Question: 3. Enter True or False for every statement below regarding Propane, 2-Methylbutane, and 2,2-Dimethylpropane. These molecules have the same molecular formula These molecules are
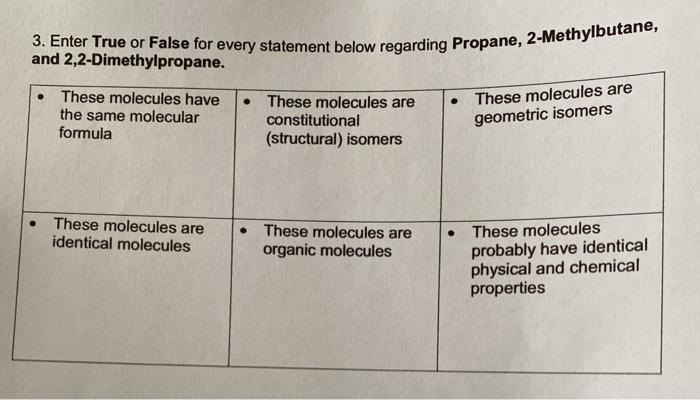

3. Enter True or False for every statement below regarding Propane, 2-Methylbutane, and 2,2-Dimethylpropane. These molecules have the same molecular formula These molecules are identical molecules These molecules are constitutional (structural) isomers These molecules are organic molecules . These molecules are geometric isomers These molecules probably have identical physical and chemical properties Pentane 2-Methylbutane 2,2-Dimethylpropane Complete the tables below for these three molecules. Pentane Alternate name (if Molecular formula given) 2-Methylbutane Alternate name Molecular formula (if given) 2-2-Dimethylpropane Alternate name (if Molecular formula given) Condensed structural formula Condensed structural formula Condensed structural formula Class of Organic Molecule Class of Organic Molecule Class of Organic Molecule
Step by Step Solution
3.47 Rating (163 Votes )
There are 3 Steps involved in it
Solution The physical properties of a class of ... View full answer

Get step-by-step solutions from verified subject matter experts


