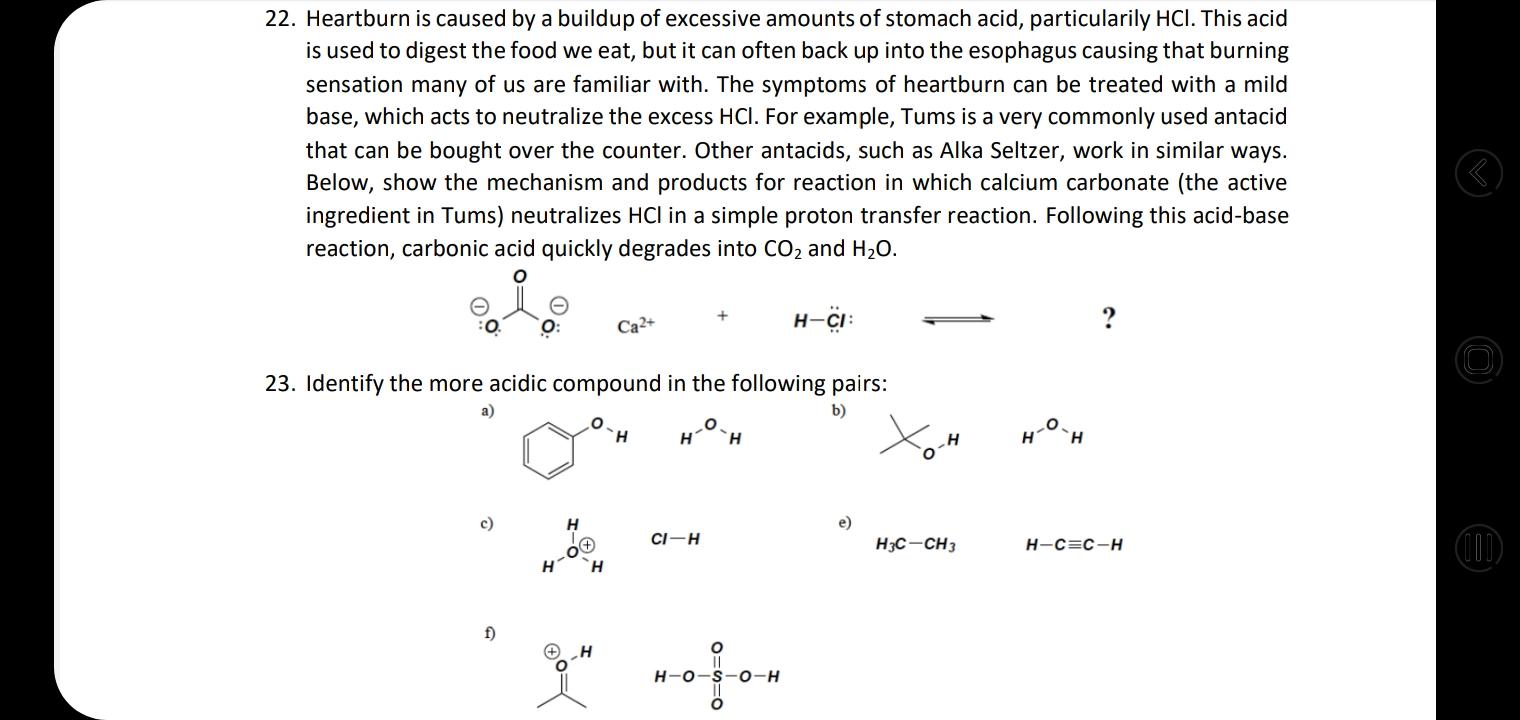
22. Heartburn is caused by a buildup of excessive amounts of stomach acid, particularily HCI. This...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
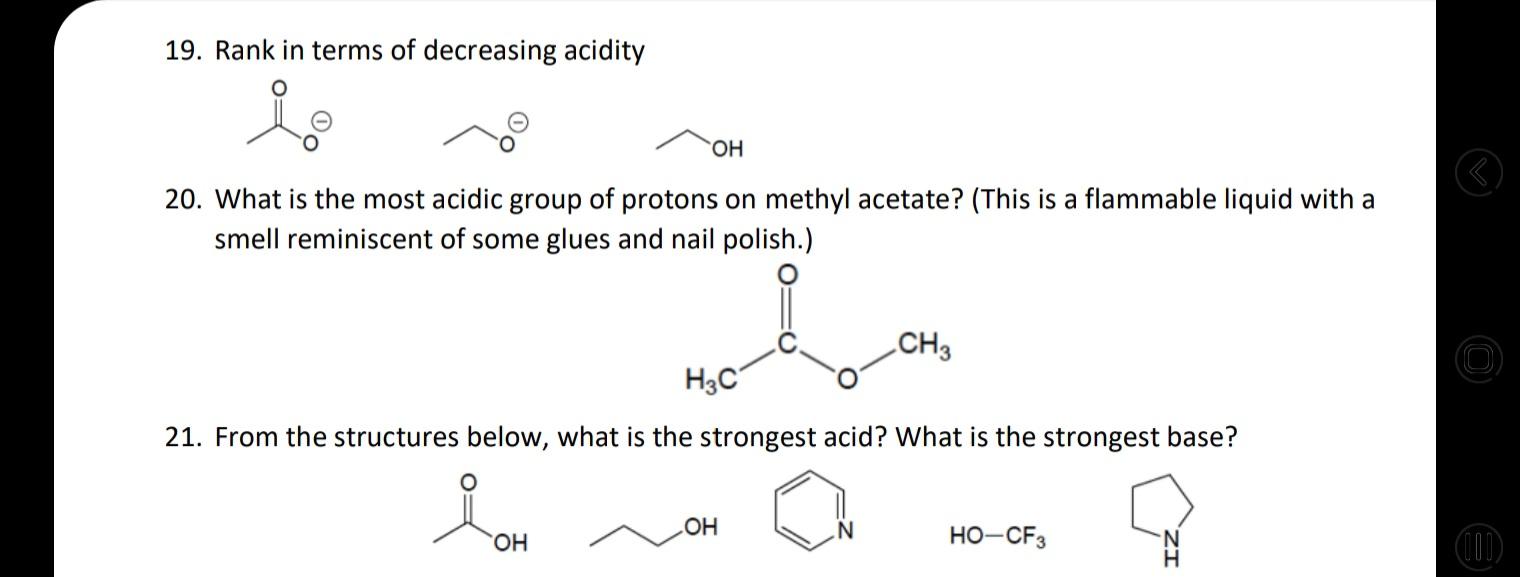
22. Heartburn is caused by a buildup of excessive amounts of stomach acid, particularily HCI. This acid is used to digest the food we eat, but it can often back up into the esophagus causing that burning sensation many of us are familiar with. The symptoms of heartburn can be treated with a mild base, which acts to neutralize the excess HCI. For example, Tums is a very commonly used antacid that can be bought over the counter. Other antacids, such as Alka Seltzer, work in similar ways. Below, show the mechanism and products for reaction in which calcium carbonate (the active ingredient in Tums) neutralizes HCI in a simple proton transfer reaction. Following this acid-base reaction, carbonic acid quickly degrades into CO2 and H20. Ca2+ H-ÇI: ? 23. Identify the more acidic compound in the following pairs: a) b) H. H. c) e) ((1) CI-H H3C-CH3 H-C=C-H H f) H-O 0-H 19. Rank in terms of decreasing acidity HO. 20. What is the most acidic group of protons on methyl acetate? (This is a flammable liquid with a smell reminiscent of some glues and nail polish.) CH3 H3C 21. From the structures below, what is the strongest acid? What is the strongest base? HO-CF3 (11) HO. H. 22. Heartburn is caused by a buildup of excessive amounts of stomach acid, particularily HCI. This acid is used to digest the food we eat, but it can often back up into the esophagus causing that burning sensation many of us are familiar with. The symptoms of heartburn can be treated with a mild base, which acts to neutralize the excess HCI. For example, Tums is a very commonly used antacid that can be bought over the counter. Other antacids, such as Alka Seltzer, work in similar ways. Below, show the mechanism and products for reaction in which calcium carbonate (the active ingredient in Tums) neutralizes HCI in a simple proton transfer reaction. Following this acid-base reaction, carbonic acid quickly degrades into CO2 and H20. Ca2+ H-ÇI: ? 23. Identify the more acidic compound in the following pairs: a) b) H. H. c) e) ((1) CI-H H3C-CH3 H-C=C-H H f) H-O 0-H 19. Rank in terms of decreasing acidity HO. 20. What is the most acidic group of protons on methyl acetate? (This is a flammable liquid with a smell reminiscent of some glues and nail polish.) CH3 H3C 21. From the structures below, what is the strongest acid? What is the strongest base? HO-CF3 (11) HO. H.
