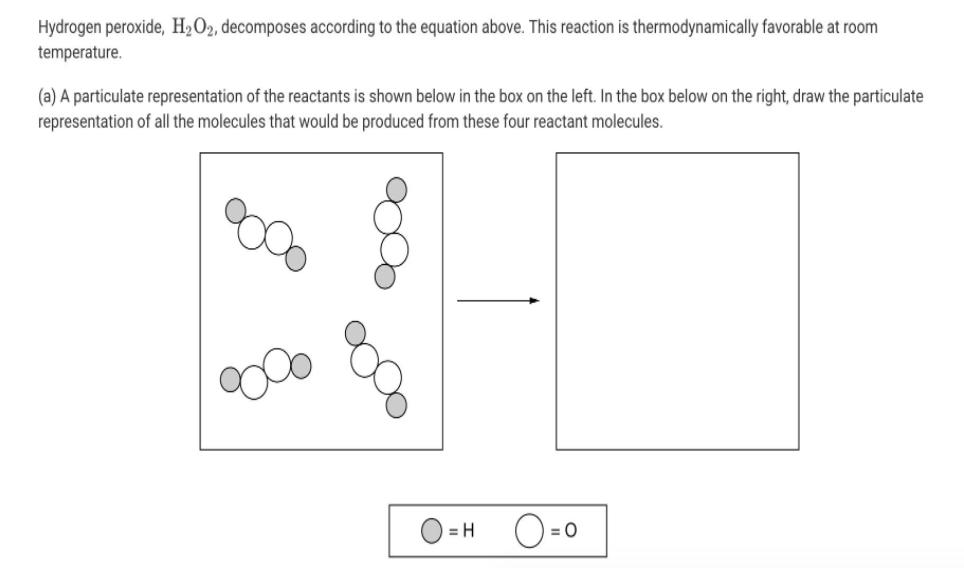
Hydrogen peroxide, H2O2, decomposes according to the equation above. This reaction is thermodynamically favorable at room...
Fantastic news! We've Found the answer you've been seeking!
Question:
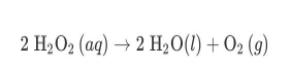
Transcribed Image Text:
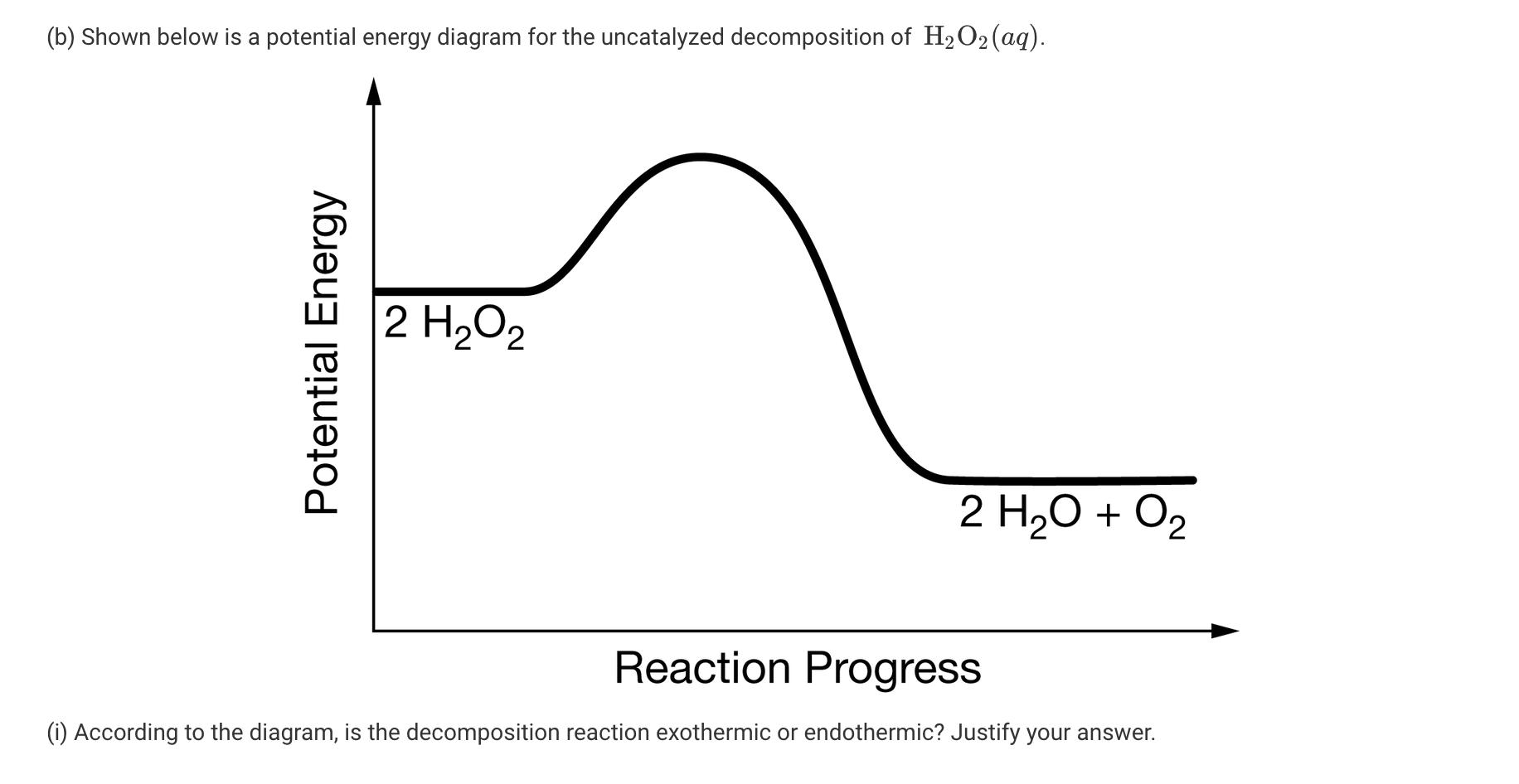
Hydrogen peroxide, H2O2, decomposes according to the equation above. This reaction is thermodynamically favorable at room temperature. (a) A particulate representation of the reactants is shown below in the box on the left. In the box below on the right, draw the particulate representation of all the molecules that would be produced from these four reactant molecules. O=0 = H 2 H2O2 (aq) → 2 H20(1) + 02 (9) (b) Shown below is a potential energy diagram for the uncatalyzed decomposition of H2O2 (aq). 2 H2O2 2 H20 + O2 Reaction Progress (i) According to the diagram, is the decomposition reaction exothermic or endothermic? Justify your answer. Potential Energy (ii) Manganese dioxide, MnO2 (s), is an insoluble substance that acts as a catalyst for the decomposition reaction. On the diagram above, draw a curve to represent the reaction as it occurs in the presence of MnO2 (s). 