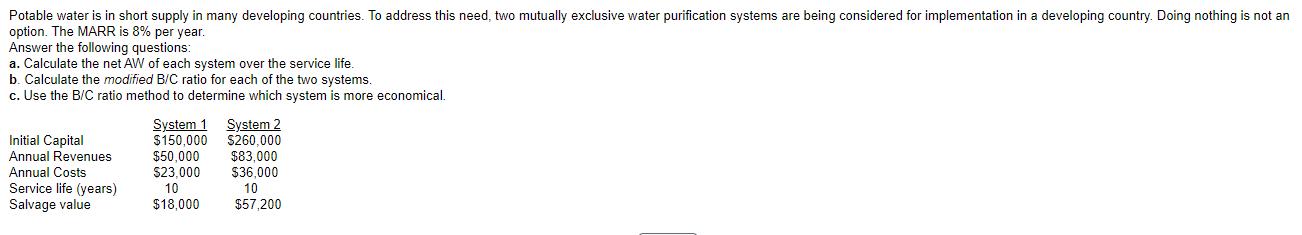
Potable water is in short supply in many developing countries. To address this need, two mutually...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
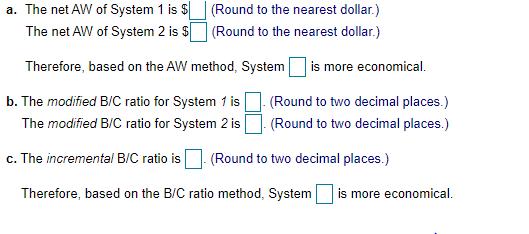
Potable water is in short supply in many developing countries. To address this need, two mutually exclusive water purification systems are being considered for implementation in a developing country. Doing nothing is not an option. The MARR is 8% per year. Answer the following questions: a. Calculate the net AW of each system over the service life. b. Calculate the modified B/C ratio for each of the two systems. c. Use the B/C ratio method to determine which system is more economical. Initial Capital Annual Revenues Annual Costs Service life (years) Salvage value System 1 System 2 $150,000 $260,000 $83,000 $36,000 10 $57,200 $50,000 $23.000 10 $18,000 a. The net AW of System 1 is $ The net AW of System 2 is $ (Round to the nearest dollar.) (Round to the nearest dollar.) Therefore, based on the AW method, System b. The modified B/C ratio for System 1 is The modified B/C ratio for System 2 is is more economical. (Round to two decimal places.) (Round to two decimal places.) c. The incremental B/C ratio is (Round to two decimal places.) Therefore, based on the B/C ratio method, System is more economical. Potable water is in short supply in many developing countries. To address this need, two mutually exclusive water purification systems are being considered for implementation in a developing country. Doing nothing is not an option. The MARR is 8% per year. Answer the following questions: a. Calculate the net AW of each system over the service life. b. Calculate the modified B/C ratio for each of the two systems. c. Use the B/C ratio method to determine which system is more economical. Initial Capital Annual Revenues Annual Costs Service life (years) Salvage value System 1 System 2 $150,000 $260,000 $83,000 $36,000 10 $57,200 $50,000 $23.000 10 $18,000 a. The net AW of System 1 is $ The net AW of System 2 is $ (Round to the nearest dollar.) (Round to the nearest dollar.) Therefore, based on the AW method, System b. The modified B/C ratio for System 1 is The modified B/C ratio for System 2 is is more economical. (Round to two decimal places.) (Round to two decimal places.) c. The incremental B/C ratio is (Round to two decimal places.) Therefore, based on the B/C ratio method, System is more economical.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these economics questions
-
In a double-slit experiment, it is found that blue light of wavelength 460nm gives a second-order maximum at a certain location on the screen. What wavelength of visible light would have a minimum at...
-
In a double-slit experiment it is found that blue light of wavelength 480 nm gives a second-order maximum at a certain location on the screen. What wavelength of visible light would have a minimum at...
-
It is found that a 6.00-m segment of a long string contains four complete waves and has a mass of 180 g. The string is vibrating sinusoidally with a frequency of 50.0 Hz and a peak-to-valley distance...
-
All administrative laws are criminal laws and enforceable as such even though they are not part of the Criminal Code. True False
-
Show that Y = a + bX, then +1 ifb >0
-
A finite-element model of a bar fixed at \(x=0\) at one end and having a mass \(m\) rigidly attached at \(x=L\) must satisfy what boundary condition?
-
The Plant Assets account of Star Media shows the following: Star Media sold plant assets at a \(\$ 10,000\) loss. Where on the statement of cash flows should Star. Media report the sale of plant...
-
Jarriot, Inc., presented two years of data for its Furniture Division and its Houseware Division. Furniture Division: Houseware Division: Required: 1. Compute the ROI and the margin and turnover...
-
2. Consider a closed economy to which the Keynesian-cross analysis applies. Consumption is given by the equation C = 200 + 2/3(YT). Planned investment, government spending and taxes all equal is 300....
-
Calculate the molar volume of saturated liquid and the molar volume of saturated vapor by the Redlich/Kwong equation for one of the following and compare results with values found by suitable...
-
Problem 3 (9 points). Let Find w = f(x, y, z) = sin(xy + x2), x = g(s, t) == w w s' t You may express these in terms of x, y, z, s, and t. and w z y=h(t, z) =t cos z.
-
Due to competition, Durango is considering lowering the unit sales price by 12% next year and increasing fixed marketing expenses by $60,000. If these changes are made, how many units must be sold...
-
What allows an accountant to match the applicable authoritative pronouncements to the accounting and auditing problems encountered? Research International Financial Reporting Standards (IFRS)...
-
Express in terms of j. -64
-
You heat 22.05 g of a solid in a test tube to 100C and add it to 50.00 g of water in a coffee-cup calorimeter with negligible heat capacity. The water temperature changes from 25.10C to 28.49C. Find...
-
A certain nearsighted person can only see objects sharply if they are no more than 1.25m in front of his corneas. What should be the focal length the contact lens that will allow him to see very...
-
use C++ Please don't use other tutors as your own work must have output for the programs Create a node class/struct. Create a queue class/struct. Members: Node - a node that tracks the front of the...
-
Les has collected stamps in his spare time for years. He purchased many of his stamps at a price much lower than the current market value. Les recently lost his job as a carpenter. Since his wife...
-
Coal and other fossil fuels usually contain sulfur compounds that produce sulfur dioxide, SO2, when burned. One possible way to remove the sulfur dioxide is to pass the combustion gases into a tower...
-
Consider the combustion (burning) of methane, CH4, in oxygen. CH4(g) + 2O2(g) CO2(g) + 2H2O(l) The heat of reaction at 25oC and 1.00 atm is 890.2 kJ. What is the change in volume when 1.00 mol CH4...
-
Is the following reaction spontaneous as written? Explain. Do whatever calculation is needed to answer the question. SO2(g) + H2(g) H2S(g) + O2(g)
-
What steps are necessary to record services performed on account for Wild Water Sports?
-
How does the QuickBooks Accountant software respond if a bill is entered with a vendor name that is not included on the vendor list?
-
What are the steps for paying sales tax?

Study smarter with the SolutionInn App


