2.4 mscf/d of the gas is compressed adiabatically and reversibly from 100 psi and 110F to...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
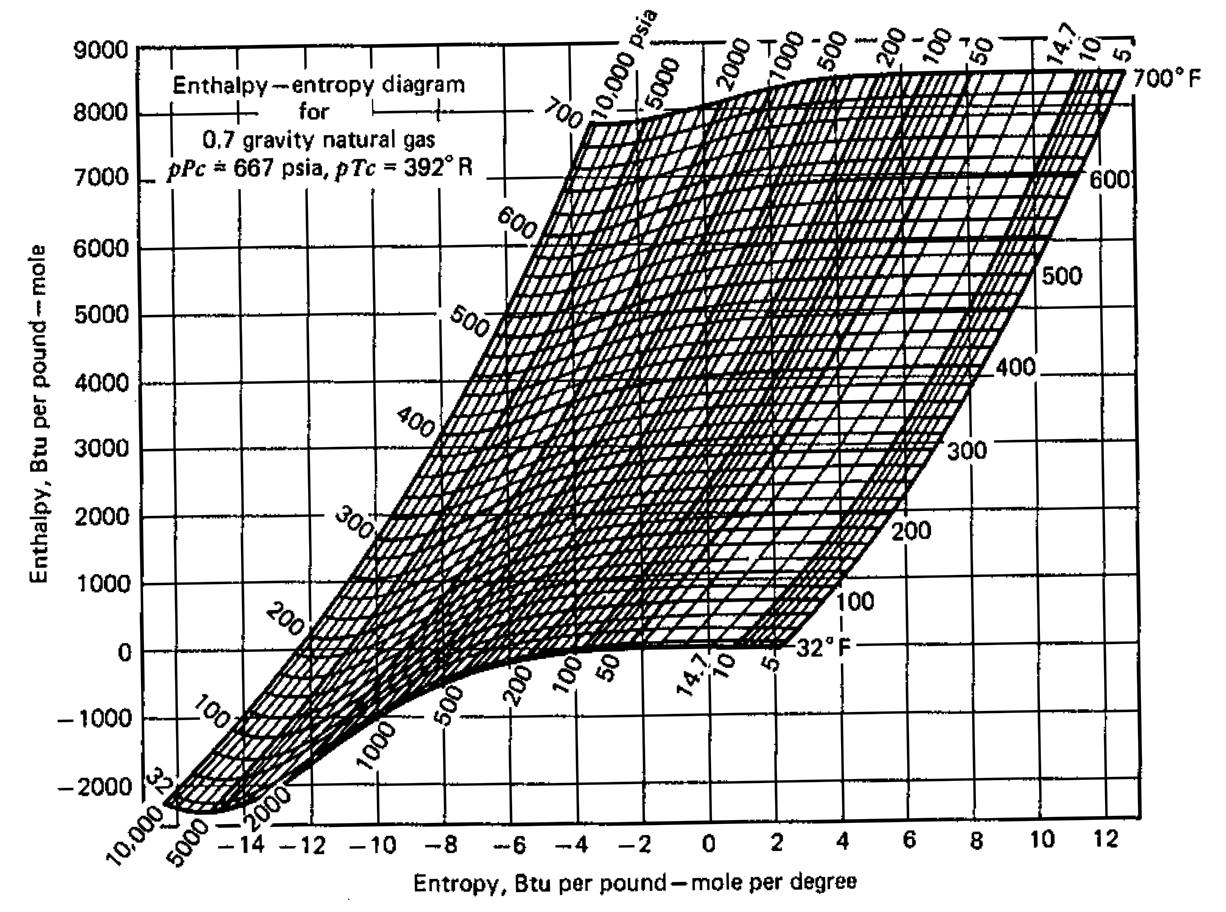
2.4 mscf/d of the gas is compressed adiabatically and reversibly from 100 psi and 110F to 1000 psi (i) What is the final temperature of this gas (ii) What is the energy requirement of this process (ii) What is the energy (in horsepower) requirement of the compressor 1 Horsepower = 2545 BTU/hr Use information provided at the end. Please explain how you calculate your answers. 700° F 9000 Enthalpy-entropy diagram for 700 8000 600. 0,7 gravity natural gas pPc * 667 psia, pTc = 392° R 7000 600 500 6000 500 400 5000 4000 400 300 3000 200 2000 100 1000 200 32° F 100 - 1000 10 12 -2000 4 8 2 -8 -6 -4 -2 - 14 -12 -10 Entropy, Btu per pound-mole per degree 200 00s 00 w00 00s 000 000, 0009 10,000 psia Enthalpy, Btu per pound -mole 2.4 mscf/d of the gas is compressed adiabatically and reversibly from 100 psi and 110F to 1000 psi (i) What is the final temperature of this gas (ii) What is the energy requirement of this process (ii) What is the energy (in horsepower) requirement of the compressor 1 Horsepower = 2545 BTU/hr Use information provided at the end. Please explain how you calculate your answers. 700° F 9000 Enthalpy-entropy diagram for 700 8000 600. 0,7 gravity natural gas pPc * 667 psia, pTc = 392° R 7000 600 500 6000 500 400 5000 4000 400 300 3000 200 2000 100 1000 200 32° F 100 - 1000 10 12 -2000 4 8 2 -8 -6 -4 -2 - 14 -12 -10 Entropy, Btu per pound-mole per degree 200 00s 00 w00 00s 000 000, 0009 10,000 psia Enthalpy, Btu per pound -mole
Expert Answer:
Answer rating: 100% (QA)
Given Initial pressure R10o psi 2 1000 psi Final ressure I psi 689476 Pa Also Init... View the full answer

Posted Date:
Students also viewed these physics questions
-
Please explain how you will be utilizing technology to make your business more effective and efficient. Explore the cloud technology concept. Discuss the importance of information technology planning...