1 Upload files (PDF, JPG, GIF, PNG, TXT, Word, Excel, Powerpoint, file formats supported) 0/2 File Limit Hydrogen peroxide, H2O2, decomposes according to the equation above. This reaction is thermodynamically favorable at room temperature. (a) A particulate representation of the reactants is shown below in the box on the left. In the box below on the right, draw the particulate representation of all the molecules that would be produced from these four reactant molecules. O=0 = H 2 H2O2 (aq) → 2 H20(1) + 02 (9) (b) Shown below is a potential energy diagram for the uncatalyzed decomposition of H2O2 (aq). 2 H2O2 2 H20 + O2 Reaction Progress (i) According to the diagram, is the decomposition reaction exothermic or endothermic? Justify your answer. Potential Energy (ii) Manganese dioxide, MnO2 (s), is an insoluble substance that acts as a catalyst for the decomposition reaction. On the diagram above, draw a curve to represent the reaction as it occurs in the presence of MnO2 (s). 1 Upload files (PDF, JPG, GIF, PNG, TXT, Word, Excel, Powerpoint, file formats supported) 0/2 File Limit
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Hydrogen bromide dissociates when heated according to the equation The equilibrium constant Kc equals 1.6 Ã 102 at 200oC. What are the moles of substances in the equilibrium mixture at 200oC...
