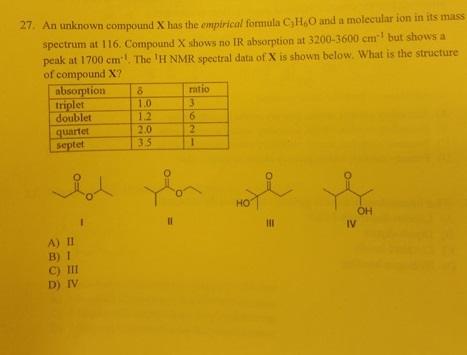
27. An unknown compound X has the empirical formula C,H,O and a molecular ion in its...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
27. An unknown compound X has the empirical formula C,H,O and a molecular ion in its mass spectrum at 116. Compound X shows no IR absorption at 3200-3600 cm¹ but shows a peak at 1700 cm. The 'H NMR spectral data of X is shown below. What is the structure of compound X? absorption triplet doublet quartet septet A) II B) 1 C) III D) IV 8 1.0 1.2 2.0 35 11 ratio 3 (6/2) 1 HO 111 OH IV 27. An unknown compound X has the empirical formula C,H,O and a molecular ion in its mass spectrum at 116. Compound X shows no IR absorption at 3200-3600 cm¹ but shows a peak at 1700 cm. The 'H NMR spectral data of X is shown below. What is the structure of compound X? absorption triplet doublet quartet septet A) II B) 1 C) III D) IV 8 1.0 1.2 2.0 35 11 ratio 3 (6/2) 1 HO 111 OH IV
Expert Answer:
Answer rating: 100% (QA)
21 Empirical formula C3H60 mas of the empirical formula 58 and since ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Treatment of compound D with LIAIH4 followed by H20 forms compound E. D shows a molecular ion in its mass spectrum at m/z 71 and IR absorptions at 3600 - 3200 and 2263 cm-1. E shows a molecular ion...
-
A compound A has a strong, broad IR absorption at 3200-3500 cm-1 and the proton NMR spectrum shown in Fig. P13.48a. Treatment of compound A with H2SO4 gives compound B, which has the NMR spectrum...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Write a program that plots average path length versus the number of random edges as random shortcuts are added to a 2-ring graph on 1,000 vertices.
-
On its recent financial statements, Hassell Fine Foods reported the following information about net sales revenue and accounts receivable (amounts in thousands): According to its Form 10-K, Hassell...
-
On January 1, 2016, Martha Carnes, fresh out of college, contributed $10,000 for a 30 percent interest in an accounting partnership. The senior partner was not attentive to the work, and the first...
-
For the system in Problem 6.7, how many hours of sunlight are needed to ensure that the battery bank is at \(100 \%\) charge at the end of the day assuming the same load? Problem 6.7 A PV battery...
-
Albany Company calculated its return on investment as 10 percent. Sales are now $300,000, and the amount of total operating assets is $480,000. Required a. If expenses are reduced by $30,000 and...
-
There is an obvious direct correlation between the rise in the number of Americans without healthcare insurance and the volume of uncompensated care provided by the medical industry. The Health and...
-
The finance director of RM plc is considering several investment projects and has collected the following information about them. Projects D and E are mutually exclusive. The capital available for...
-
What is SOAPST? Another look at explicating a poem S 1. What is the Subject? (What) In a few words or phrases, identify the general topic, content, and Ideas. The subject is about her descending to...
-
What is the result of the following? A. 1 B. 2 C. 3 D. None of the above var list new ArrayList (); list.add("Austin"); list.add("Boston"); list.add ("San Francisco"); list.removeIf (a -> a.length()...
-
What is the result of running the following program? A. 6 B. X C. The code does not compile. D. The code compiles but throws a NullPointerException at runtime. E. The code compiles but throws a...
-
Which of these elements are in the output of the following? (Choose three.) A. sugar B. minnie C. snowball D. 1 E. 2 F. 3 var q = new ArrayDeque (); q. offerFirst("snowball"); q.offer ("sugar");...
-
How many of the following are legal declarations? A. One B. Two C. Three D. Four E. Five F. Six public void greek() { [][]String alpha; [] String beta; String [] [] gamma; String[] delta[]; String...
-
Which of the following references the first and last elements in a nonempty array? A. trains[0] and trains[trains.length] B. trains[0] and trains[trains.length - 1] C. trains[1] and...
-
How often is the lifecycle sustainment plant updated? A. Before in initial operation, test and evaluation. B. At each milestone decision. C. Prior to each major change to the product of strategy. D....
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
Give the major product expected when (a) Leucine is treated with p-toluenesulfonyl chloride (tosyl chloride). (b) Alanine is heated in methanol solvent with HCI catalyst.
-
An all-suprafacial [3,3] sigma tropic rearrangement could in principle take place through either a chair-like or a boat-like transition statel When the terpene germacrone is distilled under reduced...
-
Give the structure of every stereoisomer of I,2,3- trimethylcyclohexane. Label the enantiomeric pairs and show the plane of symmetry in each a chiral stereoisomer.
-
All prototypes must be ___________ in nature. (a) Evolutionary (b) Conceptual (c) Physical (d) None of these
-
_____________ is an iterative tool for project development which produces a live working model of the system. (a) Function (b) Prototype (c) Module (d) Class
-
It is the responsibility of __________ to build prototype with suitable tool. (a) End users (b) Systems Analyst (c) Programmers (d) Managers

Study smarter with the SolutionInn App


