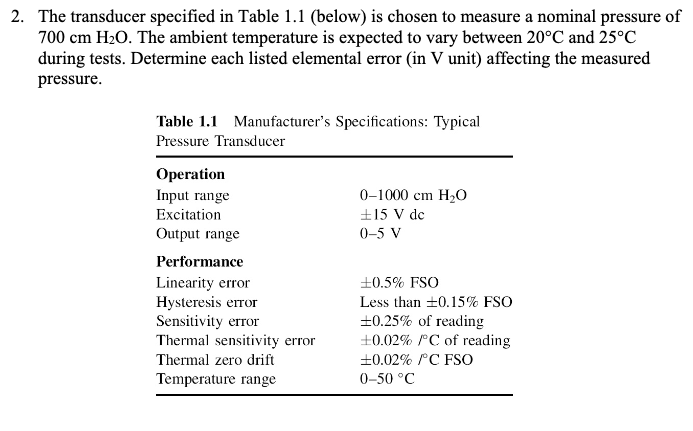
2. The transducer specified in Table 1.1 (below) is chosen to measure a nominal pressure of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. The transducer specified in Table 1.1 (below) is chosen to measure a nominal pressure of 700 cm HO. The ambient temperature is expected to vary between 20C and 25C during tests. Determine each listed elemental error (in V unit) affecting the measured pressure. Table 1.1 Manufacturer's Specifications: Typical Pressure Transducer Operation Input range Excitation Output range Performance Linearity error Hysteresis error Sensitivity error Thermal sensitivity error Thermal zero drift Temperature range 0-1000 cm HO 15 V dc 0-5 V 0.5% FSO Less than 0.15% FSO +0.25% of reading +0.02% /C of reading +0.02% /C FSO 0-50 C 2. The transducer specified in Table 1.1 (below) is chosen to measure a nominal pressure of 700 cm HO. The ambient temperature is expected to vary between 20C and 25C during tests. Determine each listed elemental error (in V unit) affecting the measured pressure. Table 1.1 Manufacturer's Specifications: Typical Pressure Transducer Operation Input range Excitation Output range Performance Linearity error Hysteresis error Sensitivity error Thermal sensitivity error Thermal zero drift Temperature range 0-1000 cm HO 15 V dc 0-5 V 0.5% FSO Less than 0.15% FSO +0.25% of reading +0.02% /C of reading +0.02% /C FSO 0-50 C
Expert Answer:
Answer rating: 100% (QA)
From the image provided you have a table of a manufacturers specifications for a typical pressure transducer The task is to determine the elemental er... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
A cylindrical tanker truck has a volume of 50 m3 when it is full. The driver can use a stick to find the depth of the contents. The following information is known: a. Find a cubic model to estimate...
-
A sieve-tray column, operating at a nominal pressure of 1 atm, is used to separate a mixture of acetone and methanol by extractive distillation using water. The column has 40 trays with a total...
-
In the accompanying Sales Transactions data, use Excel's filtering capability to extract a. all orders that used Paypal. b. all orders under $100. c. all orders that were over $100 and used a credit...
-
The Fed intervened heavily in the credit crisis. Write a short essay on whether you believe the Fed's intervention improved conditions in financial markets or made conditions worse.
-
Air flows in a 3-cm smooth tube at a rate of 0.015 m 3 /s. If T = 20C and p = 110 kPa absolute, what is the pressure drop per meter of length of tube?
-
Refer to the statements for Google in Appendix A. For the year ended December 31, 2015, what was its debt-to-equity ratio? What does this ratio tell us? Data From Statement Google In Appendix A...
-
On May 31, 2014, Terrell Company had a cash balance per books of $6,781.50. The bank statement from Home Town State Bank on that date showed a balance of $6,804.60. A comparison of the statement with...
-
The buyer at Ajax Co. is responsible for maintaining inventory of a key component which is used in the firm's manufacturing process throughout the entire 52-week year. The annual demand is 12,000...
-
The beginning inventory at Midnight Supplies and data on purchases and sales for a three month period ending March 31 are as follows: Date Transaction Number of Units Per Unit Total Jan. 1 Inventory...
-
Question 6 Ten thousand dollars is deposited in a savings account at 4.6% interest compounded continuously. How fast is the balance growing when it triples? (provide the appropriate units. 0.5 marks...
-
The city of Anville is currently home to 26000 people, and the population has been growing at a continuous rate of 5% per year. The city of Brinker is currently home to 25000 people, and the...
-
Emma Waterford is an engineer employed by Construct Pty Ltd. Emma's remuneration package includes: Salary of $90,000 per annum A company mobile phone valued at $1,800, used mostly for...
-
Liston, Incorporated had taxable income of $1 million for 2022. Compute Liston's regular tax liability.?
-
After making 30 payment of 498.47 on your car loan, you wanted to find out how much is left of your original 5 years loan at 4.1% compounded of 27000. What is the amount of the remaining balance of...
-
T corporation has 150 shareholders and was publicly traded. In 2022, the majority of T corporation stock was purchased by X company, The CEO for T corporation, Sam, was immediately asked to leave and...
-
Why is the adjusted R-squared used? a. Because it imposes a penalty for adding additional independent variables to a model. b. Because it is unbiased, unlike the R-squared measure. c. Because it is...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
Discuss how the method of Fair, Null, and Bolles [32] might be used to model the flow patterns in a rate-based model. How would the mole-fraction driving forces be computed?
-
In Example 10.4, temperatures of the gas and oil, as they pass through the absorber, increase substantially. This limits the extent of absorption. Repeat the calculations with a heat exchanger that...
-
Explain in detail, using thermodynamic principles, why the mixing of pure chemicals to form a homogeneous mixture is a so called spontaneous process, while the separation of that mixture into its...
-
In the previous two problems we found the posterior distribution of the slope of y on x, the rate of weevil infestation for endophyte infected and noninfected ryegrass. Let 1 be the slope for...
-
The moisture level of a dairy product is normally distributed with mean 15% and unknown variance 2 . A random sample of size 10 is taken and the moisture level measured. They are: 15.01 14.95 14.99...
-
The following 10 observations come from a simple linear regression model where the variance 2 = 3 2 is known. I 22 31 21 23 19 26 27 16 28 21 24.2 25.4 23.9 22.8 22.6 29.7 24.8 22.3 28.2 30.7 (a)...

Study smarter with the SolutionInn App


