Porous NiO/Ni platinum Zro,+Y,0, (solid solution) O2 The electrolyte is a ZrO/Y;O; solid solution yhat conducts oxygen ions (0) between the oxygen side and the NiO/Ni side. (a) Calculate the equilibrium oxygen partial pressure at the NiO/Ni electrode (2Ni + O, 2NIO) at 1000 K. (b) Identify the cathode and the anode in this electrochemical cell and write the half-cell reactions for the two electrodes. (Specify the direction of the reaction.) (c) What is the open circuit voltage at 1000 K when the pressure of oxygen on the oxygen electrode side is one atmosphere? (d) What is the open circuit voltage at 1000 K when the oxygen pressure on the oxygen electrode side is 1010 atm? (Reference data: For Ni +1/202 NiO AG' -244550 + 98.5 T, %3D whereAG°, is in joules and T is in Kelvin.) 2. (20%) The electrochemical cell represented in accompanying diagram consists of two electrodes. One is a solid NiO/Ni mixture. The other is a gaseous Oz electrode The porous platinum coating acts only as catalyst. Porous NiO/Ni platinum Zro,+Y,0, (solid solution) O2 The electrolyte is a ZrO/Y;O; solid solution yhat conducts oxygen ions (0) between the oxygen side and the NiO/Ni side. (a) Calculate the equilibrium oxygen partial pressure at the NiO/Ni electrode (2Ni + O, 2NIO) at 1000 K. (b) Identify the cathode and the anode in this electrochemical cell and write the half-cell reactions for the two electrodes. (Specify the direction of the reaction.) (c) What is the open circuit voltage at 1000 K when the pressure of oxygen on the oxygen electrode side is one atmosphere? (d) What is the open circuit voltage at 1000 K when the oxygen pressure on the oxygen electrode side is 1010 atm? (Reference data: For Ni +1/202 NiO AG' -244550 + 98.5 T, %3D whereAG°, is in joules and T is in Kelvin.)
Expert Answer:
Answer rating: 100% (QA)
Standard free energy change for the reaction ZrO 2 Zr O 2 at 1727 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
At 850oC and 1.000 atm pressure, a gaseous mixture of carbon monoxide and carbon dioxide in equilibrium with solid carbon is 90.55% CO by mass. Calculate Kc for this reaction at 850oC. C(s)...
-
A solar cell has an open circuit voltage value of 0.60 V with a reverse saturation current density of Jo 5 4.11 3 1010 A/ft2. (a) For a temperature of 75oF, determine the load voltage at which the...
-
One mole of oxygen gas is at a pressure of 6.00 atm and a temperature of 27.0C. (a) If the gas is heated at constant volume until the pressure triples, what is the final temperature? (b) If the gas...
-
James has $15,000 in his saving account now. (a) What is the amount he deposited one year ago in his saving account if the account has a rate of return of 10%? (b) Calculate the earned amount of...
-
A laminated item is made up of six layers. The two outer layers each have a thickness of 1.25 0.10mm and the four inner layers each have a thickness of 0.80 0.05 mm. Assume the thicknesses of the...
-
In Problems 27-30 suppose that, for a certain exam, a teacher grades on a curve. It is known that the mean is 50 and the standard deviation is 5. There are 45 students in the class. What is the...
-
The cooling effect produced by refrigeration finds application in: (i) Construction of cold storages (ii) Cooling of concrete in dams (iii) Comfort air conditioning of hospitals (iv) Liquification of...
-
Refer to Exercises 2-16A and 2-17A. In Exercises 2-16A, Dr. Rebecca Gray opened a medical practice specializing in physical therapy. During the first month of operation (January), the business,...
-
Barco Kyan Company Company Barco Company Kyan Company Data from the current year-end balance sheets Assets Cash $18,500 $ 32,000 Accounts receivable, net 36,400 Merchandise inventory 84,940 Prepaid...
-
It is October 16, 2020, and you have just taken over the accounting work of China Moon Products, whose annual accounting period ends October 31. The company?s previous accountant journalized its...
-
Discuss the important ethical issues facing Yahoo as a corporation. In your response examine the importance of institutionalizing ethics within corporations and how this may be done.
-
What changes would you make to increase your productivity and be more efficient at the same time? What can you do to reduce waste and increase satisfaction? What barriers do you anticipate may...
-
What challenges are generally associated with remote working? 2) What benefits are generally associated with remote working? 3) Given your personal experience with online college courses and/or...
-
Anna received 100 shares of India Stock as a gift from her best friend on March 23, Year 2, when the fair market value was $42 per share. Her friend had purchased the stock on February 12, Year 1,...
-
How can nonfinancial managers contribute to an organization's financial viability? Please explain.
-
Idenfy at least three common bases for materiality and calculate the range of materiality for the current year for each base. (6 marks) b. Conclude on the most appropriate materiality and include a...
-
WHS Regulations state that Safety Data Sheets (SDS) must be available for any hazardous chemicals. What information must an SDS contain? Provide a brief outline of each point.
-
Determine the volume of the parallelepiped of Fig. 3.25 when (a) P = 4i 3j + 2k, Q = 2i 5j + k, and S = 7i + j k, (b) P = 5i j + 6k, Q = 2i + 3j + k, and S = 3i 2j + 4k. P
-
Three different compounds have the same molecular formula, C2H2F2; call them A, B, and C. Compound A has a dipole moment of 2.42 D, compound B has a dipole moment of 1.38 D, and compound C has a...
-
Briefly describe what it is that the Bragg equation relates?
-
A 175-g sample of a pure liquid, liquid A, with a density of 3.00 g/mL is mixed with a 50.0-mL sample of a pure liquid, liquid B, with a density of 2.00 g/mL. What is the total volume of the mixture?...
-
What is the difference between \(O M R\) and \(O C R\) ?
-
Now look at Fowler's work on design pattems. Look at the organizational pattern created by Fowler as a way to document the structure of a solution so that its use by others is optimized. and at the...
-
Meet with the person you interviewed in mini case 1 and present them with the design prototype you created. Get their feedback on the design. Do they like it? Could they navigate the pages? What...

Study smarter with the SolutionInn App