-
Determine the specific volume (v) of the gas in a 1 m3 chamber filled with (a) Hydrogen (b) Carbon-dioxide. The pressure inside is 1 atm and the temperature is 25oC. Use the ideal gas (IG) state...
-
Refer to the case information provided at the end of Chapters 10 and 11 and the domain class diagram at the end of Chapter 9. Review and update your results from performing the tasks at the end of...
-
The jet plane starts from rest at s = 0 and is subjected to the acceleration shown. Determine the speed of the plane when it has traveled 1000 ft. Also, how much time is required for it to travel...
-
How is conceptual design different from detailed design?
-
Allison Company has always provided its customers with payment terms of 1/10, n/30. Members of its sale force have commented that competitors are offering customers 2/10, n/45. Explain what these...
-
The XYZ company sells approximately 900,000 gizmos annually. Each gizmo consists of 15 components. Management is currently evaluating which vendor to choose as the primary supplier of one of the...
-
On November 1, Essence Stores Inc. is considering leasing a building and purchasing the necessary equipment to operate a retail store. Alternatively, the company could use the funds to invest in...
-
Recently, Change Healthcare, a provider of payment cycle management and clinical information solutions, merged with Optum, a pharmacy benefit manager and subsidiary of UnitedHealth Group, in a...
-
Company A is a global company based in the United States that operates in the financial industry. Company A serves its customers with financial products, such as checking accounts, bank cards, and...
-
1. Readings: Read the assigned materials on change management, resistance, and related concepts. Familiarize yourself with the concept of employee resistance as a symptom, not the problem itself. 2....
-
PERFORMANCE GRAPH The following performance graph compares the cumulative five-year total return to shareholders on our common stock relative to the cumulative total returns of the S&P Health Care...
-
19. An article was sold for $212.00. The amount paid included a sales tax of 6%. Find the amount of sales tax on the article.
-
a) What are the benefits of private equity? What are the drawbacks? (2pts) Benefits of Private Equity - Higher Returns Potential: Private equity investments often aim for higher returns compared to...
-
Listening is important for a strong communication interaction. We can work on our own listening skills, but we can't always change the listening skills that others have. Have you ever been in a...
-
# 8 Sandra deposited $605 each month into her retirement, starting her senior year at FIT. She diversified her portfolio, so the interest rate averaged to a fixed rate of 9.75%. If she was 20 when...
-
don't do unlocking answers Compose total program that utilizes the capacities recorded underneath. Aside from the printOdd work, primary ought to print the outcomes after each capacity call to a...
-
What recommendations would you make to Big Four firms to help them (1) avoid confrontations with governmental officials in an authoritarian society and (2) deal effectively with such confrontations...
-
Derive Eq. (15-40a). Equation (15-40a) KPL.mol 1-C3XA.I.mol PL.mol NA,mol In C3=1+ C3 1-C3XA.bulk,mol. PS,mol
-
Derive the equation that is equivalent to Eqs. (15-32b) and (15-32c) in terms of a partial pressure driving force and a \(\log\) mean partial pressure difference: \[ \begin{equation*}...
-
Use the general solution in Perry's Chemical Engineers' Handbook (Wankat and Knaebel, 2019, p. 5-47) to solve the problem of a dissolving solid in a concentrated fluid using the known ratio of...

Study smarter with the SolutionInn App

