Question: 2. Qualitative analysis studies require the use of a large number of chemical reagents, each of which must be treated with care. To illustrate

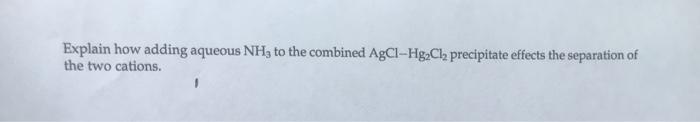
2. Qualitative analysis studies require the use of a large number of chemical reagents, each of which must be treated with care. To illustrate this statement, list the reagents used to determine and confirm the presence of Ag+ ion in a sample, and describe the safety problems associated with each. 3. Using your chemistry text or a dictionary as a reference, define the following terms as they pertain to this experiment. (1) qualitative analysis (2) precipitate (3) supernatant liquid gage Learning Explain how adding aqueous NH3 to the combined AgCl-HgCl precipitate effects the separation of the two cations.
Step by Step Solution
3.54 Rating (158 Votes )
There are 3 Steps involved in it
2 Reagents required for analysis of Ag are dil HCI NH3 KI KCrO4 and HNO3 Ag HCI AgCls AgCls 2NH3 AgN... View full answer

Get step-by-step solutions from verified subject matter experts


