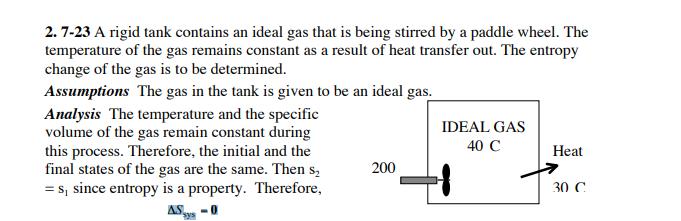
2. 7-23 A rigid tank contains an ideal gas that is being stirred by a paddle...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. 7-23 A rigid tank contains an ideal gas that is being stirred by a paddle wheel. The temperature of the gas remains constant as a result of heat transfer out. The entropy change of the gas is to be determined. Assumptions The gas in the tank is given to be an ideal gas. Analysis The temperature and the specific volume of the gas remain constant during this process. Therefore, the initial and the final states of the gas are the same. Then s, = s, since entropy is a property. Therefore, IDEAL GAS 40 C Heat 200 30 C AS -0 2. 7-23 A rigid tank contains an ideal gas that is being stirred by a paddle wheel. The temperature of the gas remains constant as a result of heat transfer out. The entropy change of the gas is to be determined. Assumptions The gas in the tank is given to be an ideal gas. Analysis The temperature and the specific volume of the gas remain constant during this process. Therefore, the initial and the final states of the gas are the same. Then s, = s, since entropy is a property. Therefore, IDEAL GAS 40 C Heat 200 30 C AS -0
Expert Answer:
Answer rating: 100% (QA)
Ans write the oelation fcr en tropy casincreaoe into the Ans write the e... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these mechanical engineering questions
-
A rigid tank contains an ideal gas at 300 kPa and 600 K. Now half of the gas is withdrawn from the tank and the gas is found at 100 kPa at the end of the process. Determine (a) the final temperature...
-
A rigid tank contains an ideal gas at 40°C that is being stirred by a paddle wheel. The paddle wheel does 200 kJ of work on the ideal gas. It is observed that the temperature of the ideal gas...
-
A pistoncylinder device contains an ideal gas. The gas undergoes two successive cooling processes by rejecting heat to the surroundings. First the gas is cooled at constant pressure until T2 = 34 T1....
-
Calculate and compare the efficiency of the two turbines of Prob. 14123. They should be the same since we are assuming dynamic similarity. However, the larger turbine will actually be slightly more...
-
Given an arrival process with l = 3.0, what is the probability that an arrival occurs in the first t = 2 time units?
-
The first ferris wheel ever built was created by Gale Ferris in 1893. The following chart shows the height of carts at various points of time. Time (min) Height (meters) 8 46 84 46 8 a) Write the...
-
Consider a $2^{2}$ factorial experiment with factors A and B. Show that $I N T(A, B)=I N T(B, A)$. That is, the interaction is symmetric in $\mathrm{B}$ and A.
-
Merck is considering launching a new drug called Laffolin. Merck has identied two possible demand scenarios: DEMAND LEVEL PROBABILITY 1 million patients.......30% 2 million patients........70% Merck...
-
XYZ Corp recorded the following Journal Entry in 2 0 1 9 : CIP 2 8 , 0 0 0 Construction Expenses 1 3 2 , 0 0 0 Revenue 1 6 0 , 0 0 0 The project is 4 5 % complete as of December 3 1 st , 2 0 1 9 ....
-
Presented below is information related to the sole proprietorship of Helen Archer, attorney. Legal service revenue2020 ...........$330,000 Total expenses2020 ........................211,000 Assets,...
-
Did paid-in capital increase or decrease from the prior year to the current year? How much? (10-K) kroger 2022 Why did paid-in capital increase or decrease during the year? (10-K)kroger 2022 Did...
-
1. Please describe clearly the application of the Library Information System diagram in a School library. Complete with examples of each subsystem in the those diagrams! 2. Look for some examples of...
-
Fill these out. Make sure when u get to the trial balance the numbers are the same when totaled. Also explain how you got transaction number 6 and 7 numbers. Trial Balance Assume this business...
-
customer defines the quality objectives for a firm and individuals decide if the objectives were met. Thinking of your experience as a customer, please describe a customer experience for a product or...
-
Properly classifying the variables that are part of a study is essential to making a correct analysis in SPSS. SPSS classifies variables as nominal, ordinal, or scale and requires you to explain any...
-
Journalize the following transactions of Cathcart Video Productions Inc.: April 19 Issued 2,000 common shares at $10 per share. July 22 Declared and paid a cash dividend of $0.60 per common share...
-
Question 4 (Mandatory) (1 point) Using the ICD-10-CM code book, identify the main term for the following diagnosis: Acute ST elevation myocardial infarction involving the inferolateral wall....
-
Difference between truncate & delete
-
Use a linear interpolation to estimate properties of ammonia to fill out the table below v [m/kg] x a) 550 b) 80 c) 0.75 20 10 0.4
-
A 400-L tank A, see figure P4.33, contains argon gas at 250 kPa, 30oC. Cylinder B, having a frictionless piston of such mass that a pressure of 150 kPa will float it, is initially empty. The valve is...
-
One means of air-conditioning hot summer air is by evaporative cooling, which is a process similar to the adiabatic saturation process. Consider outdoor ambient air at 35°C, 100 kPa, 30% relative...
-
Your boss gives you a sample of "gunk" and asks you to measure its viscosity. You do this in a cup and bob viscometer that has an outer (cup) diameter of 2 in., an inner (bob) diameter of $1.75...
-
The following data were obtained for a proprietary salad dressing tested at $22^{\circ} \mathrm{C}$ in a cup and bob viscometer (cup diameter $=4.2 \mathrm{~cm}$, bob diameter $=4.01 \mathrm{~cm}$,...
-
An inclined tube manometer with a reservoir is used to measure the pressure gradient in a large pipe carrying oil $(\mathrm{SG}=0.91)$ (see Figure $\mathrm{P} 4.5)$. The pipe is inclined at an angle...

Study smarter with the SolutionInn App


