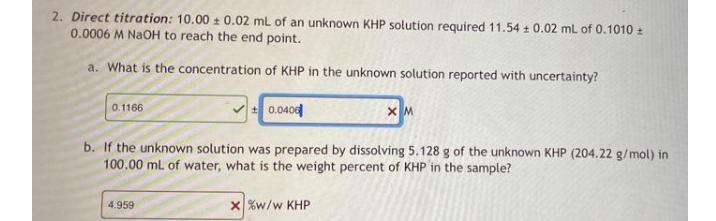
2. Direct titration: 10.00 + 0.02 mL of an unknown KHP solution required 11.54 + 0.02...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. Direct titration: 10.00 + 0.02 mL of an unknown KHP solution required 11.54 + 0.02 mL of 0.1010 0.0006 M NaOH to reach the end point. a. What is the concentration of KHP in the unknown solution reported with uncertainty? 0.040 0.1166 b. If the unknown solution was prepared by dissolving 5.128 g of the unknown KHP (204.22 g/mol) in 100.00 ml of water, what is the weight percent of KHP in the sample? x %w/w KHP 4.959 2. Direct titration: 10.00 + 0.02 mL of an unknown KHP solution required 11.54 + 0.02 mL of 0.1010 0.0006 M NaOH to reach the end point. a. What is the concentration of KHP in the unknown solution reported with uncertainty? 0.040 0.1166 b. If the unknown solution was prepared by dissolving 5.128 g of the unknown KHP (204.22 g/mol) in 100.00 ml of water, what is the weight percent of KHP in the sample? x %w/w KHP 4.959
Expert Answer:
Answer rating: 100% (QA)
Volume of unknown KHP solution V1 1000 002 mL volume of NaOH r... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Solution A was prepared by dissolving potassium acetate in methanol. Solution B was prepared by adding potassium methoxide to acetic acid. Reaction of methyl iodide either with solution A or with...
-
What is the concentration of Cu2+(aq) in a solution that was originally 0.015 M Cu(NO3)2 and 0.100 M NH3? The Cu2+ ion forms the complex ion Cu(NH3)42+. Its formation constant is given in Table 17.2.
-
A solution was prepared by dissolving 0.834 g of sulfur, S8, in 100.0 g of acetic acid, HC2H3O2. Calculate the freezing point and boiling point of the solution.
-
On January 1, 2018, the general ledger of Big Blast Fireworks includes the following account balances: The $30,000 beginning balance of inventory consists of 300 units, each costing $100. During...
-
Explain why pure liquids and solids can be ignored when writing the equilibrium-constant expression.
-
You toss a rock straight up. Compare the acceleration of the rock at the instant just after it leaves your hand with its acceleration at the instant just before it lands back in your hand, which has...
-
The pumper truck shown in Fig. P5.107 is to deliver \(1.5 \mathrm{ft}^{3} / \mathrm{s}\) to a maximum elevation of \(60 \mathrm{ft}\) above the hydrant. The pressure at the 4-in.-diameter outlet of...
-
Refer to the data in S10-8. Using only year-end figures rather than averages, compute the following for Ridgeview Employment Services: a. Net income b. Total liabilities c. Total assets (use the...
-
The growth rate for the firm's common stock is 7%. The firm's preferred stock is paying an annual dividend of $5. What is the preferred stock price if the required rate of return is 8%?
-
Why should one study operations management?
-
Problem 3- [4 points] Air is flowing at a Mach number of 2 and a pressure of 70 kPa in a two-dimensional channel. The upper wall turns towards the flow through an angle of 5 and the lower wall turns...
-
To build and run an Oracle statement, the following components can be utilized a. Statement b. Statement, PreparedStatement c. Statement, PreparedStatement, CallableStatement d. None of them
-
Think about a show you watch that has multiple episodes. How is suspense used to keep you engaged? What happens at the end of each episode that makes you want to watch the next one? Give a few...
-
Practice an upcoming presentation three timesat least once in the room where you will deliver it. Each time you practice, write notes about your observations and plans for improvements. Use the...
-
The relationship between three statement objects is: the is a subclass of the that is a subclass of the . a. CallableStatement, PreparedStatement, Statement b. Statement, CallableStatement,...
-
The database development process involves project planning, problem analysis, logical design, physical design, implementation and maintenance. (True/False)
-
Find the limit. (If the limit is infinite, enter 'o' or '-', as appropriate. If the limit does not otherwise exist, enter DNE.) lim (36t + 86t) t ||
-
The following information is for Montreal Gloves Inc. for the year 2020: Manufacturing costs Number of gloves manufactured Beginning inventory $ 3,016,700 311,000 pairs 0 pairs Sales in 2020 were...
-
Explain how a chemical equation can be used to relate the masses of different substances involved in a reaction.
-
There is often one step in a reaction mechanism that is rate-determining. What characteristic of such a step makes it rate-determining? Explain.
-
You discover that you cannot carry out a particular reaction for which you would like the enthalpy change. Does this mean that you will be unable to obtain this enthalpy change? Explain.
-
Consider the situation illustrated in Figure 25. 11. A positively charged particle is lifted against the uniform electric field of a negatively charged plate. Ignoring any gravitational interactions,...
-
A positively charged particle is moved from point A to point B in the electric field of the massive, stationary, positively charged object in Figure 25. 12. (a) Is the electrostatic work done on the...
-
Figure 25. 13 shows both the electric field lines and the equipotentials associated with the given charge distribution. (a) Is the potential at point A higher than, lower than, or the same as the...

Study smarter with the SolutionInn App


