3. An equilibrium mixture contains 1.0 mol of iron metal, Fe(s); 1.0 x 10 mol of...
Fantastic news! We've Found the answer you've been seeking!
Question:
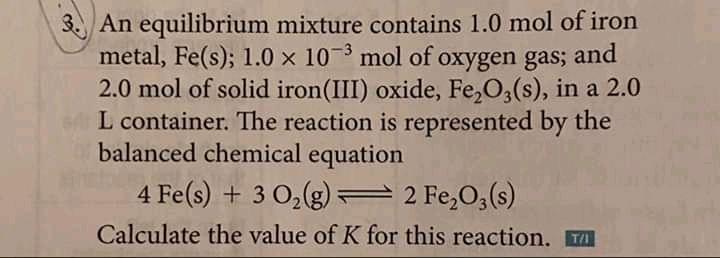
Transcribed Image Text:
3. An equilibrium mixture contains 1.0 mol of iron metal, Fe(s); 1.0 x 10 mol of oxygen gas; 2.0 mol of solid iron(III) oxide, Fe,O,(s), in a 2.0 L container. The reaction is represented by the balanced chemical equation and 4 Fe(s) + 3 0,(g) 2 Fe,O,(s) Calculate the value of K for this reaction. T 3. An equilibrium mixture contains 1.0 mol of iron metal, Fe(s); 1.0 x 10 mol of oxygen gas; 2.0 mol of solid iron(III) oxide, Fe,O,(s), in a 2.0 L container. The reaction is represented by the balanced chemical equation and 4 Fe(s) + 3 0,(g) 2 Fe,O,(s) Calculate the value of K for this reaction. T
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A gas mixture of 1 kmol carbon monoxide, 1 k mol nitrogen, and 1 k mol oxygen at 25C, 150 kPa, is heated in a constant pressure SSSF process. The exit mixture can be assumed to be in chemical...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
A metal cylinder with rigid walls contains 2.50 mol of oxygen gas. The gas is cooled until the pressure decreases to 30.0% of its original value. You can ignore the thermal contraction of the...
-
The comparative balance sheet of Merrick Equipment Co. for December 31, 20Y9 and 20Y8, is as follows: Dec. 31, 20Y9 Dec. 31, 20Y8 Assets Cash $70,720 $47,940 Accounts receivable (net) 207,230 188,190...
-
In the 1998 Minnesota gubernatorial election, there were three main candidates: Norm Coleman (the Republican), Jesse "The Body" Ventura (an Independent), and Hubert Humphrey (the Democrat). Although...
-
Boeing finances the sale of 20 Boeing jets to Lufthansa (C). Boeing is seeking short-term financing for its export sales to Germany. The Overseas Private Investment Corporation (OPIC) offers...
-
A & R Industrial Supply shows the following financial statement data for 2008, 2009, and 2010. Prior to issuing the 2010 statements, auditors found that the ending inventory for 2008 was understated...
-
Suppose that the market can be described by the following three sources of systematic risk with associated risk premiums. Factor Risk Premium Industrial production (I) ........ 6% Interest rates (R)...
-
Who or what groups are included in the micro and macroenvironments? Demographics: study the different generations and know what is important for each one. For example, which generation is the most...
-
Carl Kay is the vice-president of KM Ltd., a Canadian-controlled private corporation located in Halifax, Nova Scotia. KM operates a real estate development business constructing and selling...
-
* Q2:B/ The aim of calibration is to Meet customer requirement O Detect deterioration of accuracy O Comply with ISO standard requirements O Practice measurement procedures
-
Find the x- and y-intercepts of x-4y = -8. 4y=-8. Provide your answer below: X-intercept: V-intercept:
-
PRODUCT - DYSON V8 ORIGIN a. Identify the marketing strategies to promote our product. b. Justify your selections. c. Show how they will be used.
-
! Required information [The following information applies to the questions displayed below.] Stark company has the following adjusted accounts and normal balances at its December 31 year-end. Notes...
-
We analyzed the biceps muscle example with the angle between forearm and upper arm set at 90 (see figure below). Triceps muscle FE FB 12 Biceps muscle ma CG Wa 13 PHYSICS mb CG W Find the force...
-
Flounder Corporation had January 1 and December 31 balances as follows: 1/1/23 12/31/23 Inventory $90,000 $114,100 Accounts payable 60,400 69,800 For 2023, the cost of goods sold was $559,000....
-
1) The resultant of forces S1 and S2 is vertical and has an amplitude of 30 kN. Determine, a) S1 and S2 for a-45 and b) the value of a for which S, is minimum. (10 pt) S 30 S
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
Give the structural formula for a portion of the chain polymer obtained by the reaction of ethylene glycol, CH2OHO CH2OH with malonic acid, HOOCCH2COOH.
-
You want to make up 3.00 L of aqueous hydrochloric acid, HCl(aq), that has a pH of 2.00. How many grams of concentrated hydrochloric acid will you need? Concentrated hydrochloric acid contains 37.2...
-
The photoelectric work function for magnesium is 5.90 1019 J. (The work function is the minimum energy needed to eject an electron from the metal by irradiating it with light.) Calculate the minimum...
-
Albatross Plc, the Australian subsidiary of a British packaging company, is preparing its budget for the year to 30 June 2013. In respect of fuel oil consumption, it is desired to estimate an...
-
The following details show the direct labour requirements for the first six batches of a new product that were manufactured last month: The Management Accountant reported the following variances: The...
-
According to the Techleader blog on South Africas Mail and Guardian website, e-books are about to do to the publishing sector what iTunes did to the music industry. According to the blog post, e-book...

Study smarter with the SolutionInn App



