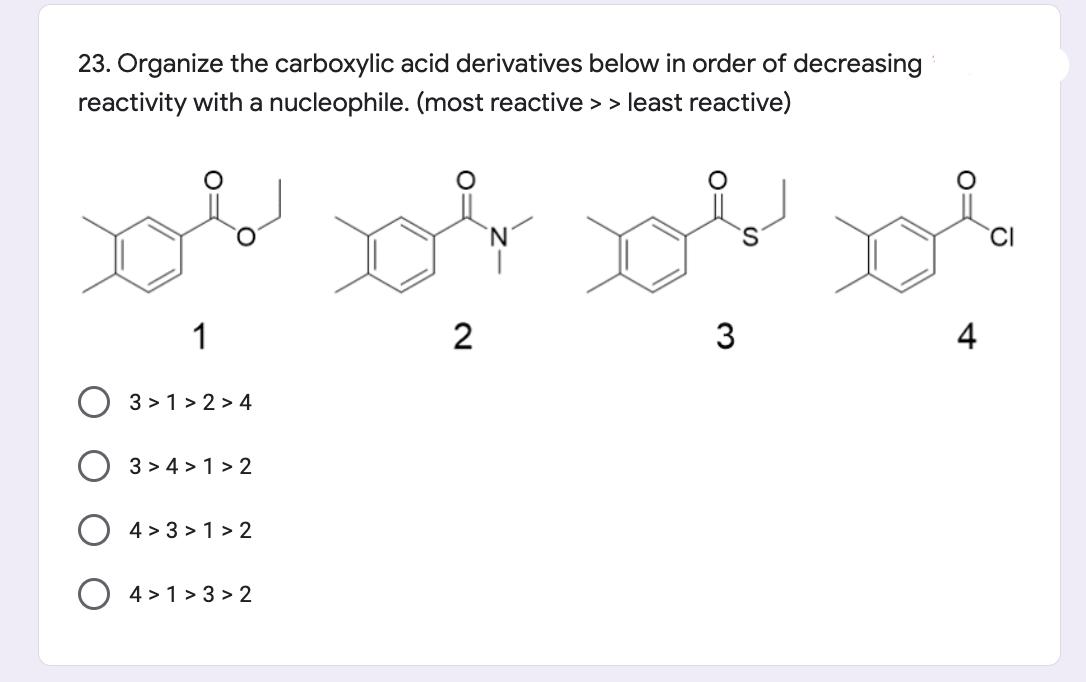
23. Organize the carboxylic acid derivatives below in order of decreasing reactivity with a nucleophile. (most...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
23. Organize the carboxylic acid derivatives below in order of decreasing reactivity with a nucleophile. (most reactive >> least reactive) CI 1 3 3 > 1 > 2 > 4 3 > 4 > 1 > 2 4 > 3 > 1 > 2 O 4 > 1 > 3 > 2 23. Organize the carboxylic acid derivatives below in order of decreasing reactivity with a nucleophile. (most reactive >> least reactive) CI 1 3 3 > 1 > 2 > 4 3 > 4 > 1 > 2 4 > 3 > 1 > 2 O 4 > 1 > 3 > 2
Expert Answer:
Answer rating: 100% (QA)
Among carboxylic acid derivatives like acid chlorides the hete... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Order these solutions in order of decreasing osmotic pressure, assuming an ideal van't Hoff factor: 0.1 M HCl, 0.1 M CaCl2, 0.05 M MgBr2, and 0.07 M Ga(C2H3O2)3
-
Rank the following compounds in order of decreasing reactivity in an E2 reaction CH3 CH3 CH3 Br Br CH3
-
List the following amides in order of decreasing reactivity toward acid-catalyzed hydrolysis: NO, CH CNH- CH CNH CH CNH NO2 CH,CNH
-
Carrefour issue its own credit card. The sales manager purchase clothes for his family for 900 using Carrefour credit card on the 1st of April. If Carrefour charge 2% per month on the amount the...
-
Due to a crisis in subprime lending, obtaining a mortgage has become difficult even for people with solid credit. In a report by the Associated Press (August 25, 2007), sales of existing homes fell...
-
What is your basic attitude toward the realities you face? Is your reality a gift or an obstacle, a friend or an enemy?
-
A company markets ethylene glycol antifreeze in halfgallon bottles. A machine fills and caps the bottles at a rate of 60 per minute. The \(68{ }^{\circ} \mathrm{F}\) ethylene glycol \(\left(ho=69.3...
-
Allred Shipping Co. acquired land, buildings, and equipment at a lump-sum price of $920,000. An appraisal of the assets at the time of acquisition disclosed the following values. Land . . . . . . . ....
-
The 18-year, $1,000 par value bonds of Waco Industries pay 7 percent interest annually. The market price of the bond is $895, and the market's required yield to maturity on a comparable-risk bond is...
-
For each information request below, formulate a single SQL query to produce the required information. In each case, you should display only the columns requested. Be sure that your queries do not...
-
Q2 For a given system: x+y+54z=110 27x+6y-z=85 6x+15y+2z=72|| (a) By using the Gauss- Jacobi method, solve the system to the 3rd iteration.
-
Find an algebraic expression equal to sec (sin (You may assume that < sin (5x 4) < ) Provide your answer below: sec(sin (5x = 4)) = (5x-4)).
-
What are some key factors in the development of the modern hospital in the United States? Is there one factor that stands out among the others through its importance?
-
6. Suppose the plates of a square parallel-plate capacitor are placed 2.0mm apart. If the area of each plate is 0.5cm, how many excess electrons will be on the negative plate if a voltage of 1.5V is...
-
Pierre is 24 years old and has always lived in Quebec. Upon graduation from Concordia last year, he landed a fantastic full-time job and decided to spend his pay cheques on buying a BMW for his first...
-
respond to this prompt individually using your opinion on what they are saying about their post .. i will attach the sample reply for you to follow. please and thank you .. basically, u have to do is...
-
Safari File Edit View History Bookmarks Window Help Candida... 109 Internsh... Question 1 ASSETS Accounts Receivable Prepaid Expenses Inventories Supplies Machinery and Equipment LIABILITIES Accounts...
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
Which is a stronger acid? a. CH3CH2CH2OH or CH3CH==CHOH b. c. CH3CH==CHCH2OH or CH3CH==CHOH d. HCCH2OH or CH COH CH CH2CH2NH or CH3CH CHNH3
-
When the following compound is hydrated in the presence of acid, the unreacted alkene is found to have retained the deuterium atoms: What does the preceding statement tell you about the mechanism of...
-
The hydrolysis of glycinamide is catalyzed by [Co(ethylenediamine)2]2+ Propose a mechanism for this reaction. Co2+ H2NCH2CNH2 H20
-
A valve has a failure rate of 0.06 failures per year. A periodic inspection performed once a year can detect 60% of the failures. The valve is operated for 10 years before it is removed from service...
-
An instrument has a failure rate of 0.015 failures per year. All failures are immediately detectable. The MTTR is 24 hours. What is steady state unavailability?
-
A failure report states that a safety transmitter signaled a high pressure in a vessel and initiated a trip. Other pressure measurements did not indicate any high pressure. A diagnostic in the safety...

Study smarter with the SolutionInn App