-
A sample of nitrosyl bromide (NOBr) decomposes according to the equation An equilibrium mixture in a 5.00-L vessel at 100 oC contains 3.22 g of NOBr, 3.08 g of NO, and 4.19 g of Br2. (a) Calculate...
-
A particle moves according to the equation x = 10 t 2 where x is in meters and t is in seconds (a) Find the average velocity for the time interval from 2.00 to 3.00 s. (b) Find the average velocity...
-
Imagine you discover a yeast mutant that exhibits a general inability to grow and thrive compared to wild-type yeast. You predict that the mutant has a defect in a basic process necessary for...
-
You find a container of 27 old pens in your school supplies and continue to test them (without replacement), until you find one that works. If each individual pen works 25% of the time (regardless of...
-
Ovit, Inc., has preferred stock with a price of $18.14 and a dividend of $1.26 per year. What is its dividend yield?
-
Define the following : (a) Indicated power (b) Brake power (c) Mechanical efficiency (d) Indicated and brake thermal efficiency (e) Relative efficiency
-
Bird-Bath, Inc., experienced four situations for its supplies. Compute the amounts that have been left blank for each situation. For situations 1 and 2, journalize the needed transaction. Consider...
-
Consider the spring assembly shown in Fig. ( a ) Obtain the global stiffness matrix for the full spring assembly. Take the determinant of the matrix you obtained ( you can do this using software, e ....
-
2. A double-spool turbofan engine is used to power an aircraft flying at speed of 250 m/s at an altitude of 11,000 m, where ambient temperature is -50C and pressure is 22.7 kPa. As shown below, the...
-
Three moles of a neen gas are at a temperature of 455 K. Calculate the average kinetic energy per atom, the root-mean-square (ms) speed of atoms in the gas, and the internal energy of the gas. HINT...
-
Le Co. paid $1,210,000 for the net assets of Chi Corp. At the time of the acquisition the following information was available related to Chi's balance sheet: Book Fair Value Value $ Current $500,000...
-
A carpenter uses roughly 15,000 sanding pads in one year. The set-up costs are $15.00 and carrying costs are estimated at 2%. Each pad costs $4.50. What is the economic order quantity? (Round decimal...
-
What does the budget constraint framework suggest when income rises? What does the budget constraint framework suggest when price changes? Include a brief explanation of what the results of income...
-
A bond issued by Google Corp. has a face value of $1000, matures in 4 years and has a 7 percent annual coupon. It currently sells at a yield to maturity of 8 percent. Calculate: (a) Current yield.
-
2. The stock price for Parkside Industries is currently $88.75. The company's PE ratio is expected to remain between 10 and 12 for the next five years. The current dividend is $4.28 per share...
-
11) You still live in the house below. It is a basic cape style. The house is 24'x36'. The front walls are 8' tall. And the peak of the house is 20' tall. Your furnace has just died and cannot be...
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
The breakdown of the ozone layer is promoted by the emission into the atmosphere of a. carbon dioxide from the burning of fossil fuels b. sulfuric acid from the burning of fossil fuels c. unburned...
-
The interaction responsible for the structures of molecules, liquids, and solids is the a. gravitational interaction b. strong interaction c. weak interaction d. electromagnetic interaction
-
The binding energy per nucleon in the chlorine isotope 3157 CI is 8.5 MeV. What is its atomic mass?
-
Why are none of the bulbs in Figure 31. 24 lit? Data from Figure 31. 24 Figure 31.24 (1) (iii) NNN
-
In Figure 31. 25, identify the energy conversions that occur between points \(A\) and \(B, B\) and \(\mathrm{C}, \mathrm{C}\) and \(\mathrm{D}\), and \(\mathrm{D}\) and \(\mathrm{A}\). Data from...
-
In Figure 31. 26, bulb B is brighter than bulb C, which in turn is brighter than bulb A. Rank, largest first, \((a)\) the magnitudes of the potential differences across the bulbs, \((b)\) the...

Study smarter with the SolutionInn App