Expert Answer:

Related Book For 

Money, Banking, Financial Markets and Institutions
ISBN: 978-0538748575
1st edition
Authors: Michael Brandl
Posted Date:
Students also viewed these chemistry questions
-
Identify the more acidic compound in each of the following pairs: (a) CF3CH2CO2H or CF3CH2CH2CO2H (b) CH3CH2CH2CO2H or CH3CCCO2H
-
Identify the more acidic compound in each of the following pairs: (a) CF3CH2CO2H or CF3CH2CH2CO2H (b) CH3CH2CH2CO2H or CH3CPCCO2H (c) (d) (e) (f) (g) CO2H CO2H or CO,H CO2H o F- COH CO.H -CO-H or...
-
In grasshoppers, rosy body color is caused by a recessive mutation; the wild-type body color is green. If the gene for body color is on the X chromosome, what kind of progeny would be obtained from a...
-
Assume you are considering opening a retail business. You are trying to decide whether to have a traditional brick-and-mortar store or to sell only online. Explain how the activities and costs differ...
-
Consider two bonds with $1,000 face values that carry coupon rates of 8%, make annual coupon payments, and exhibit similar risk characteristics. The first bond has 5 years to maturity whereas the...
-
How is new technology impacting the TV industry?
-
For each of the following situations, calculate the \(z\)-statistic \((z)\), make a decision about the null hypothesis (reject, do not reject), and indicate the level of significance \((p>.05, p
-
Use the income statement for Hargrove Dynamics, Inc., for the past year ended December 31, 2011, and the information from the comparative balance sheets shown for the beginning and the end of the...
-
What is the return on an Investment that costs $500 today and will return $800 at the end of 4 years? What long will it take a $100 investment to double to $200 if the interest rate is 6%?
-
Dakota Company experienced the following events during Year 2: 1. Acquired $20,000 cash from the issue of common stock. 2. Paid $22,500 cash to purchase land. 3. Borrowed $8,000 cash. 4. Provided...
-
What is the difference between an exam and a test?
-
What are full-service supplier firms? What types of services do they provide?
-
Who are the top 3 firms in marketing research based on revenue? What is their revenue?
-
What is a multinational corporation?
-
Describe the difference between client-side and supply-side research. What other terms are used to refer to companies that are involved in supply-side research?
-
What effects do relative inflation rates have on relative interest rates?
-
2. a. List THREE (3) methods in which an interface designer can assist the use to manage user's attention. SIO NOTE: [NOTE: For each, draw and explain.] and explain.) din [6 marks] b. Information is...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
During the Reagan Administration in the 1980s, while the US government was running large government budget deficits, the rest of the world was also bringing large amounts of their savings to the...
-
If the ECB is pursuing an expansionary monetary policy, it will do which of the following? a. Lower the minimum reserve requirements b. Lower the interest rate paid by its deposit facility c....
-
Which of the following explain why exchange rates may overshoot? a. Incomes change more slowly than exchange rates. b. Exchange rates change more slowly than market prices of goods and services. c....
-
A running mountain lion can make a leap 10.0 m long, reaching a maximum height of 3.0 m. a. What is the speed of the mountain lion just as it leaves the ground? b. At what angle does it leave the...
-
Emily throws a soccer ball out of her dorm window to Allison, who is waiting below to catch it. If Emily throws the ball at an angle of 30 below horizontal with a speed of 12 m/s, how far from the...
-
In punting a football, the kicker tries to maximize both the distance of the kick and its hang timethe time that the ball is in the air. A kicker gets off a great punt with a hang time of 5.0 s that...

Study smarter with the SolutionInn App


